Search
Page Contents
We are committed to the extensive development of tyramide signal amplification (TSA) in combination with our Alexa Fluor dyes to achieve high-resolution signal amplification in cell and tissue applications. TSA—sometimes called CARD, for Catalyzed Reporter Deposition—is an enzyme-mediated detection method that utilizes the catalytic activity of horseradish peroxidase (HRP) to generate high-density labeling of a target protein or nucleic acid sequence in situ.![]() TSA has been reported to increase detection sensitivity up to 100-fold, as compared with conventional avidin–biotinylated enzyme complex (ABC) procedures.
TSA has been reported to increase detection sensitivity up to 100-fold, as compared with conventional avidin–biotinylated enzyme complex (ABC) procedures.![]() Moreover, for multiparameter detection of targets in either live or fixed cells or tissues, TSA can be combined with several other important technologies, including our nucleic acid labeling kits (Labeling Oligonucleotides and Nucleic Acids—Section 8.2), primary and secondary antibodies, avidin and lectin conjugates (Antibodies, Avidins and Lectins—Chapter 7), cytoskeletal stains (Probes for Cytoskeletal Proteins—Chapter 11) , organelle probes (Probes for Organelles—Chapter 12) and cell tracers (Fluorescent Tracers of Cell Morphology and Fluid Flow—Chapter 14). The Zenon Horseradish Peroxidase Antibody Labeling Kits (Zenon Technology: Versatile Reagents for Immunolabeling—Section 7.3, Zenon Antibody Labeling Kits—Table 7.7), which are described below, are of particular utility when used in combination with TSA technology.
Moreover, for multiparameter detection of targets in either live or fixed cells or tissues, TSA can be combined with several other important technologies, including our nucleic acid labeling kits (Labeling Oligonucleotides and Nucleic Acids—Section 8.2), primary and secondary antibodies, avidin and lectin conjugates (Antibodies, Avidins and Lectins—Chapter 7), cytoskeletal stains (Probes for Cytoskeletal Proteins—Chapter 11) , organelle probes (Probes for Organelles—Chapter 12) and cell tracers (Fluorescent Tracers of Cell Morphology and Fluid Flow—Chapter 14). The Zenon Horseradish Peroxidase Antibody Labeling Kits (Zenon Technology: Versatile Reagents for Immunolabeling—Section 7.3, Zenon Antibody Labeling Kits—Table 7.7), which are described below, are of particular utility when used in combination with TSA technology.
TSA labeling is a combination of three (or four) elementary processes (Figure 6.2.1) that typically comprise:
- Binding of a probe to the target via immunoaffinity (proteins) or hybridization (nucleic acids) followed by secondary detection of the probe with an HRP-labeled antibody or streptavidin conjugate. Peroxidase conjugates of other targeting proteins such as lectins and receptor ligands are likely to be suitable for labeling targets, as is endogenous peroxidase activity.
Unconjugated HRP is also useful as a neuronal tracer, and can be combined with TSA
.
- Activation of multiple copies of a labeled tyramide derivative by HRP. Most often a fluorescent or biotinylated tyramide has been used; however, labeling with other hapten-conjugated tyramides
or with polymeric reagents, including tyramide-conjugated gold particles, has also been reported.
- Covalent coupling of the resulting highly reactive, short-lived tyramide radicals to residues (principally the phenol moiety of protein tyrosine residues) in the vicinity of the HRP–target interaction site, resulting in minimal diffusion-related loss of signal localization (Figure 6.2.2). In a unique application, fluorescein-labeled tyramine has been used to detect protein oxidation by reactive oxygen species (ROS, Generating and Detecting Reactive Oxygen Species—Section 18.2) in fibroblasts exposed to oxidative stress
and in the extracellular proteins of endothelial cells exposed to an oxidative burst from phorbol myristate acetate–activated neutrophils.
In direct TSA protocols, the fluorescent signal can be immediately detected, resulting in both excellent spatial resolution (Figure 6.2.3) and high signal intensity. When using a hapten-labeled tyramide such as biotin-XX tyramide, a subsequent detection step is required using a bioconjugate that recognizes the hapten, in this case a fluorescent streptavidin (Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6). Alternatively, the hapten-labeled tyramide can be detected using an alkaline phosphate– or HRP-labeled hapten recognizer in conjunction with a fluorogenic or chromogenic substrate (![]() ), resulting in another enzyme-amplified detection step. Chemiluminescent detection of an HRP-deposited biotin tyramide has also been reported.
), resulting in another enzyme-amplified detection step. Chemiluminescent detection of an HRP-deposited biotin tyramide has also been reported.![]() The streptavidin conjugate of NANOGOLD 1.4 nm gold clusters (N24918, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) has been used to make biotin tyramide conjugates visible in light and electron microscopy.
The streptavidin conjugate of NANOGOLD 1.4 nm gold clusters (N24918, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) has been used to make biotin tyramide conjugates visible in light and electron microscopy.![]() The antibody and streptavidin conjugates of Alexa Fluor FluoroNanogold 1.4 nm gold clusters (Secondary Immunoreagents—Section 7.2, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) can also be used with hapten-labeled tyramides for correlated fluorescence, light and electron microscopy studies.
The antibody and streptavidin conjugates of Alexa Fluor FluoroNanogold 1.4 nm gold clusters (Secondary Immunoreagents—Section 7.2, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) can also be used with hapten-labeled tyramides for correlated fluorescence, light and electron microscopy studies.
The signal amplification conferred by the turnover of multiple tyramide substrates per peroxidase label translates into practical benefits, namely ultrasensitive detection of low-abundance targets in fluorescence in situ hybridization,![]() immunohistochemistry,
immunohistochemistry,![]() neuroanatomical tracing
neuroanatomical tracing![]() and other applications. For example, we have utilized TSA and Alexa Fluor 488 tyramide to detect expression of low-abundance epidermal growth factor (EGF) and estrogen receptors by flow cytometry with far greater sensitivity than can be obtained using a directly labeled EGF probe (Figure 6.2.4) or fluorophore- or hapten-labeled antibodies to the estrogen receptor (Figure 6.2.5). Application of TSA resulted in significantly increased detectability of estrogen receptors in urinary bladder carcinomas, as compared with conventional immunohistochemical analysis.
and other applications. For example, we have utilized TSA and Alexa Fluor 488 tyramide to detect expression of low-abundance epidermal growth factor (EGF) and estrogen receptors by flow cytometry with far greater sensitivity than can be obtained using a directly labeled EGF probe (Figure 6.2.4) or fluorophore- or hapten-labeled antibodies to the estrogen receptor (Figure 6.2.5). Application of TSA resulted in significantly increased detectability of estrogen receptors in urinary bladder carcinomas, as compared with conventional immunohistochemical analysis.![]()
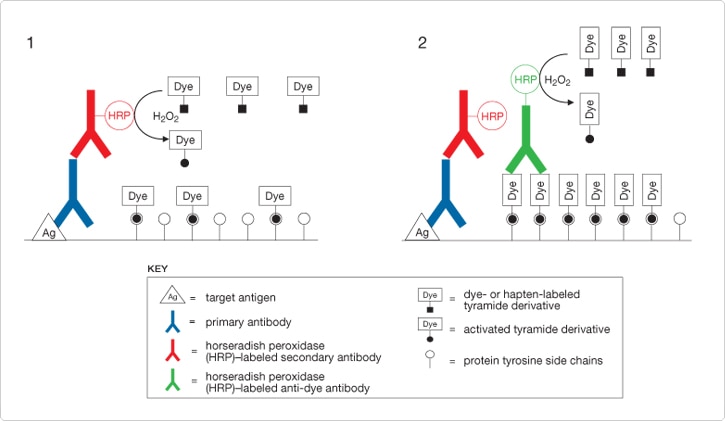
Figure 6.2.1 Schematic representation of TSA detection methods applied to immunolabeling of an antigen. The antigen is detected by a primary antibody, followed by a horseradish peroxidase (HRP)–labeled secondary antibody in conjunction with a dye-labeled tyramide, resulting in localized deposition of the activated tyramide derivative (Stage 1). Further dye deposition , and therefore higher levels of signal amplification, can be generated by detecting dye deposited in Stage 1 with a HRP-labeled anti-dye antibody in conjunction with a dye-labeled tyramide (Stage 2).
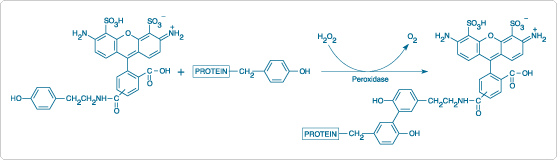
Figure 6.2.2 Coupling of Alexa Fluor 488 tyramide to protein tyrosine side chains via peroxidase-mediated formation of an O,O'-dityrosine adduct.
 | Figure 6.2.3 Golgi in HeLa cells detected with Alexa Fluor 546 tyramide. Cells were fixed and permeabilized, then labeled with anti–human Golgin-97 antibody (A21270) and detected using HRP-conjugated goat anti–mouse IgG antibody and Alexa Fluor 546 tyramide, which are components of the TSA Kit #3 (T20913). The nuclei were counterstained using DAPI (D1306, D3571, D21490). The images were acquired using filters appropriate for DAPI and Alexa Fluor 546 and processed using MetaMorph software from Universal Imaging Corp. |
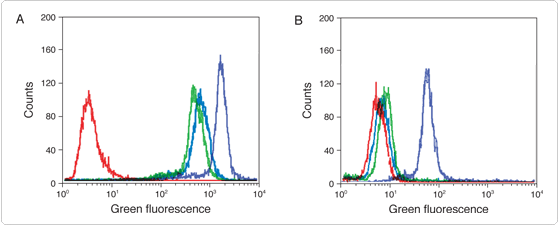
Figure 6.2.4 Detection of epidermal growth factor (EGF) receptors directly or with signal amplification. Cells expressing high (A431 cells, panel A) and low (NIH 3T3 cells, panel B) levels of EGF receptors were either directly labeled with the preformed Alexa Fluor 488 complex of biotinylated epidermal growth factor (E13345, blue) or indirectly labeled with biotinylated EGF (E3477) followed by either Alexa Fluor 488 streptavidin (S11223, green) or HRP-conjugated streptavidin and Alexa Fluor 488 tyramide (purple), components of our TSA Kit #22 (T20932).
 | Figure 6.2.5 Enhancement of estrogen receptor detection sensitivity by tyramide signal amplification (TSA). SKBR3 cells with characteristically low levels of estrogen receptor expression were fixed, permeabilized and treated with H2O2 to inhibit endogenous peroxidase activity. A mouse anti–human estrogen receptor monoclonal antibody (Chemicon) was labeled with the Alexa Fluor 488 dye or with biotin using our Zenon Alexa Fluor 488 Mouse IgG1 Labeling Kit (Z25002) or Zenon Biotin-XX Mouse IgG1 Labeling Kit (Z25052), respectively. Detection of estrogen receptors using the labeled antibodies was performed on a Becton Dickinson FACScan flow cytometer with excitation at 488 nm. The cellular fluorescence intensity histograms represent detection with Alexa Fluor 488 dye–labeled antibodies (green), biotinylated antibodies coupled to Alexa Fluor 488 streptavidin (S11223, red) and biotinylated antibodies coupled to HRP–streptavidin and Alexa Fluor 488 tyramide (TSA Kit #22, T20932; orange). The blue histogram represents unstained cells. |
TSA Kits
We have developed a number of TSA Kits that combine the versatile tyramide signal amplification technology with our high-performance Alexa Fluor tyramides, Oregon Green 488 tyramide or biotin-XX tyramide (Tyramide Signal Amplification (TSA) Kits—Table 6.1). Each kit provides sufficient materials to stain 50–150 slide preparations and includes the following components:
- Tyramide labeled with an Alexa Fluor dye, Oregon Green 488 dye or biotin-XX
- HRP-conjugated anti–mouse IgG antibody, anti–rabbit IgG antibody or streptavidin
- Amplification reaction buffer
- H2O2 reaction additive
- TSA blocking reagent
- Detailed protocols for tyramide labeling (Tyramide Signal Amplification (TSA) Kits)
Our fluorescent and biotin-XX tyramides are not currently available as stand-alone reagents.
Zenon Horseradish Peroxidase Antibody Labeling Kits
Our Zenon Horseradish Peroxidase Antibody Labeling Kits, available for mouse IgG (Z25054, Z25154, Z25254), rabbit IgG (Z25354) and human IgG (Z25454), make it possible to quantitatively label even submicrogram quantities of a primary antibody with HRP immediately before it is applied to the sample (Zenon Technology: Versatile Reagents for Immunolabeling—Section 7.3, Zenon Antibody Labeling Kits—Table 7.7, Figure 6.2.6). Antibodies labeled with HRP using these Zenon Antibody Labeling Kits can replace the HRP-labeled goat anti–mouse IgG and goat anti–rabbit IgG antibody conjugates in any of the TSA Kits containing these secondary detection reagents.
 | Figure 6.2.6 Labeling scheme utilized in the Zenon Antibody Labeling Kits. A) An unlabeled IgG antibody is incubated with the Zenon labeling reagent, which contains a fluorophore-labeled, Fc-specific anti-IgG Fab fragment. B) This labeled Fab fragment binds to the Fc portion of the IgG antibody. C) Excess Fab fragment is then neutralized by the addition of a nonspecific IgG, preventing crosslabeling by the Fab fragment in experiments where primary antibodies of the same type are present. Note that the Fab fragment used for labeling need not be coupled to a fluorophore, but could instead be coupled to an enzyme (such as HRP) or to biotin. |
Zenon Antibody Labeling Kit Enhanced with TSA Technology
For mouse IgG1 primary antibodies, we have developed the Zenon Alexa Fluor 488 Mouse IgG1 Labeling Kit enhanced with TSA technology (Z25090), which provides the necessary reagents from both the Zenon Horseradish Peroxidase Mouse IgG1 Labeling Kit and the Alexa Fluor 488 TSA Kit, for researchers who want both the ease of labeling mouse IgG1 antibodies with Zenon labeling reagents and the signal amplification afforded by TSA technology. Each kit provides sufficient reagents for 25 labelings, including:
- Zenon HRP mouse IgG1 labeling reagent
- Zenon mouse IgG blocking reagent
- Alexa Fluor 488 tyramide
- Dimethylsulfoxide (DMSO)
- TSA blocking reagent
- TSA amplification buffer
- Hydrogen peroxide (H2O2)
- Detailed protocols for Zenon complex formation and tyramide labeling (Enhanced Zenon Mouse IgG Labeling Kits)
The Zenon HRP mouse IgG1 labeling reagent contains Fab fragments of goat IgG antibodies directed against the Fc portion of intact mouse IgG1 antibodies. These Fab fragments have been purified to ensure their selectivity for the Fc portion of the mouse IgG1 antibody and then labeled with HRP. This Zenon HRP mouse IgG1 labeling reagent is simply mixed with any mouse IgG1 primary antibody to form the Fab–mouse IgG1 complexes, which can be used for immunolabeling similar to that of primary antibodies covalently labeled with HRP. TSA technology is then used to detect the target-bound Fab–mouse IgG1 complex. Each HRP label on the Fab–mouse IgG1 complexes can activate multiple copies of the Alexa Fluor 488 tyramide to produce short-lived tyramide radicals that are highly reactive with residues near the interaction site, yielding an amplified green-fluorescent signal with minimal diffusion.
Other Horseradish Peroxidase Conjugates for Secondary Detection
For use in signal amplification of antibody- or biotin-labeled targets, we offer the horseradish peroxidase conjugates of:
- Streptavidin and NeutrAvidin biotin-binding protein (S911, A2664; Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6)
- Goat anti–mouse IgG antibody (A10551, A10668, A10677, A10685, G21040)
- Goat anti–mouse F(ab')2 fragment (F21453)
- Goat anti–rabbit IgG antibody (G21234)
- Goat anti–rabbit F(ab')2 fragment (A10547)
- Goat anti–rat IgG antibody (A10549)
- Goat anti–rat F(ab')2 fragment (A10548)
- Mouse anti–human IgG antibody (A10648, A10654)
- Rabbit anti–goat IgG antibody (R21459)
- Rabbit anti–mouse IgG antibody (R21455)
A more thorough discussion of our secondary antibodies can be found in Secondary Immunoreagents—Section 7.2 (Summary of Molecular Probes secondary antibody conjugate—Table 7.1).
TSA technology has been used successfully for over two decades,![]() and there are many reports of the use of biotin tyramide for indirect target labeling and fluorescent tyramides for direct target labeling. Direct labeling methods have the considerable advantage of saving a second step in the detection scheme. Moreover, labeling targets with fluorescent tyramides instead of biotin tyramide has the further advantage of avoiding amplification of endogenous biotin in cells and tissues, such as we have observed in mitochondrial staining with streptavidin conjugates in the absence of a biotinylated probe (
and there are many reports of the use of biotin tyramide for indirect target labeling and fluorescent tyramides for direct target labeling. Direct labeling methods have the considerable advantage of saving a second step in the detection scheme. Moreover, labeling targets with fluorescent tyramides instead of biotin tyramide has the further advantage of avoiding amplification of endogenous biotin in cells and tissues, such as we have observed in mitochondrial staining with streptavidin conjugates in the absence of a biotinylated probe (![]() ). In most of the early reports, the biotin tyramide used did not have the additional 14-atom spacer that we utilize in our biotin-XX tyramide to make the probe more accessible to avidin conjugates, nor are the specific fluorescent dyes available in our TSA Kits. Therefore, the specific methods described in these references should be considered guides rather than definitive protocols, and results using our TSA reagents may differ from those reported. In our experience, the Alexa Fluor 488 tyramide (Tyramide Signal Amplification (TSA) Kits—Table 6.1) provides greater signal and significantly greater photostability than fluorescein tyramide, and the other Alexa Fluor tyramides also yield intense staining of targets.
). In most of the early reports, the biotin tyramide used did not have the additional 14-atom spacer that we utilize in our biotin-XX tyramide to make the probe more accessible to avidin conjugates, nor are the specific fluorescent dyes available in our TSA Kits. Therefore, the specific methods described in these references should be considered guides rather than definitive protocols, and results using our TSA reagents may differ from those reported. In our experience, the Alexa Fluor 488 tyramide (Tyramide Signal Amplification (TSA) Kits—Table 6.1) provides greater signal and significantly greater photostability than fluorescein tyramide, and the other Alexa Fluor tyramides also yield intense staining of targets.
Immunohistochemical Detection Using TSA
TSA detection can be applied to a variety of immunohistochemical specimen preparations, including crytostat sections, formaldehyde-fixed paraffin-embedded sections, plastic-embedded sections and cultured cells. In immunohistochemical applications (![]() ), sensitivity enhancements derived from TSA allow primary antibody dilutions to be increased—up to a 1:1,000,000 antibody dilution was possible in one reported case,
), sensitivity enhancements derived from TSA allow primary antibody dilutions to be increased—up to a 1:1,000,000 antibody dilution was possible in one reported case,![]() although a 5- to 50-fold increase over the normal dilution factor is more common
although a 5- to 50-fold increase over the normal dilution factor is more common ![]() —in order to reduce nonspecific background signals.
—in order to reduce nonspecific background signals.![]() Additionally, the strong signal amplification provided by the TSA method can overcome relatively high autofluorescence of cells and tissues.
Additionally, the strong signal amplification provided by the TSA method can overcome relatively high autofluorescence of cells and tissues.![]() Furthermore, because TSA and diaminobenzidine (DAB) oxidation are both peroxidase-mediated reactions, TSA is readily adaptable for correlated fluorescence and electron microscopy studies.
Furthermore, because TSA and diaminobenzidine (DAB) oxidation are both peroxidase-mediated reactions, TSA is readily adaptable for correlated fluorescence and electron microscopy studies.![]() The significantly lower detection threshold of TSA, as compared with fluorescent secondary antibodies, allows detection of two targets by primary antibodies raised in the same host species, without substantial crosstalk between the signals.
The significantly lower detection threshold of TSA, as compared with fluorescent secondary antibodies, allows detection of two targets by primary antibodies raised in the same host species, without substantial crosstalk between the signals.![]() The first target was detected using TSA and a primary antibody concentration that was so low that it was essentially undetectable by fluorescent secondary antibodies; the second target was then detected by conventional secondary immunofluorescence labeling.
The first target was detected using TSA and a primary antibody concentration that was so low that it was essentially undetectable by fluorescent secondary antibodies; the second target was then detected by conventional secondary immunofluorescence labeling.
Fluorescence In Situ Hybridization Using TSA
The increased sensitivity afforded by TSA (Figure 6.2.7) can be critically important for detecting relatively short oligonucleotide probes and low-abundance mRNAs by fluorescence in situ hybridization ![]() (FISH). Cosmid detection in formalin-fixed, paraffin-embedded sections is cumbersome, and the ability to use smaller cosmid probes of less than 1000 bases in conjunction with TSA detection technology is likely to be an important technique for FISH.
(FISH). Cosmid detection in formalin-fixed, paraffin-embedded sections is cumbersome, and the ability to use smaller cosmid probes of less than 1000 bases in conjunction with TSA detection technology is likely to be an important technique for FISH.![]() TSA is also faster than traditional FISH detection schemes, allowing definitive results to be obtained within a single day. In addition, a two-stage amplification method for ultrasensitive mRNA detection has been reported that combines TSA detection of biotinylated riboprobes with alkaline phosphatase–mediated fluorescence generation using Molecular Probes ELF 97 phosphatase substrate
TSA is also faster than traditional FISH detection schemes, allowing definitive results to be obtained within a single day. In addition, a two-stage amplification method for ultrasensitive mRNA detection has been reported that combines TSA detection of biotinylated riboprobes with alkaline phosphatase–mediated fluorescence generation using Molecular Probes ELF 97 phosphatase substrate ![]() (Phosphatase-Based Signal Amplification Techniques—Section 6.3).
(Phosphatase-Based Signal Amplification Techniques—Section 6.3).
TSA, however, is not a panacea for FISH sensitivity problems. Because both specifically and nonspecifically bound probe signals are amplified, TSA will not compensate for suboptimal hybridization conditions. Optimal probe concentrations are typically 2- to 10-fold lower for TSA-detected FISH than for conventional immunocytochemical detection procedures, again saving on the cost of expensive hybridization probes.![]() Typically, FISH probes are labeled by indirect methods that use streptavidin- or antibody-conjugated HRP. Techniques for direct labeling of oligonucleotide probes have been developed to eliminate background signals due to nonspecific binding of peroxidase conjugates.
Typically, FISH probes are labeled by indirect methods that use streptavidin- or antibody-conjugated HRP. Techniques for direct labeling of oligonucleotide probes have been developed to eliminate background signals due to nonspecific binding of peroxidase conjugates.![]()
As with some other detection systems, TSA technology allows several probes to be hybridized simultaneously to identify multiple targets. Signal development using multicolored fluorescent tyramides must then be carried out sequentially, with a peroxidase inactivation step between each TSA reaction to prevent crosstalk ![]() (
(![]() ). TSA amplification followed by peroxidase inactivation through mild acid treatment with 0.01 M HCl for 10 minutes at room temperature
). TSA amplification followed by peroxidase inactivation through mild acid treatment with 0.01 M HCl for 10 minutes at room temperature ![]() and then reapplication of TSA using a fluorescent tyramide of a different fluorescent color has been used for triple-labeled in situ hybridization.
and then reapplication of TSA using a fluorescent tyramide of a different fluorescent color has been used for triple-labeled in situ hybridization.![]()
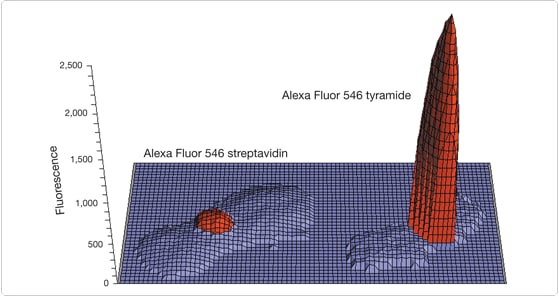
Figure 6.2.7 Digital image analysis comparison of in situ–hybridized biotinylated α-satellite probes detected using TSA Kit #23 (T20933) with HRP–streptavidin and Alexa Fluor 546 tyramide (right) or Alexa Fluor 546 streptavidin (S11225, left). Both images were converted to pixel intensity values using MetaMorph software (Universal Imaging Corporation) and transferred to a Microsoft Excel spreadsheet for plotting. Alexa Fluor 546 dye and DAPI (counterstain) intensity values are shown in red and blue, respectively. Alexa Fluor 546 dye intensity values below 35% of maximum were omitted for clarity.
Detection of Hapten-Labeled Tyramides
When a tyramide labeled with a hapten such as biotin-XX is used for TSA in an indirect labeling technique, a signal-generation reagent or scheme is necessary. A fluorescent tyramide such as our Oregon Green 488 tyramide can also be utilized as a hapten for subsequent detection and amplification by an anti–fluorescein/Oregon Green dye antibody (Anti-Dye and Anti-Hapten Antibodies—Section 7.4). Various reagents and reagent combinations have been reported for detecting enzyme-deposited biotin tyramide or fluorescein tyramide that should be equally suitable for use with our biotin-XX tyramide and Oregon Green 488 tyramide, including:
- Streptavidin conjugate of alkaline phosphatase (S921, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) in combination with NBT/BCIP
(N6495, N6547; Phosphatase-Based Signal Amplification Techniques—Section 6.3)
- Streptavidin conjugate of HRP (S911, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) or the rabbit anti–fluorescein/Oregon Green dye antibody conjugate of HRP
(A21253, Anti-Dye and Anti-Hapten Antibodies—Section 7.4) in combination with a traditional chromogenic peroxidase substrate such as diaminobenzidine (DAB)
(
)
- Fluorescent conjugates of avidin or streptavidin (Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6).
- Qdot nanocrystal streptavidin conjugates
(Qdot Nanocrystals—Section 6.6)
- Diaminobenzidine (DAB) Histochemistry Kits (D22185, D22187; see below), for direct use with biotin-XX tyramide or conversion of fluorescent signals to permanent staining
- NANOGOLD and Alexa Fluor FluoroNanogold conjugates of antibodies (Secondary Immunoreagents—Section 7.2) and streptavidin (Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6), for target localization using a combination of light and electron microscopy

Anti–fluorescein/Oregon Green dye antibody conjugates of HRP (Anti-Dye and Anti-Hapten Antibodies—Section 7.4, Anti-fluorophore antibodies and their conjugates—Table 7.8) have been used with fluorescein-labeled probes and TSA to detect:
Similarly, our anti–Green Fluorescent Protein (anti-GFP) antibodies (Anti–Epitope Tag and Anti-Reporter Antibodies—Section 7.5) can be used in combination with TSA for ultrasensitive immunocytochemical detection of GFP in situations where the expression level is insufficient for direct fluorescence detection.![]()
Double and Sequential Amplification with TSA
To achieve greater signal amplification, sequential rounds of amplification can be achieved using TSA ![]() (Figure 6.2.1). For example, in the first round biotin-XX tyramide can be deposited on a target using one of our biotin-XX tyramide TSA Kits (Tyramide Signal Amplification (TSA) Kits—Table 6.1). In a subsequent step, the peroxidase conjugate of streptavidin that is used in TSA Kit #21 (T20931) is used again, but this time in combination with an Alexa Fluor tyramide, Oregon Green 488 tyramide or another round of biotin-XX tyramide. Presumably, this amplification can be continued for at least a third round, although some loss of spatial resolution may result. Biotin tyramide that has first been deposited at the binding site of a biotin-labeled riboprobe using the streptavidin conjugate of HRP has been further amplified with the streptavidin conjugate of alkaline phosphatase (S921, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) in conjunction with ELF 97 phosphate for the ultrasensitive detection of a scarce leptin receptor mRNA.
(Figure 6.2.1). For example, in the first round biotin-XX tyramide can be deposited on a target using one of our biotin-XX tyramide TSA Kits (Tyramide Signal Amplification (TSA) Kits—Table 6.1). In a subsequent step, the peroxidase conjugate of streptavidin that is used in TSA Kit #21 (T20931) is used again, but this time in combination with an Alexa Fluor tyramide, Oregon Green 488 tyramide or another round of biotin-XX tyramide. Presumably, this amplification can be continued for at least a third round, although some loss of spatial resolution may result. Biotin tyramide that has first been deposited at the binding site of a biotin-labeled riboprobe using the streptavidin conjugate of HRP has been further amplified with the streptavidin conjugate of alkaline phosphatase (S921, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) in conjunction with ELF 97 phosphate for the ultrasensitive detection of a scarce leptin receptor mRNA.![]() In another example demonstrating the versatility of the TSA technology, several labeling technologies were combined sequentially to detect the HIV-1 virus:
In another example demonstrating the versatility of the TSA technology, several labeling technologies were combined sequentially to detect the HIV-1 virus:![]()
- In situ hybridization with a 15-base peptide nucleic acid probe labeled with a single fluorescein dye
- Complexation with an HRP conjugate of anti-fluorescein antibody
- Incubation with biotin tyramide
- Incubation with the streptavidin conjugate of HRP
- Re-incubation with biotin tyramide
- Detection with the Alexa Fluor 488 conjugate of streptavidin (S11223, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6)
Alternatively, for detection by light microscopy, the sample was incubated with the streptavidin conjugate of HRP in conjunction with DAB instead of Alexa Fluor 488 streptavidin.
Additional Tips on Using TSA Technology
Use of the TSA technology is not without its precautions. Among these is the possibility of endogenous peroxidase activity in certain cells, especially eosinophils.![]() This activity can be at least partially blocked by incubation with 0.3–3% hydrogen peroxide for about 60 minutes. Second, when using biotin-XX tyramide, endogenous biotinylated proteins are a potential problem (
This activity can be at least partially blocked by incubation with 0.3–3% hydrogen peroxide for about 60 minutes. Second, when using biotin-XX tyramide, endogenous biotinylated proteins are a potential problem (![]() ). Third, because of the significant signal amplification capability of TSA, nonspecific binding of labeled hybridization probes, antibodies and other targeting probes can lead to unacceptably high background staining. This nonspecific staining can be alleviated to some degree with appropriate blocking reagents.
). Third, because of the significant signal amplification capability of TSA, nonspecific binding of labeled hybridization probes, antibodies and other targeting probes can lead to unacceptably high background staining. This nonspecific staining can be alleviated to some degree with appropriate blocking reagents.![]() Furthermore, the high sensitivity of TSA permits antibodies and nucleic acid probes to be highly diluted, far below the amount required for target saturation, thus reducing nonspecific background. Antibody and nucleic acid probe dilution can also substantially reduce the cost of an assay and the amount of a rare material required for staining.
Furthermore, the high sensitivity of TSA permits antibodies and nucleic acid probes to be highly diluted, far below the amount required for target saturation, thus reducing nonspecific background. Antibody and nucleic acid probe dilution can also substantially reduce the cost of an assay and the amount of a rare material required for staining.![]()
Mammalian cells and tissues contain biotin-dependent carboxylases, which are required for a variety of metabolic functions. These biotin-containing enzymes produce substantial background signals when biotin–streptavidin detection systems are used to identify cellular targets ![]() (
(![]() ). Because the TSA technology is so sensitive, we recommended preblocking endogenous biotin in cells with our Endogenous Biotin-Blocking Kit (E21390, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) when using TSA Kits containing biotin-XX tyramide and the streptavidin conjugate of HRP. The Endogenous Biotin-Blocking Kit provides streptavidin and biotin solutions in convenient dropper bottles and an easy-to-follow protocol; sufficient material is provided for approximately one hundred 18 mm × 18 mm glass coverslips.
). Because the TSA technology is so sensitive, we recommended preblocking endogenous biotin in cells with our Endogenous Biotin-Blocking Kit (E21390, Avidin, Streptavidin, NeutrAvidin and CaptAvidin Biotin-Binding Proteins and Affinity Matrices—Section 7.6) when using TSA Kits containing biotin-XX tyramide and the streptavidin conjugate of HRP. The Endogenous Biotin-Blocking Kit provides streptavidin and biotin solutions in convenient dropper bottles and an easy-to-follow protocol; sufficient material is provided for approximately one hundred 18 mm × 18 mm glass coverslips.
Improvement of TSA detection by post-incubation heating has been reported.![]() Addition of viscosity-increasing dextran sulfate, poly(vinyl alcohol), poly(ethylene glycol) or poly(vinyl pyrrolidone) to the medium is reported to decrease diffusion of the phenoxy radical intermediate, resulting in superior localization of the signal.
Addition of viscosity-increasing dextran sulfate, poly(vinyl alcohol), poly(ethylene glycol) or poly(vinyl pyrrolidone) to the medium is reported to decrease diffusion of the phenoxy radical intermediate, resulting in superior localization of the signal.![]() Hybridization probes that are directly labeled with HRP are reportedly useful for lowering nonspecific binding when working with labeled tyramides.
Hybridization probes that are directly labeled with HRP are reportedly useful for lowering nonspecific binding when working with labeled tyramides.![]() Endogenous peroxidase can be sufficient to yield labeling at the site of this activity in cells, as in the case of eosinophils.
Endogenous peroxidase can be sufficient to yield labeling at the site of this activity in cells, as in the case of eosinophils.![]() The review by Speel, Hopman and Komminoth gives additional practical suggestions and references.
The review by Speel, Hopman and Komminoth gives additional practical suggestions and references.![]()
DAB Histochemistry Kits
The use of HRP for enzyme-amplified immunodetection—commonly referred to as immunoperoxidase labeling—is a well-established standard histochemical technique.![]() The most widely used HRP substrate for these applications is diaminobenzidine (DAB), which generates a brown-colored polymeric oxidation product localized at HRP-labeled sites. The DAB reaction product can be visualized directly by bright-field light microscopy or, following osmication, by electron microscopy. We offer DAB Histochemistry Kits for detecting mouse IgG primary antibodies (D22185) and biotinylated antibodies and tracers (D22187). Each kit contains sufficient materials to stain approximately 200 slides, including:
The most widely used HRP substrate for these applications is diaminobenzidine (DAB), which generates a brown-colored polymeric oxidation product localized at HRP-labeled sites. The DAB reaction product can be visualized directly by bright-field light microscopy or, following osmication, by electron microscopy. We offer DAB Histochemistry Kits for detecting mouse IgG primary antibodies (D22185) and biotinylated antibodies and tracers (D22187). Each kit contains sufficient materials to stain approximately 200 slides, including:
- Diaminobenzidine (DAB)
- HRP-labeled goat anti–mouse IgG antibody (in Kit D22185) or streptavidin (in Kit D22187) conjugate
- H2O2 reaction additive
- Blocking reagent
- Staining buffer
- Detailed staining protocols (Diaminobenzidine Histochemistry Kits)
Luminol and MCLA: Chemiluminescent Peroxidase Substrates
Nonisotopic immunoassays utilizing peroxidase conjugates and the chemiluminescent horseradish peroxidase substrate luminol (L8455) have provided a rapid and sensitive method for quantitating a wide variety of analytes, including cholesterol,![]() digoxin
digoxin ![]() and acetylcholine.
and acetylcholine.![]() Addition of trace amounts of luciferin (L2911, L2912, L2916; Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6) has been shown to considerably enhance the sensitivity in the assay of thyroxine, digoxin, α-fetoprotein and other analytes.
Addition of trace amounts of luciferin (L2911, L2912, L2916; Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6) has been shown to considerably enhance the sensitivity in the assay of thyroxine, digoxin, α-fetoprotein and other analytes.![]() A method that employs luminol has been developed for the quantitation of very limiting samples of human DNA from single hairs, saliva, small blood stains and paraffin-embedded and fixed tissue sections. Using a biotinylated oligodeoxynucleotide probe to membrane-immobilized DNA, horseradish peroxidase streptavidin and luminol, researchers have detected 150 pg of human DNA.
A method that employs luminol has been developed for the quantitation of very limiting samples of human DNA from single hairs, saliva, small blood stains and paraffin-embedded and fixed tissue sections. Using a biotinylated oligodeoxynucleotide probe to membrane-immobilized DNA, horseradish peroxidase streptavidin and luminol, researchers have detected 150 pg of human DNA.![]()
MCLA (M23800) is principally utilized as a superoxide-sensitive chemiluminescent probe (Generating and Detecting Reactive Oxygen Species—Section 18.2). MCLA has also been utilized for the determination of both horseradish peroxidase ![]() and myeloperoxidase.
and myeloperoxidase.![]()
The Amplex ELISA Development Kits for Mouse IgG (A33851) and for Rabbit IgG (A33852) provide a comprehensive set of components for creating fluorescence-based ELISAs using mouse and rabbit detection antibodies, respectively.![]() This assay is based on the Amplex UltraRed reagent, a fluorogenic substrate for horseradish peroxidase (HRP) that reacts with H2O2 in a 1:1 stoichiometric ratio to produce a brightly fluorescent and strongly absorbing reaction product (excitation/emission maxima ~568/581 nm). Because the Amplex UltraRed peroxidation product has long-wavelength spectra, there is little interference from the blue or green autofluorescence found in most biological samples.
This assay is based on the Amplex UltraRed reagent, a fluorogenic substrate for horseradish peroxidase (HRP) that reacts with H2O2 in a 1:1 stoichiometric ratio to produce a brightly fluorescent and strongly absorbing reaction product (excitation/emission maxima ~568/581 nm). Because the Amplex UltraRed peroxidation product has long-wavelength spectra, there is little interference from the blue or green autofluorescence found in most biological samples.
With a high extinction coefficient, good quantum efficiency and resistance to autooxidation, the fluorescence-based Amplex UltraRed reagent delivers better sensitivity and a broader assay range than colorimetric reagents. In a sandwich ELISA format, we can routinely detect 1 pg of C-reactive protein using HRP-conjugated goat anti–rabbit IgG antibody (Figure 6.2.8); this detection limit is 25-fold lower than that obtained from the same sandwich ELISA format using the common colorimetric reagent TMB.
Each Amplex ELISA Development Kit contains:
- Amplex UltraRed reagent
- Dimethylsulfoxide (DMSO)
- Concentrated phosphate-buffered saline (PBS), pH 7.2
- Horseradish peroxidase conjugate of goat anti–mouse IgG antibody (in A33851) or goat anti–rabbit IgG antibody (in A33852)
- Amplex stop reagent
- Hydrogen peroxide (H2O2)
- 0.1 M sodium bicarbonate buffer, pH ~9.3
- Bovine serum albumin (BSA)
- Tween 20
- Nunc-Immuno MaxiSorp U96 plate
- Detailed protocols (Amplex ELISA Development Kit for Mouse IgG with Amplex UltraRed Reagent, Amplex ELISA Development Kit for Rabbit IgG with Amplex UltraRed Reagent)
Sufficient reagents are provided in each kit for 500 microplate assays in a 96-well fluorescence microplate format (100 µL per assay).
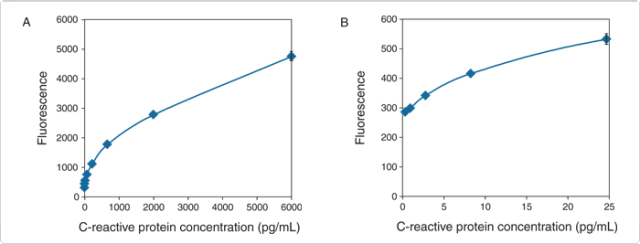
Figure 6.2.8 Detection range of C-reactive protein (CRP) using the Amplex ELISA Development Kit for Rabbit IgG (A33852). The sandwich ELISA was carried out using a mouse anti-CRP capture antibody, CRP in a concentration range between 6000 pg/mL and 0.10 pg/mL and a rabbit polyclonal anti-CRP primary antibody (10 µL per well of a 50 ng/mL solution). The Z' factor analysis of the data obtained gives a lower detection level for CRP in this assay of 1 pg/mL or 0.1 pg/well (based on a well volume of 100 µL in the sandwich ELISA).
For Research Use Only. Not for use in diagnostic procedures.