Search
When Inflammatory Cytokines Are Unbalanced
Investigate the Role of Inflammation in Disease States Using Immunoassays
(See a complete list of products discussed in this article.)
As part of the immune response, inflammation plays an important role in defending the body against pathogens such as viruses, bacteria, fungi, and other parasites. However, inappropriate activation of inflammatory processes is an underlying contributor to many common pathological conditions. For example, autoimmune diseases arise when our immune system mistakes our cells or tissues for pathogens and attacks them. In addition, tumor proliferation and metastasis may occur when inflammatory cytokines create a microenvironment conducive to cancer progression. Figure 1 shows a simplified representation of cytokine interactions during inflammation.
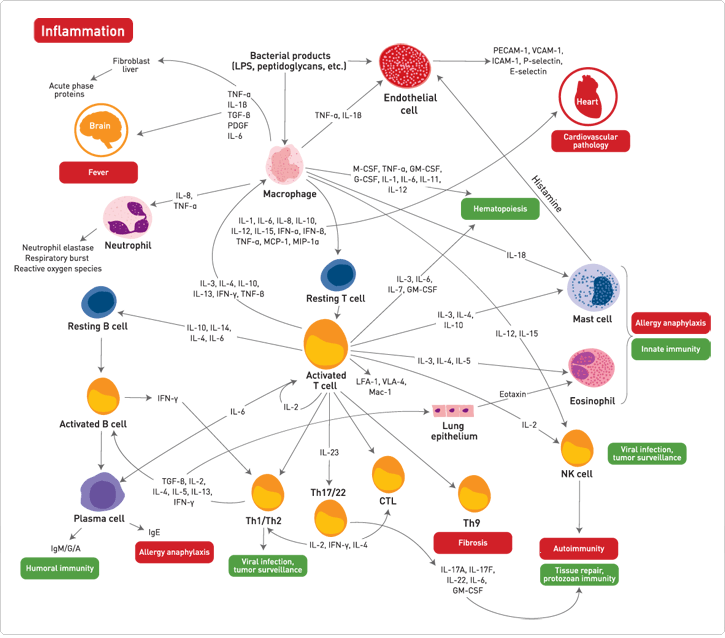
Figure 1. Simplified representation of cytokine interactions during inflammation.
Acute Versus Chronic Inflammation
Acute inflammation is a short-lived response that is characterized by extravasation of leukocytes, erythrocytes, and plasma components into the injured tissue. Left unchecked, the acute inflammatory process can lead to chronic inflammation.
Unlike acute inflammation, chronic inflammation is characterized primarily by tissue infiltration by lymphocytes and macrophages. Chronic inflammation is closely associated with allergy, atherosclerosis, cancer, arthritis, and Alzheimer’s disease, as well as autoimmune diseases. The process of acute inflammation is well defined, but the causes of chronic inflammation and its associated molecular and cellular pathways are still not well understood [1].
The Critical Balance Between Pro- and Anti‑inflammatory Mediators
The overall effect of an inflammatory response is dictated by the balance between pro- and anti-inflammatory mediators (Figure 2). Pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α are responsible for early responses and amplify inflammatory reactions, whereas anti-inflammatory cytokines, which include IL-4, IL-10, and IL-13, have the opposite effect in that they limit the inflammatory responses (Table 1). The increasing complexity of pro- and anti-inflammatory cytokine/chemokine networks has made it crucial to examine them in relevant functional groups rather than individually (Figure 1).
To facilitate the investigation of these pro- and anti-inflammatory cytokines, we have developed a large portfolio of single and multiplex assay kits designed to profile and quantitate levels of secreted cytokine proteins in body fluids such as plasma and serum, as well as in in vitro culture supernatants.
 | Figure 2. Inflammation is required for a healthy immune response, but it is also responsible for the pathogenesis of several diseases. The balance between pro- and anti-inflammatory cytokines plays a critical role in the body’s response to an inflammatory stimulus. |
Table 1. Pro- and anti-inflammatory mediators. From Bone et al. [2].
Pro-inflammatory mediators | ||
|---|---|---|
|
|
|
Anti-inflammatory mediators | ||
|
|
|
Multiplex Cytokine Assay Panels for the Luminex® Platform
The Novex® multiplex cytokine assay panels enable the accurate, reproducible, and sensitive profiling of multiple secreted cytokines, chemokines, and growth factors simultaneously in a single well using the Luminex® platform. Available for analysis of human, monkey, mouse, and rat proteins, these assay panels provide more data from each sample of plasma, serum, or tissue culture supernatant than do ELISAs, an important feature when sample size is limited.
Furthermore, the Novex® cytokine panels are available either with antibody-coated polystyrene capture beads or with the recently introduced antibody-coated magnetic capture beads, which facilitate automation, decrease hands-on time, and increase throughput and precision. We have used the Human Cytokine Magnetic 25-Plex Panel to analyze the cytokine and chemokine levels in seven different serum samples in a single experiment (Figure 3). Luminex® technology has become increasingly adopted for the savings it provides in time, cost, and sample volume, and it now has been cited in over 7,000 scientific publications. These Novex® assay panels are designed for use with the Luminex® 100⁄200™, FLEXMAP 3D®, and MAGPIX® systems.
 | Figure 3. Seven serum samples are analyzed in a single experiment using the Human Cytokine Magnetic 25-Plex Panel. Serum samples from seven different individuals were assayed with the Novex® Human Cytokine Magnetic 25-Plex Panel to determine the levels of 25 different cytokines and chemokines simultaneously. Measurements were performed using the Luminex® 200™ system. |
Singleplex Cytokine ELISA Kits
Novex® ELISA kits can be used for single-protein measurement to further evaluate individual cytokine targets. To supplement our colorimetric ELISA kits, we have added chemiluminescence-based ELISA kits for improved sensitivity and a broader dynamic range. These Novex® Chemi ELISA kits, which employ a chemiluminescent substrate as an indicator of antigen levels, undergo rigorous validation for sensitivity, specificity, precision, and lot-to-lot consistency.
The Whole Picture
Dissecting the complex process of inflammation requires a multiplex approach using tools that are designed to work together. Learn more about our Novex® immunoassay reagents and kits.
Ordering Information
| Product | Targets | Quantity | Cat. No. |
|---|---|---|---|
| Human Cytokine Magnetic 25-Plex Panel | Eotaxin, GM-CSF, IFN-α, IFN-γ, IL-1β, IL-1RA, IL-2, IL-2R, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12 (p40/p70), IL-13, IL-15, IL-17, IP-10, MCP‑1, MIG, MIP-1α, MIP-1β, RANTES, TNF-α | 100 tests | LHC0009M |
| Monkey Cytokine Magnetic 28-Plex Panel | EGF, eotaxin, FGF-basic, G-CSF, GM-CSF, HGF, IFN-γ, IL-1β, IL-1RA, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12, IL-15, IL-17, I-TAC, MCP-1, MDC, MIF, MIG, MIP-1α, MIP-1β, RANTES, TNF-α, VEGF | 100 tests | LPC0003M |
| Mouse Cytokine Magnetic 20-Plex Panel | FGF basic, GM-CSF, IFN-γ, IL-1α, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12 (p40/p70), IL-13, IL-17, IP-10, KC, MCP-1, MIG, MIP-1α, TNF-α, VEGF | 100 tests | LMC0006M |
| Rat Cytokine 10-Plex Panel | GM-CSF, IL-1α, IFN-γ, IL-1β, IL-2, IL-4, IL-6, IL-10, IL-12 (p40/p70), TNF-α | 100 tests | LRC0002 |
| Hu IL-1β Chemi ELISA Kit | Hu IL-1β | 96 tests | KHC0019 |
| Hu IL-6 Chemi ELISA Kit | Hu IL-6 | 96 tests | KHC0069 |
Resources | |
| Article Download Get a copy of this article as it appears in the print version of BioProbes 67. | Learn More About: |
For Research Use Only. Not for use in diagnostic procedures.