Search
Colorimetric ReadiLAMP Kit for Optimized SARS-CoV-2 Detection and Surveillance Using LAMP Assay

Invitrogen Colorimetric ReadiLAMP™ kit, SARS-CoV-2
- Detection is simple, intuitive and fast: colorimetric LAMP reaction changes color from purple to blue if SARS-target is present. No instrumentation is required for this assay, except for a 65℃ heat source.
- Utilizes non-invasive saliva samples or swab samples
- Ability to detect varying SARS-CoV-2 viral strains
- It can be utilized either of two ways – direct assay using crude samples or assay based on RNA purified from saliva and swabs.
- It allows input of up to 50% purified RNA per LAMP reaction to achieve maximal sensitivity in SARS-CoV-2 detection. See kit performance data.
- Process 1-96 samples at a time: Customers can process more samples with the same number of technicians, using individual tubes, tubes strips, or 96 well plates.
Product details for the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2
 Colorimetric ReadiLAMP™ kit, Colorimetric ReadiLAMP™ kit, 100 reactions |  Colorimetric ReadiLAMP™ kit, 1000 reactions | |
| Reactions | 100 reactions and 1000 reactions | |
| Test duration | ~30 mins – Direct LAMP assay ~1 hour – LAMP using purified RNA | |
| Sample types | Saliva and Swabs | |
| Detection | Colorimetric readout | |
| Sensitivity | Direct: ≤250 copies/25 uL LAMP reaction Purified: ≤100 copies/25 uL LAMP reaction | |
| Controls | Human Control Primer Mix and SARS-CoV-2 Control RNA | |
| Test format | 1-96 samples at a time | |
| Kit Contents | 100 Reactions | 1000 reactions |
| Kit storage temperature | –20℃ | |
| Instrument required | 65℃ heating block with heated lid | |
| Regulatory classification | RUO | |
| Catalogue Number | 100 reactions (Cat. No. A52539) or 1,000 reactions (Cat. No. A52544) | |
Why use the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2?
- Simplicity: Fast and simple protocol, easy to read results and robust system based on colorimetric indicators.
- Range of Samples types:
- Sample ranges from saliva, nasal swabs, nasopharyngeal swab, and other swabs.
- Sensitivity and variant coverage:The kit allows input of up to 50% purified RNA per LAMP reaction enabling maximal sensitivity in SARS-CoV-2 detection.
- Direct: ≤ 250 copies/25 µL LAMP reaction
- Purified: ≤ 100 copies/25 µL LAMP reaction
- Various SARS-CoV-2 variant coverage
- Speed to result and protocol flexibility – choose from 2 workflows:
- Direct LAMP protocol provides results in about 30 minutes using crude saliva or transport media samples.
- Purified RNA-based LAMP assay provides results in about 60 minutes: 30 minutes purification, followed by 30 minutes LAMP assay.
- Throughput
- Allows users to optimize their workflow for variable sample numbers (1-96 sample batches) and consumable types (individual tubes, strip tubes, 96-well plates) for efficient sample processing and rapid turn-around times.

Protocols and workflows for using the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2
Use this kit to either perform direct assay or purified assay. Either method yields quality results that are easy to interpret with blue being positive Sars-CoV-2 reactions and violet being negative results.

Direct Method: SARS-CoV-2 in crude saliva/NP samples

Indirect Method: SARS-CoV-2 detection workflow for purified RNA samples

Performance data for Invitrogen Colorimetric ReadiLAMP™ Kit, Sars-CoV-2
See the results of using our kit for various sample types and target infectious disease analytes. With color coded positive and negative results, interpreting the data is simple.

Figure 1. The Colorimetric ReadiLAMP Kit enables highly specific SARS-CoV-2 detection.
Samples from 12 unique nasopharyngeal donors (D1 – D12) were supplemented with γ-irradiated SARS-CoV-2 isolate USA-WA1/2020 (BEI Resources), Influenza A H1N1, or Influenza A H3N2 (ATCC) or 5.0 μL NATtrol™ Respiratory Pathogen Panel-1 or Panel-2 (RP1/RP2, Zeptometrix). RNA was purified using the MagMAX Viral/Pathogen II Nucleic Acid Isolation Kit and the Thermo Scientific KingFisher Flex Magnetic Particle Processor. The 25 μL Colorimetric ReadiLAMP reactions were incubated at 65 °C for 30 min. In all samples supplemented with SARS-CoV-2, a positive blue color change was observed, while all other virally supplemented samples and controls remained a negative purple color. [RP1 contains the following viral particles and bacteria: Influenza A H3N2 (Brisbane/10/07), Influenza A H1N1 (NY/02/2009), Rhinovirus (Type 1A), Adenovirus (Type 3), Parainfluenza (Type 1), Parainfluenza (Type 4), Metapneumovirus (Peru 6-2003), Chlamydia pneumoniae (CWL-029), Mycoplasma pneumoniae (M129), and Coxsackievirus (Type A1). Whereas RP2 contains the following viral particles and bacteria: from Influenza A H1 (New Caledonia/20/99), Influenza B (Florida/02/06), RSV (Type A), Parainfluenza (Type 2), Parainfluenza (Type 3), Coronavirus (HKU-1 recombinant), Coronavirus (OC43), Coronavirus (NL63), Coronavirus (229E), Bordetella pertussis (A639).]

Figure 2. The Colorimetric ReadiLAMP Kit efficiently detects multiple SARS-CoV-2 strains.
Fifteen strains of SARS-CoV-2 RNA were obtained and evaluated in the Colorimetric ReadiLAMP assay. Each 25 μL reaction included 2.5 x 101 – 1.0 x 104 copies of SAR-CoV-2 in a background of 50 ng human genomic DNA. After 65 °C incubation for 30 min, 80% of strains were detected at a sensitivity of 25 copies/ reaction with all strains being detected at 50 copies/ reaction. These detectable strains, obtained from ATCC, were Hong Kong (NR-52388), Italy (NR-52498), England (NR-52499), Singapore (NR-52501), Germany (NR-52502), USA-IL (NR-52503), USA-CA1 (NR-52504), USA-AZ (NR-52505), USA-WI (NR-52506), USA-CA3 (NR-52507), USA-CA4 (NR-52508), USA-CA2 (NR-52509), Chile-Santiago (NR-52510), USA-New York (NR-53518), and USA-WA (NR-52285).
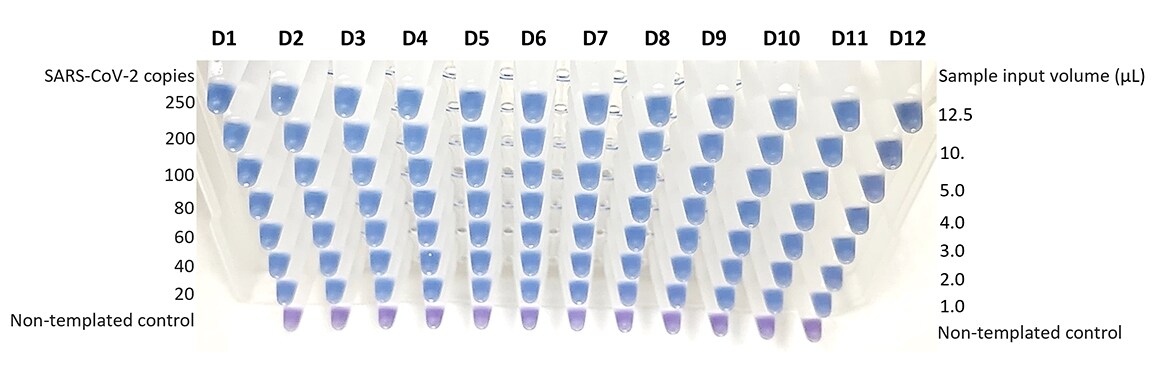
Figure 3. Using saliva-derived samples, the Colorimetric ReadiLAMP kit detections as few as 20 copies SARS-CoV-2 per reaction.
Samples from 12 unique saliva donors (D1 - D12) were supplemented with γ-irradiated SARS-CoV-2 isolate USA-WA1/2020 (BEI Resources). RNA was purified using the MagMAX Viral/Pathogen II Nucleic Acid Isolation Kit and the Thermo Scientific KingFisher Flex Magnetic Particle Processor. The 25 μL Colorimetric ReadiLAMP reactions were assembled using 1.0 – 12.5 μL input sample containing 0 – 250 copies of SARS-CoV-2 per reaction. After a 65 °C incubation for 30 min, all samples containing SARS-CoV-2 RNA transitioned to blue, positive reactions, while non-templated reactions remained purple. To increase sensitivity, sample input volume may be increased to 50 % of the total reaction volume (12.5/ 25 μL) with the caveat that individual reaction sensitivity is dependent on sample type, donor, and RNA purity. For saliva, the minimum range that can be robustly detected varies from 20 – 100 copies/ reaction for uncomplicated to challenging samples, respectively.
Product details for the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2
 Colorimetric ReadiLAMP™ kit, Colorimetric ReadiLAMP™ kit, 100 reactions |  Colorimetric ReadiLAMP™ kit, 1000 reactions | |
| Reactions | 100 reactions and 1000 reactions | |
| Test duration | ~30 mins – Direct LAMP assay ~1 hour – LAMP using purified RNA | |
| Sample types | Saliva and Swabs | |
| Detection | Colorimetric readout | |
| Sensitivity | Direct: ≤250 copies/25 uL LAMP reaction Purified: ≤100 copies/25 uL LAMP reaction | |
| Controls | Human Control Primer Mix and SARS-CoV-2 Control RNA | |
| Test format | 1-96 samples at a time | |
| Kit Contents | 100 Reactions | 1000 reactions |
| Kit storage temperature | –20℃ | |
| Instrument required | 65℃ heating block with heated lid | |
| Regulatory classification | RUO | |
| Catalogue Number | 100 reactions (Cat. No. A52539) or 1,000 reactions (Cat. No. A52544) | |
Why use the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2?
- Simplicity: Fast and simple protocol, easy to read results and robust system based on colorimetric indicators.
- Range of Samples types:
- Sample ranges from saliva, nasal swabs, nasopharyngeal swab, and other swabs.
- Sensitivity and variant coverage:The kit allows input of up to 50% purified RNA per LAMP reaction enabling maximal sensitivity in SARS-CoV-2 detection.
- Direct: ≤ 250 copies/25 µL LAMP reaction
- Purified: ≤ 100 copies/25 µL LAMP reaction
- Various SARS-CoV-2 variant coverage
- Speed to result and protocol flexibility – choose from 2 workflows:
- Direct LAMP protocol provides results in about 30 minutes using crude saliva or transport media samples.
- Purified RNA-based LAMP assay provides results in about 60 minutes: 30 minutes purification, followed by 30 minutes LAMP assay.
- Throughput
- Allows users to optimize their workflow for variable sample numbers (1-96 sample batches) and consumable types (individual tubes, strip tubes, 96-well plates) for efficient sample processing and rapid turn-around times.

Protocols and workflows for using the Invitrogen Colorimetric ReadiLAMP™ Kit, SARS-CoV-2
Use this kit to either perform direct assay or purified assay. Either method yields quality results that are easy to interpret with blue being positive Sars-CoV-2 reactions and violet being negative results.

Direct Method: SARS-CoV-2 in crude saliva/NP samples

Indirect Method: SARS-CoV-2 detection workflow for purified RNA samples

Performance data for Invitrogen Colorimetric ReadiLAMP™ Kit, Sars-CoV-2
See the results of using our kit for various sample types and target infectious disease analytes. With color coded positive and negative results, interpreting the data is simple.

Figure 1. The Colorimetric ReadiLAMP Kit enables highly specific SARS-CoV-2 detection.
Samples from 12 unique nasopharyngeal donors (D1 – D12) were supplemented with γ-irradiated SARS-CoV-2 isolate USA-WA1/2020 (BEI Resources), Influenza A H1N1, or Influenza A H3N2 (ATCC) or 5.0 μL NATtrol™ Respiratory Pathogen Panel-1 or Panel-2 (RP1/RP2, Zeptometrix). RNA was purified using the MagMAX Viral/Pathogen II Nucleic Acid Isolation Kit and the Thermo Scientific KingFisher Flex Magnetic Particle Processor. The 25 μL Colorimetric ReadiLAMP reactions were incubated at 65 °C for 30 min. In all samples supplemented with SARS-CoV-2, a positive blue color change was observed, while all other virally supplemented samples and controls remained a negative purple color. [RP1 contains the following viral particles and bacteria: Influenza A H3N2 (Brisbane/10/07), Influenza A H1N1 (NY/02/2009), Rhinovirus (Type 1A), Adenovirus (Type 3), Parainfluenza (Type 1), Parainfluenza (Type 4), Metapneumovirus (Peru 6-2003), Chlamydia pneumoniae (CWL-029), Mycoplasma pneumoniae (M129), and Coxsackievirus (Type A1). Whereas RP2 contains the following viral particles and bacteria: from Influenza A H1 (New Caledonia/20/99), Influenza B (Florida/02/06), RSV (Type A), Parainfluenza (Type 2), Parainfluenza (Type 3), Coronavirus (HKU-1 recombinant), Coronavirus (OC43), Coronavirus (NL63), Coronavirus (229E), Bordetella pertussis (A639).]

Figure 2. The Colorimetric ReadiLAMP Kit efficiently detects multiple SARS-CoV-2 strains.
Fifteen strains of SARS-CoV-2 RNA were obtained and evaluated in the Colorimetric ReadiLAMP assay. Each 25 μL reaction included 2.5 x 101 – 1.0 x 104 copies of SAR-CoV-2 in a background of 50 ng human genomic DNA. After 65 °C incubation for 30 min, 80% of strains were detected at a sensitivity of 25 copies/ reaction with all strains being detected at 50 copies/ reaction. These detectable strains, obtained from ATCC, were Hong Kong (NR-52388), Italy (NR-52498), England (NR-52499), Singapore (NR-52501), Germany (NR-52502), USA-IL (NR-52503), USA-CA1 (NR-52504), USA-AZ (NR-52505), USA-WI (NR-52506), USA-CA3 (NR-52507), USA-CA4 (NR-52508), USA-CA2 (NR-52509), Chile-Santiago (NR-52510), USA-New York (NR-53518), and USA-WA (NR-52285).

Figure 3. Using saliva-derived samples, the Colorimetric ReadiLAMP kit detections as few as 20 copies SARS-CoV-2 per reaction.
Samples from 12 unique saliva donors (D1 - D12) were supplemented with γ-irradiated SARS-CoV-2 isolate USA-WA1/2020 (BEI Resources). RNA was purified using the MagMAX Viral/Pathogen II Nucleic Acid Isolation Kit and the Thermo Scientific KingFisher Flex Magnetic Particle Processor. The 25 μL Colorimetric ReadiLAMP reactions were assembled using 1.0 – 12.5 μL input sample containing 0 – 250 copies of SARS-CoV-2 per reaction. After a 65 °C incubation for 30 min, all samples containing SARS-CoV-2 RNA transitioned to blue, positive reactions, while non-templated reactions remained purple. To increase sensitivity, sample input volume may be increased to 50 % of the total reaction volume (12.5/ 25 μL) with the caveat that individual reaction sensitivity is dependent on sample type, donor, and RNA purity. For saliva, the minimum range that can be robustly detected varies from 20 – 100 copies/ reaction for uncomplicated to challenging samples, respectively.
Related Products
- KingFisher Flex System
- KingFisher Apex System
- PureLink Viral RNA and DNA Mini
- MyBlock Mini Digital Dry Bath w /heated lids
- Veriti 96-Well Thermal Cycler
- MiniAmp Thermal Cycler
- SimpliAmp Thermal Cycler
- MagMAX Viral/Pathogen II (MVP II) Nucleic Acid Isolation Kit
- SpeciMAX Saliva Collection Kits
- 96-Well Plates
- 8 Stripe tubes
Resources
For Research Use Only. Not for use in diagnostic procedures.
