Search
Gibco Hibernate Media

Hibernate media when supplemented with B-27 and GlutaMAX-I supplements allow for the manipulation of neurons at ambient CO2 for at least 48 hours while retaining their viability. Hibernate media can also be used to preserve viable brain tissue for up to a month when stored at 4°C and has also been shown to be a suitable transport media to ship various tissues and biological specimens including umbilical cord tissue. There are two types of Hibernate media, Hibernate-A and Hibernate-E, formulated to be used for post natal and embryonic neurons, respectively. These two products have similar formulation except for a difference in osmolality, Hibernate-A has a higher range of osmolality than Hibernate-E.
Taking your work to the clinic? Be sure to check out CTS Hibernate-A Medium and CTS Hibernate-E Medium
Survival of rat hippocampal and cortical neurons at ambient CO2 when cultured in Hibernate-E and B-27
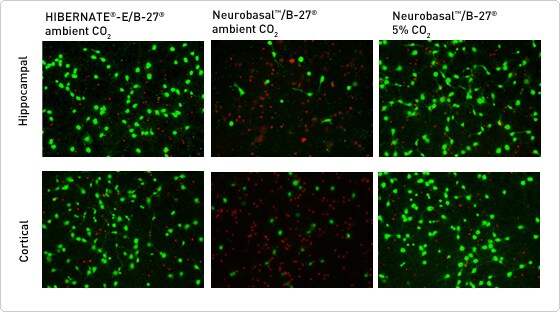
Figure 1. Live/Dead cell viability assay showing live cells stained with Calcein-AM (green) and dead cells with EthD-1 (red). The viability of primary rat hippocampal and cortical neurons maintained in HIBERNATE-E Medium at ambient conditions was equivalent to the viability of neurons in Neurobasal Medium in a standard 5% CO2 incubator. Corresponding cultures of neurons maintained in Neurobasal Medium at ambient CO2 conditions showed dramatic loss of viability at 48 hours.
Quantification of neuronal survival at 24 and 48h in Hibernate media
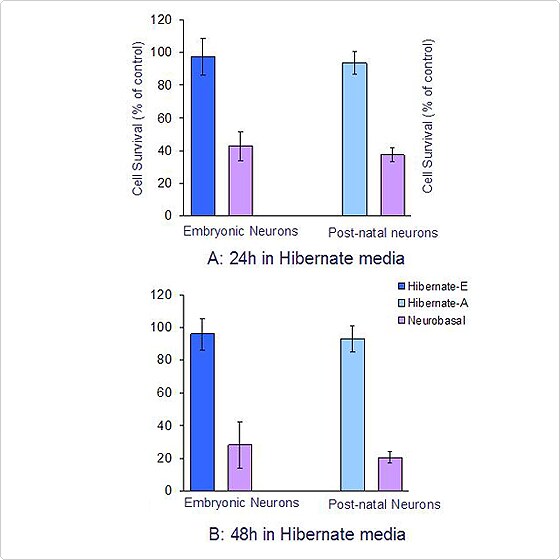
Figure 2. The viability of embryonic rat cortical neurons in Hibernate-E Medium, and post natal (P1) cortical neurons was maintained in Hibernate-A Medium at ambient conditions after 4 days in standard culture conditions. Cells maintained in Hibernate media at ambient CO2 showed less than 10% viability loss as compared to the control i.e., neurons in Neurobasal media in a 5% CO2 incubator. Neurons maintained in complete Neurobasal media at ambient CO2 conditions showed more than 70% viability loss by 48 hours. Calculated means and standard errors were tested for significance by ANOVA (n=3, p<0.001).
Assessment of physiological functionality when cells are maintained in Hibernate-E media
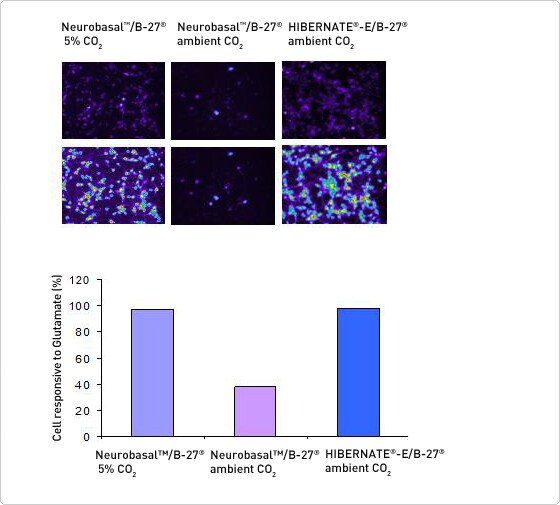
Figure 3. Glutamate-induced changes in intracellular calcium levels were measured with the Fluo-4 Direct Calcium Assay Kit. Pseudocolored images before (top) and after (bottom) challenge with 500 μM glutamate are shown. Purple represents low and red represents high intracellular calcium. The bar graph indicates that most neurons in Neurobasal Medium at 5% CO2 responded to glutamate (116/119), few neurons in Neurobasal Medium kept at ambient CO2 responded to glutamate (5/13) and most of the neurons kept at ambient CO2 in Hibernate-E Medium responded to glutamate (98/100).
Hibernate media can be used to transport tissues and biological specimens including umbilical cord tissue
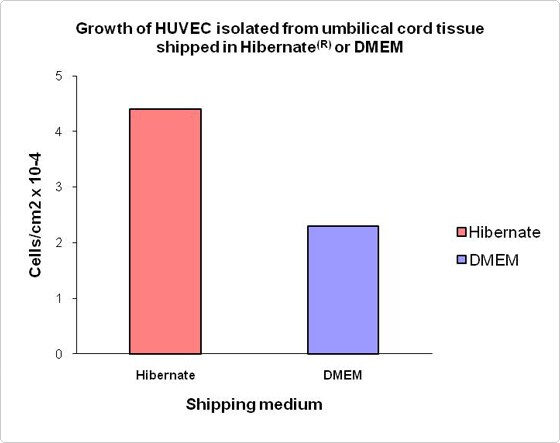
Figure 4. Growth of HUVECs from tissue shipped in Hibernate medium or DMEM. Freshly isolated human umbilical cords were cut in half. One half was shipped overnight in wet ice in Hibernate-E medium, while the other half was shipped in DMEM under the same conditions. Cords were unpacked, examined for any damage and then both halves were trimmed to equal lengths. HUVECs were isolated and cultured from each half by standard protocols using the same reagent, medium, and supplement. Cultures were grown until 80% confluent, then both sets were harvested and the cells were counted. The graph demonstrates that the growth of HUVEC from cords shipped in Hibernate-E medium is significantly better than growth of cells from cords shipped in DMEM.
Citations
- Brewer, GJ and Price, PJ (1996) Viable cultured neurons in ambient carbon dioxide and hibernation storage for a month. Neuroreports 7: 1509–1512.
- Brewer, GJ Isolation and culture of adult rat hippocampal neurons. (1997) J Neurosci Methods 71 14–155.
- Kivell, BM, McDonald, FJ, Miller, JH (2001) Method for serum-free culture of late fetal and early postnatal rat brainstem neurons. Brain Res Protoc 6: 91–99.
- Vieira, M, Christensen, BL, Feng, A.S., Kollmar, R.(2007) Survival and stimulation of neurite outgrowth in a serum-free culture of spiral ganglion neurons from adult mice. Hear Res 230:17–23.
- Brewer, GJ and LeRoux, PD (2007) Human primary brain tumor cell growth inhibition in serum-free medium optimized for neuron survival. Brain Res 1157:156–66.
- Williams, SN and Undieh, AS (2009) Dopamine D1-like receptor activation induces brain-derived neurotrophic factor protein expression. Neuroreport 20:606–10.
- Kotekar, AS (2008) Histone Modifications, but Not Nucleosomal Positioning, Correlate with Major Histocompatibility Complex Class I Promoter Activity in Different Tissues In Vivo. Mol Cell Biol 28: 7323–7336.
- Kelly, CM et al. (2007) Striatal graft projections are influenced by donor cell type and not the immunogenic background. Brain 130:1317–1329.
- Misgeld, T et al. (2005) Agrin promotes synaptic differentiation by counteracting an inhibitory effect of neurotransmitter. PNAS 102:11088–11093.
- Vukicevic, M and Kellenberger, S (2004) Modulatory effects of acid-sensing ion channels on action potential generation in hippocampal neurons. Am J Physiol Cell Physiol 287:C682–C690.
For Research Use Only. Not for use in diagnostic procedures.