Search
Combining the Click-iT® EdU Assay with Signal Amplification for Sensitive Detection of Nascent mtDNA
Changes in mitochondrial DNA (mtDNA) copy number are not only important for normal biological processes, but are also implicated in drug toxicity, aging, and a wide range of diseases including cancer, diabetes, and neurodegeneration. The regulation of mtDNA replication and its role in mitochondrial biogenesis remains a mystery [1]. Methods used to study the regulation of nuclear DNA replication are, in theory, applicable to mtDNA applications, but have faced challenges in practice. A new technique that combines the Click-iT® EdU assay with signal amplification technology is demonstrated for the sensitive and reliable measurement of nascent mtDNA synthesis.
Advantages Abound with Click-iT® EdU
Traditional methods for monitoring nascent nuclear DNA synthesis follow the incorporation of a nucleoside analog of thymidine into DNA. Detection is accomplished with either radioactive [3H]-thymidine labeling, antibody-based detection of BrdU (5-bromo-2’-deoxyuridine) incorporation, or click chemistry–based detection of EdU (5-ethynyl-2’-deoxyuridine) incorporation. Directly visualizing mtDNA synthesis with any of these analogs can be challenging due to the weak signal obtained from the ~17 kb mitochondrial genome, even when nuclear DNA replication is inhibited as part of the labeling and detection strategy.
Of the three thymidine analog–based methods described, the Click-iT® EdU assay is by far the easiest to perform and offers the most benefits [2]. This assay eliminates not only the use of radioactivity but also the harsh DNA denaturation step required with the BrdU method (Figure 1). EdU detection is based on a click reaction—a copper(I)-catalyzed reaction between an azide and an alkyne [3]. EdU contains an alkyne that reacts with a fluorescent azide to form a very stable reaction product.
Figure 1. Detection of incorporated EdU with Alexa Fluor® 488 azide dye compared to detection of incorporated BrdU with an anti-BrdU antibody. The small size of the Alexa Fluor® 488 azide dye eliminates the need to denature DNA in order for the detection reagent to gain access to the modified base.
Signal Amplification Exposes mtDNA Synthesis
In order to visualize nascent mtDNA, the Click-iT® EdU assay requires signal amplification (Figure 2). Tyramide Signal Amplification (TSA®) technology (PerkinElmer) is compatible with Click-iT® EdU and has been reported to increase detection sensitivity up to 100-fold compared to conventional avidin-biotinylated enzyme complex (ABC) procedures [4].
TSA® is an enzyme-mediated detection method that utilizes the catalytic activity of horseradish peroxidase (HRP) to generate high-density labeling of a target antigen (Figure 3). In this technique, the target antigen is the fluorescent azide (Oregon Green® 488 or tetramethylrhodamine (TAMRA™) dye) used to detect DNA that has incorporated EdU. An anti-dye antibody (either HRP-conjugated or unconjugated) followed by a secondary antibody–HRP conjugate is combined with a TSA® analog with fluorescence emission comparable to the original fluorescent azide. When amplifying the Oregon Green® 488 dye signal, a green-fluorescent Alexa Fluor® 488 TSA® kit is recommended. When amplifying the TAMRA™ dye signal, either Alexa Fluor® 555 or Alexa Fluor® 546 dye can be used to generate a red-orange fluorescence emission similar to the TAMRA™ dye.
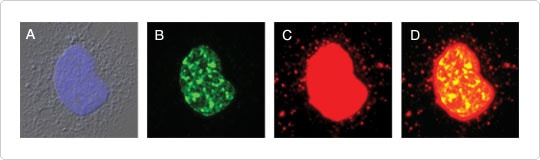
Figure 2. mtDNA synthesis visualized using Click-iT® EdU and signal amplification. F11 neuroblastoma cells were incubated with 10 μM EdU from the Click-iT® EdU Microplate Assay for 24 hr. EdU incorporated into DNA was detected using the Oregon Green® 488 azide included in the kit. The signal was amplified using the anti–Oregon Green® HRP-conjugated antibody (also a component of the kit) together with the red-fluorescent Alexa Fluor® 594 tyramide from TSA® Kit #5. (A) Nucleus stained with DAPI. (B) EdU incorporation without signal amplification, showing nuclear DNA synthesis. (C) EdU incorporation with signal amplification, revealing mtDNA in addition to nuclear DNA synthesis. (D) Overlay of panels B and C. Image contributed by Stephen I. Lentz and Eva L. Feldman, Departments of Internal Medicine and Neurology, University of Michigan.
 | Figure 3. Schematic representation of TSA® detection. |
Click-iT® EdU and TSA® Detection In Action
Mitochondrial biogenesis enables neurons to meet changing energy loads and to redistribute mitochondria throughout the neuron. One approach for monitoring mitochondrial biogenesis is to measure the rate of mtDNA replication. The method of amplifying the Click-iT® EdU signal with TSA® technology was recently used in combination with the pan-neuronal marker αTuj1 to visualize mtDNA synthesis in adult dorsal root ganglion neurons (Figure 4) [5].
By combining the streamlined Click-iT® EdU assay with ultrasensitive TSA® detection, a superior method is established to explore the biology of mtDNA replication and turnover, and for toxicological profiling of drugs which inhibit mtDNA synthesis.
 | Figure 4. mtDNA replication in dissociated dorsal root ganglion neurons. Neurons plated on glass coverslips were incubated with 10 μM EdU. Following fixation and permeabilization, endogenous peroxidase activity was blocked and EdU was detected with Oregon Green® 488 azide. The signal was amplified using HRP-conjugated rabbit antibody against Oregon Green® 488 and Alexa Fluor® 488 tyramide (green) from TSA® Kit #12. The pan-neuronal marker αTuj1 was detected with a mouse primary antibody and visualized with an Alexa Fluor® 594 goat anti-mouse secondary antibody (red). Nuclei were stained with DAPI (blue). Image contributed by Stephen I. Lentz and Eva L. Feldman, Departments of Internal Medicine and Neurology, University of Michigan. |
- Clay Montier LL, Deng J, Bai Y (2009) J Genet Genomics 36:125–131.
- Yu Y, Arora A, Min W et al (2009) J Immunol Methods 350:29–35.
- Salic A, Mitchison TJ (2008) Proc Natl Acad Sci U S A 105:2415–2420.
- Van Heusden J, de Jong P, Ramaekers F et al. (1997) J Histochem Cytochem 45:315–319.
- Lentz SI, Edwards JL, Backus C et al (2009) J Histochem Cytochem Nov 3 Epub.
See a complete listing of the products discussed in this article.
View products


