Search
Related Tables
Get Chapter Downloads from The Molecular Probes Handbook, 11th edition |
Bifunctional "crosslinking" reagents contain two reactive groups, thereby providing a means of covalently linking two target groups. The reactive groups in a chemical crosslinking reagent (Chemical Crosslinking Reagents—Section 5.2) typically belong to the classes of functional groups—including succinimidyl esters, maleimides and iodoacetamides—described in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1 and Thiol-Reactive Probes—Chapter 2.
In contrast, one of the reactive groups in each of our photoreactive crosslinking reagents (Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3) requires light activation before reacting with a target group. Crosslinking of a biopolymer (such as an antibody, enzyme, avidin or nucleic acid) to a low molecular weight molecule (such as a drug, toxin, peptide or oligonucleotide) or to another biopolymer yields a stable heteroconjugate. This bioconjugate can serve as a detection reagent in a wide variety of research and diagnostic assays or as an immunogen designed to elicit antibody production. Crosslinking reagents are also useful for probing the spatial relationships and interactions within and between biomolecules.
In homobifunctional crosslinking reagents (Chemical Crosslinking Reagents—Section 5.2), the reactive groups are identical. These reagents couple like functional groups—typically two thiols, two amines, two acids or two alcohols—and are predominantly used to form intramolecular crosslinks or to prepare polymers from monomers. When used to conjugate two different biomolecules, for example an enzyme to an antibody, these relatively nonspecific reagents tend to yield high molecular weight aggregates.
In heterobifunctional crosslinking reagents (Chemical Crosslinking Reagents—Section 5.2, Molecular Probes heterobifunctional crosslinkers—Table 5.1), the reactive groups have dissimilar chemistry, allowing the formation of crosslinks between unlike functional groups (Figure 5.1.1). As with homobifunctional crosslinking reagents, heterobifunctional crosslinking reagents can still form multiple intermolecular crosslinks to yield high molecular weight aggregates, but conjugations that use these reagents can be more easily controlled so as to optimize the stoichiometry of the target molecules. Thus, heterobifunctional crosslinking reagents are very useful for preparing conjugates between two different biomolecules.
The photoreactive crosslinking reagents (Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3) are a special subset of the heterobifunctional crosslinking reagents. Upon UV illumination, these reagents react with nucleophiles or form C–H insertion products (Figure 5.1.2, Figure 5.1.3, Figure 5.1.4).
An additional variation is the "zero-length" crosslinking reagent—a reagent that forms a chemical bond between two groups without itself being incorporated into the product (Figure 5.1.5). The water-soluble carbodiimide EDAC (E2247, Chemical Crosslinking Reagents—Section 5.2), which is used to couple carboxylic acids to amines, is an example of a zero-length crosslinking reagent.
A noncovalent interaction between two molecules that has very slow dissociation kinetics can also function as a crosslink. For example, reactive derivatives of phospholipids can be used to link the liposomes or cell membranes in which they are incorporated to antibodies or enzymes. Biotinylation and haptenylation reagents (Biotin and Hapten Derivatives—Chapter 4) can also be thought of as heterobifunctional crosslinking reagents because they comprise a chemically reactive group as well as a biotin or hapten moiety that binds with high affinity to avidin or an anti-hapten antibody, respectively. Similarly, avidin, streptavidin, NeutrAvidin biotin-binding protein and CaptAvidin biotin-binding protein (Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) can tightly bind up to four molecules of a biotinylated target.

Figure 5.1.1 Schematic illustration of the heterobifunctional crosslinker succinimidyl acetylthioacetate (SATA): A) attachment to an aminosilane-modified surface, B) deprotection with base and C) reaction with a thiol-reactive biomolecule.

Figure 5.1.2 Photoreactive crosslinking reaction of a simple aryl azide.

Figure 5.1.3 Photoreactive crosslinking reaction of a fluorinated aryl azide.

Figure 5.1.4 Photoreactive crosslinking reaction of a benzophenone derivative.
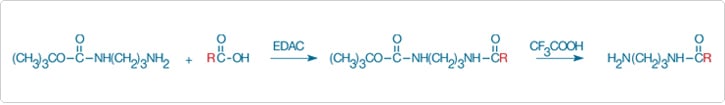 Figure 5.1.5 Conversion of a carboxylic acid group into an aliphatic amine. The activated carboxylic acid is derivatized with a half-protected aliphatic diamine (mono-N-(t-BOC)-propylenediamine), usually in an organic solvent, followed by removal of the t-BOC–protecting group with trifluoroacetic acid.
Figure 5.1.5 Conversion of a carboxylic acid group into an aliphatic amine. The activated carboxylic acid is derivatized with a half-protected aliphatic diamine (mono-N-(t-BOC)-propylenediamine), usually in an organic solvent, followed by removal of the t-BOC–protecting group with trifluoroacetic acid.
In addition to the photoreactive crosslinking reagents that are briefly mentioned above and described in detail in Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3, we prepare photoactivatable probes. Flash photolysis of photoactivatable or "caged" probes provides a means of controlling the release—both spatially and temporally—of biologically active products or other reagents of interest.![]() The chemical caging process may also confer membrane permeability on the caged ligand, as is the case for caged cAMP
The chemical caging process may also confer membrane permeability on the caged ligand, as is the case for caged cAMP ![]() and caged luciferin.
and caged luciferin.![]() Our selection of caged nucleotides, chelators, second messengers and neurotransmitters has tremendous potential for use with both live cells and isolated proteins (Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3).
Our selection of caged nucleotides, chelators, second messengers and neurotransmitters has tremendous potential for use with both live cells and isolated proteins (Photoactivatable Reagents, Including Photoreactive Crosslinkers and Caged Probes—Section 5.3).
We prepare caged versions of biologically active molecules, as well as caged fluorescent dyes that are essentially nonfluorescent until after photolysis. These caged fluorophores have proven useful for photoactivation of fluorescence (PAF) experiments, which are analogous to fluorescence recovery after photobleaching (FRAP) experiments except that the fluorophore is activated upon illumination rather than bleached. Measuring the fluorescent signal of the photoactivated fluorophore against a dark background is intrinsically more sensitive than measuring a dark photobleached region against a bright field.![]()
For Research Use Only. Not for use in diagnostic procedures.