Search

Ribonuclease Inhibitors (RNase Inhibitors) |
Ribonuclease (RNase) inhibitors are recombinant enzymes used to inhibit RNase activity during experiments. RNase inhibitors are commonly used as a precautionary measure in enzymatic manipulations of RNA to inhibit and control for RNases. Small amounts of RNases can co-purify with isolated RNA, leading to compromised experimental results in downstream applications. Such contamination can also be introduced via tips, tubes, and other reagents used in procedures, which is why it is important to consider the use of RNase inhibitors in your experiments, especially if you are working with RNA.
What is an RNase inhibitor?
A ribonuclease inhibitor protein is a molecule that binds to and inactivates ribonucleases to protect RNA from degradation. It is composed of a series of leucine-rich repeats that form a horseshoe-shaped structure.
RNase inhibitors are recombinant enzymes that inhibit the activity of RNases. Since RNases are ubiquitous in the laboratory environment—on your skin, in the air, on anything touched by bare hands, or on anything left open to the air—it is important to make an effort to prevent or eliminate RNase contamination, which can be done with the use of RNase inhibitors.
How much RNase inhibitor to use?
RNase inhibitors are typically sold in units, and one unit (depending on the RNase inhibitor you use) is defined as the amount of protein to inhibit 5 ng RNase A.
Based on experimental design and the input amount of RNA, the amounts of RNase inhibitor to use will vary. A quick calculation will need to be made first to decide the amount required for your experiment. Note: What's in a unit? Most enzymes and proteins are sold based upon their unit activity. Looking closely at not only the manufacturer's unit definition, but more importantly at their unit assay, generates a better understanding of how the product will function in your specific application. Unfortunately, more often than not, the attempt to apply an established unit assay to your own system can lead to issues because unit assays are designed to quantitate a specific function of the product. Such is the case for ribonuclease inhibitors.
The traditional unit assay for ribonuclease inhibitors has been the cyclic cytidine monophosphate (CMP) assay. While this assay does demonstrate inhibition of RNase A, it is limited in that it provides only an indirect measure of RNA degradation. Because cCMP is an artificial substrate for RNase, its hydrolysis does not always correlate with actual RNA degradation. In addition, this assay can only be used for RNase A-type ribonucleases and their inhibitors because cCMP is not a substrate for many other ribonucleases.
In addition, some manufacturers define a unit via a functional assay that directly measures the ability of the inhibitor to block RNA degradation. A radiolabeled RNA is exposed to various RNases with and without the inhibitor. The results are analyzed on a polyacrylamide gel to determine the degree to which RNA degradation is inhibited. This functional assay provides direct information about the inhibitor's ability to block RNA degradation.
Types of nuclease inhibitors
Nuclease inhibitors encompass both RNase and DNase inhibition. We offer several nuclease (which includes DNase and RNase) inhibitors: Invitrogen SUPERase•In RNase Inhibitor, RNaseOUT Recombinant Ribonuclease Inhibitor, RNAsecure RNase Inactivation Reagent, and RNase Inhibitor (RI). See which inhibitor or reagent is right for your research by viewing the selection guide below:
| SUPERase•In RNase inhibitor | RNaseOUT recombinant ribonuclease inhibitor | RNAsecure RNase inactivation reagent | RNase Inhibitor (RI) | RNase Inhibitor (RI), cloned | |
 |  |  |  |  | |
| Mechanism | This is a protein-based inhibitor of non-human origin that non-covalently binds and inhibits the most common and troublesome RNases, including RNase A, B, C, 1, and T1 | This is a non-competitive inhibitor of pancreatic-type ribonucleases such as RNase A | This is a non-enzymatic reagent that will irreversibly inactivate RNases in solution | This is a 50 kDa recombinant enzyme used to inhibit RNase activity | This is a recombinant human protein produced in E. coli and inhibitor of neutral pancreatic RNase A type enzymes |
| Sizes available |
SUPERase•IN RNase inhibitor for RNase A, B, C, 1, and T1
SUPERase•In RNase inhibitor is a protein-based inhibitor of non-human origin that non-covalently binds and inhibits the most common and troublesome RNases including RNase A, B, C, 1, and T1. SUPERase•In can be used in any application where RNase contamination could be problematic. It is ideal for in vitro transcription and translation, cDNA synthesis, RT-PCR, and preparation of RNase-free antibodies. Because it inhibits a broader range of RNases than traditional RNase inhibitors, SUPERase•In is one of the most effective RNase inhibitors available, providing a higher level of protection against degradation (Figure 1). Main features of SUPERase•In RNase inhibitor include:
- Robust protein-based RNase inhibitor
- Inhibits RNases A, B, C, 1, and T1
- Remains active over a broad range of conditions—up to 65°C and from pH range 5.5–8.5
- Requires no DTT, but is functional in DTT concentrations as high as 200 mM DTT
- Retains activity in multiple freeze-thaw cycles
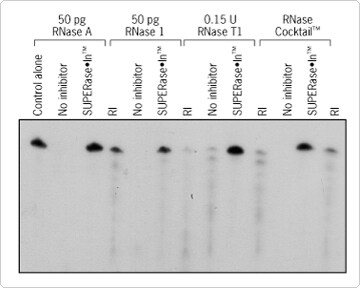
Figure 1. SUPERase•In vs. Placental Ribonuclease Inhibitor (RI). A 32P-labeled RNA probe was incubated for 30 minutes at 37°C in the presence of the indicated nucleases and either SUPERase•In or RI. Both the SUPERase•In and the RI were added at a concentration of 1 U/µl. Presence of signal indicates RNase activity.
RNaseOUT inhibitor for RNase A, B, and C
RNaseOUT recombinant ribonuclease inhibitor is purified by affinity chromatography from a recombinant strain of E. coli expressing a cloned porcine liver gene. This inhibitor has a very high binding affinity for pancreatic-type RNases, such as RNase A.
- Non-competitive, recombinant, protein-based RNase inhibitor
- Inhibits RNase A, B, and C
RNAsecure reagent for RNases A, B, and C
Traditionally, diethyl pyrocarbonate (DEPC) has been used to treat solutions that come into contact with RNA. DEPC treatment is time-consuming, possibly hazardous, and only eliminates RNases present at the time of DEPC treatment. In addition, some solutions (e.g., primary amine containing compounds such as Tris) cannot be treated with DEPC at all. Adding to that, DEPC must be inactivated by autoclaving post-treatment to prevent it from interfering with downstream enzymatic reactions. RNAsecure reagent eliminates these problems: RNAsecure can be used on virtually any solution and does not require post-treatment autoclaving. A unique feature of RNAsecure is that reheating after the initial treatment will reactivate the RNase-destroying agent to eliminate any new contaminants.
- Non-enzymatic reagent that irreversibly inactivates RNases in solutions that
- Cannot be treated with DEPC
- Cannot be autoclaved
- Contain enzymatic reactions prior to adding the enzyme
- Does not require post-treatment autoclaving
- Reheating after initial treatment reactivates the RNase-destroying agent to minimize new contamination
Choose from a variety of RNase Inhibitors (RIs)
Our selection of RNase inhibitors come in different unit sizes, from 1,000 to 10,000. Our RI features include:
- Recombinant enzyme or protein
- Inhibits RNases A, B, and C
- Does not inhibit RNase I, T1, T2, H, U1, U2, or CL3
- Requires a minimum of 1 mM DTT
- Effective pH range: 5.0–8.0
SUPERase•IN RNase inhibitor for RNase A, B, C, 1, and T1
SUPERase•In RNase inhibitor is a protein-based inhibitor of non-human origin that non-covalently binds and inhibits the most common and troublesome RNases including RNase A, B, C, 1, and T1. SUPERase•In can be used in any application where RNase contamination could be problematic. It is ideal for in vitro transcription and translation, cDNA synthesis, RT-PCR, and preparation of RNase-free antibodies. Because it inhibits a broader range of RNases than traditional RNase inhibitors, SUPERase•In is one of the most effective RNase inhibitors available, providing a higher level of protection against degradation (Figure 1). Main features of SUPERase•In RNase inhibitor include:
- Robust protein-based RNase inhibitor
- Inhibits RNases A, B, C, 1, and T1
- Remains active over a broad range of conditions—up to 65°C and from pH range 5.5–8.5
- Requires no DTT, but is functional in DTT concentrations as high as 200 mM DTT
- Retains activity in multiple freeze-thaw cycles

Figure 1. SUPERase•In vs. Placental Ribonuclease Inhibitor (RI). A 32P-labeled RNA probe was incubated for 30 minutes at 37°C in the presence of the indicated nucleases and either SUPERase•In or RI. Both the SUPERase•In and the RI were added at a concentration of 1 U/µl. Presence of signal indicates RNase activity.
RNaseOUT inhibitor for RNase A, B, and C
RNaseOUT recombinant ribonuclease inhibitor is purified by affinity chromatography from a recombinant strain of E. coli expressing a cloned porcine liver gene. This inhibitor has a very high binding affinity for pancreatic-type RNases, such as RNase A.
- Non-competitive, recombinant, protein-based RNase inhibitor
- Inhibits RNase A, B, and C
RNAsecure reagent for RNases A, B, and C
Traditionally, diethyl pyrocarbonate (DEPC) has been used to treat solutions that come into contact with RNA. DEPC treatment is time-consuming, possibly hazardous, and only eliminates RNases present at the time of DEPC treatment. In addition, some solutions (e.g., primary amine containing compounds such as Tris) cannot be treated with DEPC at all. Adding to that, DEPC must be inactivated by autoclaving post-treatment to prevent it from interfering with downstream enzymatic reactions. RNAsecure reagent eliminates these problems: RNAsecure can be used on virtually any solution and does not require post-treatment autoclaving. A unique feature of RNAsecure is that reheating after the initial treatment will reactivate the RNase-destroying agent to eliminate any new contaminants.
- Non-enzymatic reagent that irreversibly inactivates RNases in solutions that
- Cannot be treated with DEPC
- Cannot be autoclaved
- Contain enzymatic reactions prior to adding the enzyme
- Does not require post-treatment autoclaving
- Reheating after initial treatment reactivates the RNase-destroying agent to minimize new contamination
Choose from a variety of RNase Inhibitors (RIs)
Our selection of RNase inhibitors come in different unit sizes, from 1,000 to 10,000. Our RI features include:
- Recombinant enzyme or protein
- Inhibits RNases A, B, and C
- Does not inhibit RNase I, T1, T2, H, U1, U2, or CL3
- Requires a minimum of 1 mM DTT
- Effective pH range: 5.0–8.0
Technical bulletins
TechNotes articles
For Research Use Only. Not for use in diagnostic procedures.