Search
Fluorescence Microscopy Tools to Interrogate Internalization Pathways
In mammalian cells, as in other eukaryotes, the plasma membrane serves as an effective barrier separating the cell from the extracellular milieu. This membrane encloses the cell and protects it from invading pathogens. The presence of such an effective barrier means that specialized mechanisms have evolved to allow translocation of materials across the plasma membrane. These processes, including endocytosis, phagocytosis, and macropinocytosis, serve a highly diverse set of functions that regulate a plethora of cellular events, including nutrient acquisition, receptor desensitization, and antigen presentation. Paralleling the evolution of endocytotic processes, infectious agents, such as bacteria and viruses, have evolved ways to subvert the plasma membrane, thereby gaining access to the cell. This article describes fluorescence-based tools to interrogate routes of molecules and microbes—whether critical to cell health or pathogenic—across the plasma membrane, as well as their final destination after entering the cell.pHrodo™ Reagents
Molecules and pathogens that encounter the plasma membrane are funneled into membrane invaginations that subsequently bud inward to form endosomes (or phagosomes in the case of microbe internalization by macrophages). These intracellular structures are rapidly acidified, and this acidification is exploited by pHrodo™ reagents (Figure 1). The intensity of red emission of pHrodo™ dye is dependent on the ambient pH; a more acidic environment results in brighter emission. Conjugation of this dye to dextran or microbes has led to useful tools to study endocytosis and phagocytosis [1,2]. The reactive succinimidyl ester form of the dye (pHrodo™ SE) represents a versatile sensor that can be attached to a number of molecules, including tumor-targeting molecules such as avidin [3].
The reactive form of pHrodo™ dye can also be used to label whole cells, for example to monitor engulfment of apoptotic cells [4]. Conjugation of pHrodo™ dye to viruses holds great promise for investigating both the biology of viral entry and small molecules that hinder the entrance and invasion of viral species into mammalian cells. Figure 2 shows cells infected with BacMam 2.0 CellLight® MAP4-GFP virus labeled with pHrodo™ dye. Internalized BacMam particles can be seen inside a cell (Figure 2A). No detectable signal was seen in cells bathed in unlabeled virus or buffer containing pHrodo™ SE (data not shown). Imaging of the cells 24 hours later shows that the viruses retain biological activity, as the cells show strong Green Fluorescent Protein (GFP) fluorescence along microtubules originating from the virally expressed MAP4 (CellLight® MAP4-GFP, Figure 2B).
 | Figure 1. Tracking of internalization. When microorganisms or particles labeled with pHrodo™ dye are internalized by phagocytosis or endocytosis, acidification of the vesicles results in emission of fluorescence. |
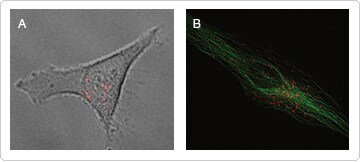
Figure 2. Visualizing internalization.CellLight® MAP4-GFP, a BacMam virus, was labeled in serum-free buffer with pHrodo™ SE. U20S cells were placed on a microscope stage inside a climate control chamber, and 50 µL of labeled virus (100 particles/cell) was added. A fluorescent image acquired with the appropriate filters (TRITC) for pHrodo™ dye is shown in a differential interference contrast overlay (A). Red fluorescence from the labeled viruses can be seen within intracellular vesicles. Cells were returned to the incubator overnight, and were imaged again the following day. Fluorescence from CellLight® MAP4-GFP (microtubules) can be seen, as can residual pHrodo™ staining from viral remnants (B), showing that labeled viruses retain functionality following pHrodo™ SE labeling.
CellLight® Reagents
The endosome is rarely the final destination for internalized molecules. Once inside the cell, molecules escape the endosomal compartment either by causing lysis of the endosome or through fusion with another organelle. Endosomal lysis releases cargo into the cytoplasm, whereas membrane fusion typically traffics cargo on to other organelles. These molecules will traverse the endocytotic pathway, trafficking either back to the plasma membrane or continuing on to either the lysosome or the Golgi apparatus. Fluorescence microscopy is an ideal method to easily and noninvasively monitor these highly dynamic trafficking events. The CellLight® reagents allow labeling of a number of intracellular organelles, including endosomes (Figures 3, 4), lysosomes, Golgi apparatus, ER, and peroxisomes (Figure 4A), with either GFP or Red Fluorescent Protein (RFP). CellLight® reagents use fluorescent proteins that survive fixation and permeabilization, and therefore they can be used in combination with any antibody. Life Technologies offers antibodies validated for imaging intracellular organelles such as the Golgi apparatus (Figure 4C), peroxisomes (Figure 4B), ER (Figure 4B), and recycling endosomes (Figure 4C). Labeling of transferrin, epidermal growth factor, or low-density lipoprotein with Alexa Fluor® dyes provides a useful means of visualizing ligand internalization.
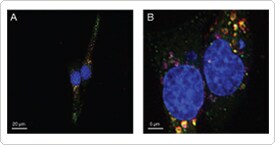
Figure 3. Combining markers for endocytosis. HeLa cells were transduced with CellLight® Early Endosome-GFP (green) and left overnight. The following day, cells were serum-starved in the presence of desferrioxamine for 24 hr. Cells were then incubated in Dulbecco’s phosphate-buffered saline containing 25 µg/mL Alexa Fluor® 647–labeled transferrin (red) and 20 µg/mL pHrodo™ dextran 10,000 MW (purple) for 5 min at 37°C to visualize endosomes, and with Hoechst 33342 to stain nuclei (blue). The cells were washed once and imaged.
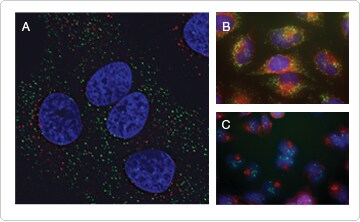
Figure 4. Demarcation of cellular structures. (A) Visualization of peroxisomes and endosomes in live HeLa cells. Cells were transduced with CellLight® Peroxisome-GFP (green). The following day, cells were incubated in 20 µg/mL pHrodo™ dextran 10,000 MW (red) for 20 min. (B, C) Cells were fixed and processed for immunocytochemistry using the SelectFX® Alexa Fluor® 488 Endoplasmic Reticulum Labeling Kit (the Alexa Fluor® 488 secondary antibody was replaced with Alexa Fluor® 555 goat anti–mouse IgG antibody) and the SelectFX® Alexa Fluor® 488 Peroxisome Labeling Kit (green) (B) or stained for the Golgi apparatus (red) and recycling endosomes (green) with antibodies against golgin-97 and Rab11 (C). Cells were stained with Hoechst 33342 (blue) to label nuclei.
Understanding Internalization
Visualizing how molecules that have been endocytosed are trafficked in the cell is a valuable approach to better understand their fate. pHrodo™ and CellLight® reagents are thus powerful tools for studying the roles of endocytosis in disease and health.
References
For Research Use Only. Not for use in diagnostic procedures.
- Learn more about pHrodo™ Indicators
- Find other Probes for Endocytosis
Get a copy of this article as it appears in the print version of BioProbes 64.
See a complete listing of the products discussed in this article.
