Search
StemPro MSC SFM—Human Mesenchymal Stem Cell Culture Medium

StemPro MSC SFM—the first serum-free medium (SFM) specially formulated for the growth and expansion of human mesenchymal stem cells (hMSCs)
- Superior expansion efficiency
- Better quality hMSCs
- Better lot-to-lot consistency
First serum-free medium for hMSCs
- Superior efficiency of hMSC expansion at high cell densities—less medium, surface area, and time
- Better quality cells—primitive phenotype, retained hMSC surface marker expression, normal gene expression profiles, and maintained CFU-F and tri-lineage mesoderm differentiation potential beyond passage 5
- Batch-to-batch consistency with each lot—produced under cGMP and qualified using a hMSC performance assay
- No or little adaptation required from serum-supplemented medium
Superior hMSC expansion at high cell densities
The ability to scale up hMSC production will be critical for any viable cell therapy. StemPro MSC SFM enables hMSC scale up from 1 million to 1 billion cells using 85% less medium, 92% less surface area, 16% less time, 25% less passaging and less effort (less plates, passaging and no pre-qualification of FBS) versus classical medium (DMEM + 10% MSC-Qualified FBS).
Better quality hMSCs with a primitive phenotype
Human MSCs grown in StemPro MSC SFM exhibit a primitive morphology compared to control culture (Figure 1).
While hMSCs grown in classical medium have a flattened cell morphology and reach confluency between 1.0–3.0 x 104 cells/cm2, hMSCs grown in StemPro MSC SFM have a much smaller, spindle-shaped morphology and can reach densities >1.0 x 105 cells/cm2.
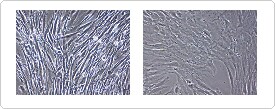
Figure 1. Morphology of hMSCs when grown in StemPro MSC SFM or classical medium (DMEM + 10% MSC-Qualified FBS.
Differentiation potential after 5 passages
hMSCs cultured in StemPro MSC SFM retained the tri-lineage mesoderm differentiation potential (adipocyte, chondrocyte, and osteoblast) beyond passage 5 (Figure 2).
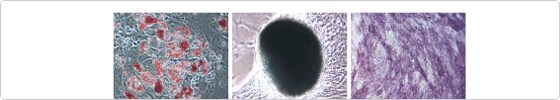
Figure 2. hMSC differentiation potential over long-term passaging in StemPro MSC SFM hMSCs cultured in StemPro MSC SFM (Passage 5) were seeded into adipogenic, chondrogenic, or osteogenic differentiation medium for 14 days, revealing adipocytes (Oil Red O–lipid stain), chondrocytes (Alcian Blue–glycosaminoglycan stain) and osteoblasts (Alkaline Phosphatase–cell surface glycoprotein stain)
CELLstart substrate for serum-free hMSC culture
CELLstart substrate is designed for use with StemPro MSC SFM to support hMSC attachment when growing hMSCs under serum-free conditions. CELLstart substrate is the first xeno-free substrate for stem cell culture, is produced under cGMP and offers consistent lot-to-lot performance.
For Research Use Only. Not for use in diagnostic procedures.