Search
Going Viral
Fluorescent Probes to Image Viruses and Their Host Cell Interactions
(See a complete list of products discussed in this article.)
By definition, the virus life cycle is intertwined with that of its host cell. These small infectious particles can only replicate from within a living cell, and they have devised strategies that allow them to infect organisms as simple as bacteria and as complex as plants and animals. Untangling the viral replication pathways from host cell functions is not a straightforward sorting process because viruses hijack cell machinery upon infection, creating cell conditions more conducive for virus production. Thus, the overarching goal of viral research is to understand the interactions between virus and host cell in the context of infection. Advances in imaging technologies, in combination with genetic and genomic approaches, have enabled the detailed characterization of viruses and their interactions with host cells. Given the growing number of selective probes, labeling technologies, and emission colors available, fluorescence assays are uniquely suited to this multifaceted analysis. We discuss an array of fluorescent probes that have been used in published studies, as well as more recently developed techniques that show promise for the advancement of viral research.
Visualizing Viral Entry
To investigate both the biology of viral entry and small molecules that hinder viral entrance into mammalian cells, researchers have conjugated the bright and photostable Alexa Fluor® dyes [1,2] and the pH-sensitive pHrodo™ dye to viral coat proteins, creating specific sensors of endocytosis (Figure 1). The pHrodo™ dye is nonfluorescent at neutral pH and fluoresces bright red in acidic environments such as endosomes.
CellLight® reagents—ready-to-use fluorescent protein constructs targeted to specific subcellular structures—are also important tools for analyzing viral entry and subsequent interactions with cell organelles. Alone or in multiplex studies with the reactive Alexa Fluor® or pHrodo™ dyes mentioned above, CellLight® fluorescent proteins can be used to label early or late endosomes, the plasma membrane, cytoskeletal proteins, and other organelles for colocalization studies with fluorescently labeled viral proteins.
CellTracker™ reagents are another option for labeling cells before or after viral infection [3]. These mildly thiol-reactive probes freely diffuse through membranes of live cells and form membrane-impermeant fluorescent adducts with glutathione and other intracellular proteins. Many cell types loaded with CellTracker™ probes are both fluorescent and viable for at least 24 hours and often through several cell divisions. Moreover, the fluorescent adducts survive aldehyde fixation, allowing subsequent immunocytochemical analysis while also facilitating the safe handling and analysis of cells containing pathogens.
 | Figure 1. Visualizing internalization of a BacMam particle with the pHrodo™ dye. CellLight® MAP4-GFP, a BacMam particle encoding the MAP4-GFP fusion, was labeled in serum-free buffer with pHrodo™ SE. U2OS cells were placed on a microscope stage inside a climate control chamber, and 50 µL of labeled BacMam particles (100 particles/cell) were added. (A) The pHrodo™ dye is shown in a differential interference contrast overlay. Red fluorescence from the labeled BacMam particles can be seen within intracellular vesicles. (B) Cells were imaged again the following day. Green-fluorescent microtubules labeled with CellLight® MAP4-GFP can be seen, as can pHrodo™ staining from the internalized BacMam particles. |
Imaging Viral and Host Transcription/Translation
Little is understood about the sites of viral RNA synthesis, including the transport and fate of nascent RNA inside and outside viral replication complexes. Click-iT® RNA assays allow detection of nascent RNA through the use of the alkyne-modified nucleoside EU (5-ethynyl uridine), which is fed to cells, incorporated into RNA during transcription, and detected with an Alexa Fluor® azide via a “click” reaction between the alkyne and azide. The Click-iT® RNA imaging and HCS kits provide advantages over traditional antibody-based BrU or BrUTP assays for studying viral RNA synthesis in infected cells: EU does not generally require a transfection reagent, and the small size of the Alexa Fluor® azide relative to the anti-BrdU antibody enables more effective detection of both ssRNA and dsRNA. After host transcription is inhibited (e.g., with actinomycin D), the levels of viral RNA synthesis and the subcellular localization of RNA can be measured using the Click-iT® RNA imaging and HCS kits, which should prove valuable in the study of viral gene expression and genome replication.
Many RNA viruses inhibit host transcription as a general strategy to evade antiviral responses, and this suppression can be evaluated with click chemistry–based assays. For instance, Kalveram et al. [4] recently used the Click-iT® RNA reagents to demonstrate that the Rift Valley fever virus (RVFV) NSs protein, which localizes to the nucleus and inhibits assembly of the basal transcription factor TFIIH, suppresses cellular RNA synthesis (Figure 2). This transcriptional suppression was visualized using fluorescence imaging and quantitated using flow cytometry analysis.
Click chemistry–based assays can also be employed for the detection of newly synthesized proteins. In this procedure, cellular proteins are metabolically labeled with L-azidohomoalanine, and then these newly synthesized proteins are detected using a click reaction with fluorescent azides. Groskreutz et al. [5] recently employed this technique to demonstrate that respiratory syncytial virus (RSV) infection does not suppress host protein translation, and thus as a consequence maintains viral protein translation.
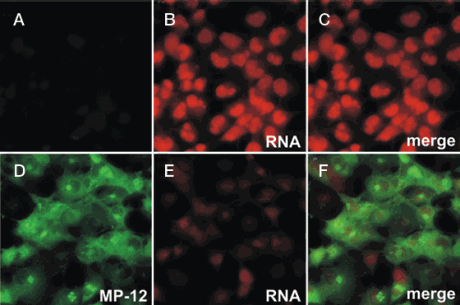 Figure 2. Rift Valley fever virus suppresses host transcription. 293 cells were either mock infected (A–C) or infected (D–E) with the MP-12 virus strain at an MOI of 3. (A, D) Viral protein expression was visualized by immunostaining with a polyclonal anti-RVFV antiserum followed by an Alexa Fluor® 488 dye–labeled secondary antibody (green). (B, E) Fifteen hours post-infection, cells were treated with 1 mM 5-ethynyl uridine (EU) for 1 hr, followed by fixation and permeabilization. Labeled RNA was detected by click chemistry with Alexa Fluor® 594 azide (red). (C, E) The labeled RNA image was overlaid on the corresponding labeled viral protein image. Figure reproduced with permission from the Journal of Virology [4] and Tetsuro Ikegami, University of Texas Medical Branch, Galveston, Texas. Figure 2. Rift Valley fever virus suppresses host transcription. 293 cells were either mock infected (A–C) or infected (D–E) with the MP-12 virus strain at an MOI of 3. (A, D) Viral protein expression was visualized by immunostaining with a polyclonal anti-RVFV antiserum followed by an Alexa Fluor® 488 dye–labeled secondary antibody (green). (B, E) Fifteen hours post-infection, cells were treated with 1 mM 5-ethynyl uridine (EU) for 1 hr, followed by fixation and permeabilization. Labeled RNA was detected by click chemistry with Alexa Fluor® 594 azide (red). (C, E) The labeled RNA image was overlaid on the corresponding labeled viral protein image. Figure reproduced with permission from the Journal of Virology [4] and Tetsuro Ikegami, University of Texas Medical Branch, Galveston, Texas. |
Tracking Viral Proteins in Live Cells
Studies of the localization and transport of viral proteins have been traditionally performed via the fusion of a fluorescent protein to one of the virus’ structural proteins. However, large protein tags can potentially disrupt viral protein function or replicative capacity, and segmented genomes such as influenza do not allow for fluorescent protein incorporation without compromising replication. Recent advances in imaging techniques have overcome these limitations through the use of biarsenical labeling: the introduction of a small tetracysteine (TC) tag, CCPCGG, which is specifically recognized by membrane-permeant biarsenical dyes that fluoresce upon binding to the TC motif. The existence of two dyes with distinct fluorescence emissions, FlAsH (green) and ReAsH (red), allows for dual labeling of existing and newly synthesized protein.
Biarsenical labeling is currently one of the most widely used methods to visualize newly synthesized viral proteins by imaging, and has been used in a plethora of recent studies investigating viral protein localization. For example, Das et al. [6] used this technique for imaging both existing and newly synthesized pools of the vesicular stomatitis virus (VSV) M protein (Figure 3), demonstrating that the M protein is transported from its site of cytoplasmic synthesis to the plasma membrane within 30 minutes. In similar approaches, Li et al. [7] used biarsenical labeling to visualize nuclear import of the influenza A NS1 protein, and Rudner et al. [8] used the technique to image the HIV Gag protein in live HeLa cells. Additionally, the HIV Gag protein was visualized with biarsenical dye staining in dendritic cells [9]. In this study, the FlAsH dye preferentially stained the cytosolic uncleaved Gag polyprotein, whereas monoclonal antibodies to Gag preferentially stained endocytosed, mature virions; using a combination of the two stains enabled distinct imaging of early versus late stages of the viral life cycle and trafficking between cells (Figure 4).
 | Figure 3. Imaging existing and nascent M protein of the vesicular stomatitis virus. BHK-21 cells infected with VSV-∆M-Mtc were treated with ReAsH-EDT2 for 30 min at 4 hr post-infection (A, D), washed, and then labeled immediately (B) or 1 hr later (E) with FlAsH-EDT2 for 30 min. Merged images of cells stained with ReAsH and FlAsH are shown in panels C and F. Figure reproduced with permission from the Journal of Virology [6] and Asit Pattnaik, University of Nebraska, Lincoln, Nebraska. |
 | Figure 4. HIV trafficking between dendritic cells. A synapse can be seen forming between a green-fluorescent, FlAsH-positive infected dendritic cell and an uninfected recipient, both stained with DAPI nuclear counterstain. Note the transfer of mature virus (red-fluorescent staining using anti-p24 (clone 183) that only detects mature HIV) to the neighboring uninfected recipient cell engaged in the synapse. Images are fluorescence overlays without (left) or with (right) differential interference contrast. Figure reprinted by permission from Macmillan Publishers Ltd on behalf of Cancer Research UK: Nature Methods [9], copyright 2008, and from Stuart Turville, The Kirby Institute, University of New South Wales, Australia. |
Detecting Virus-Mediated Cell Cycle Arrest
There is increasing evidence linking viral infection to cell cycle arrest, though the mechanisms of arrest vary widely between viruses [10]. The Click-iT® EdU cell proliferation assay is a powerful method to assess cell cycle arrest in virus-infected cells. Instead of the EU used in the Click-iT® RNA assays described above, the modified nucleoside EdU (5-ethynyl-2′-deoxyuridine) is incorporated during DNA synthesis and detected using an Alexa Fluor® azide in a click reaction. In a recent study, the Click-iT® EdU flow cytometry assay was used to characterize the impact of hepatitis C virus (HCV) infection on cell cycle in human hepatoma cells [11], demonstrating arrest between G2 and mitosis. Similarly, Wu et al. [12] used the Click-iT® EdU assay to investigate the interaction between human respiratory syncytial virus (HRSV) and the cell cycle in human airway epithelial cells, although arrest in this case occurred at the G0/G1 phase.
Measuring Influenza Neuraminidase Activity
Neuraminidase (NA) enzymes cleave the glycosidic linkages in neuramic acids and are expressed on the surface of the influenza viruses. NA inhibitors are commonly used as antiviral drugs targeting influenza, and recent attention has focused on the emergence of virus strains that are resistant to these inhibitors. The NA-Fluor®, NA-XTD™, and NA-Star® Influenza Neuraminidase Assay Kits provide fluorescent or chemiluminescent detection reagents for quantitating the sensitivity of influenza virus isolates to NA inhibitors (Figure 5). NA activity has been used as the basis for high-throughput screening assays to identify new antiviral compounds and drug-resistant influenza strains [13].
 | Figure 5. Detection of neuraminidase inhibitor resistance. A half-log dilution series of oseltamivir was incubated with four dilutions of influenza virus culture supernatants and assayed with the NA-Star® Influenza Neuraminidase Inhibitor Resistance Detection Kit. VR-1469 (influenza A/H1N1, strain A/PR/8/34, ATCC) and VR-1535 (influenza B, strain B/Lee/40) were cultured on MDCK cells. |
Advancing Virus Research
In addition to the tools mentioned here, we offer a myriad of kits for the assessment of cell health in response to viral infection, including assays for viability, apoptosis, and oxidative stress.
References
- Zhang SL, Tan HC, Hanson BJ et al. (2010) J Virol Methods 167:172–177.
- Kumari K, Gulati S, Smith DF et al. (2007) Virol J 4:42.
- Yang W, McCrae MA (2012) Arch Virol 157:305–314.
- Kalveram B, Lihoradova O, Ikegami T (2011) J Virol 85:6234–6243.
- Groskreutz DJ, Babor EC, Monick MM et al. (2010) J Biol Chem 285:24023–24031.
- Das SC, Panda D, Nayak D et al. (2009) J Virol 83:2611–2622.
- Li Y, Lu X, Li J et al. (2010) J Virol 84:7204–7213.
- Rudner L, Nydegger S, Coren LV et al. (2005) J Virol 79:4055–4065.
- Turville SG, Aravantinou M, Stössel H et al. (2008) Nat Methods 5:75–85.
- Davy C, Doorbar J (2007) Virology 368:219–226.
- Kannan RP, Hensley LL, Evers LE et al. (2011) J Virol 85:7989–8001.
- Wu W, Munday DC, Howell G et al. (2011) J Virol 85:10300–10309.
- Eichelberger MC, Hassantoufighi A, Wu M et al. (2008) Virol J 5:109.
Resources | |
| Article Download Get a copy of this article as it appears in the print version of BioProbes 67. | Learn More About: |
For Research Use Only. Not for use in diagnostic procedures.