Search
The relatively new discovery that pluripotent human cells can be derived from somatic cells through the expression of exogenous genes has generated great excitement and promise in the fields of biology and cell therapy [1–5]. These induced pluripotent stem cells (iPSCs) can be used to further our understanding of basic cellular mechanisms, discover new therapeutic drugs, and treat illnesses through cell therapy. Unlike embryonic stem cells (ESCs), iPSCs do not present any cultural or ethical barriers for use, are derived from somatic cells that are plentiful, and can potentially overcome immune rejection issues when used for cell therapy. Invitrogen’s broad technology platform provides optimized tools for all aspects of iPSC research—from the isolation and expansion of somatic cells to the validation and differentiation of iPSCs. Complete growth systems for isolating and expanding somatic cells are optimized for either keratinocytes or fibroblasts. For reprogramming, the drug-inducible ViraPower™ HiPerform™ iPSC lentiviral particles enable precise gene delivery into your desired cell type, and the Neon™ Transfection System allows efficient delivery of vectors into somatic cells.
Isolating and Expanding Keratinocytes
The most common cell types used to generate induced pluripotent stem cells are derived from skin. Although the majority of studies to date have utilized fibroblasts for reprogramming efforts, more recently it has been demonstrated that epidermal keratinocytes can be reprogrammed with higher frequency [1]. This may be particularly significant for cell therapy applications because keratinocytes are the predominant cell type in skin, especially in the adult, and exhibit a longer in vitro lifespan. A variety of culture media and supplements are available to support the growth of normal human skin cells, as well as reagents for their isolation, subculture, and cryopreservation (Table 1).Two growth systems—extended-lifespan and classic—are available for keratinocyte culture. The extended-lifespan growth system family features the most advanced basal medium for human keratinocyte culture, EpiLife® Basal Medium. This medium more than doubles the in vitro lifespan of human keratinocytes when compared to classic growth media (Figure 1). When combined with a single-addition growth supplement, EpiLife® Medium supports the rapid proliferation of human keratinocytes with culture lifespans of greater than 50 population doublings, even in samples from adult donors.
Supplements available for EpiLife® Basal Medium range from the defined, animal origin–free Supplement S7, to the standard supplement (Human Keratinocyte Growth Supplement) containing bovine pituitary extract. The classic keratinocyte growth systems include the tried-and-true Keratinocyte-SFM, which contains bovine pituitary extract, and the defined, xeno-free medium Defined Keratinocyte-SFM.
 | Figure 1. Extended lifespan of keratinocytes grown in EpiLife® medium compared to growth in a competitor’s medium. Normal Neonatal Human Keratinocytes were grown in EpiLife® medium and in a keratinocyte medium from a leading competitor. Cultures grown in EpiLife® medium demonstrated population doublings over an extended period compared to cells grown in the competitor’s medium. |
Table 1. Cells and Reagents for Somatic Cell Isolation, Expansion, and Reprogramming.
| Product | Quantity |
|---|---|
| KERATINOCYTE ISOLATION AND EXPANSION | |
| Human Epidermal Keratinocytes, adult (HEKa) | ≥500,000 viable cells |
| Human Epidermal Keratinocytes, neonatal (HEKn) | ≥500,000 viable cells |
| EpiLife® Medium | 500 mL |
| Supplement S7 | 5 mL |
| Human Keratinocyte Growth Supplement | 5 mL, 100X |
| EpiLife® Defined Growth Supplement | 5 mL, 100X |
| Defined Keratinocyte-SFM | 500 mL |
| Keratinocyte-SFM | 500 mL |
| FIBROBLAST ISOLATION AND EXPANSION | |
| Human Dermal Fibroblasts, adult (HDFa) | ≥500,000 viable cells |
| Human Dermal Fibroblasts, neonatal (HDFn) | ≥500,000 viable cells |
| Medium 106 | 500 mL |
| Low Serum Growth Supplement | 10 mL, 50X |
| Synth-a-Freeze® Cryopreservation Medium | 50 mL |
| Fetal Bovine Serum, ES Cell-Qualified | 500 mL |
| SOMATIC CELL REPROGRAMMING | |
| iPSC Lentivirus—Nanog | 100 μL, 6.92 x 107 particles |
| iPSC Lentivirus—Klf4 | 100 μL, 9.90 x 107 particles |
| iPSC Lentivirus—Lin28 | 100 μL, 3.64 x 107 particles |
| iPSC Lentivirus—Sox2 | 100 μL, 1.57 x 105 particles |
| iPSC Lentivirus—Oct4 | 100 μL, 6.50 x 106 particles |
| iPSC Lentivirus—cMyc | 100 μL, 4.90 x 107 particles |
Isolating and Expanding Fibroblasts
For fibroblasts, both low-serum and classic serum–containing media are available (Table 1). Medium 106 has been optimized to support the growth of dermal fibroblasts in a reduced-serum environment. When combined with Low Serum Growth Supplement, Medium 106 supports the optimal growth of dermal fibroblasts in a 2% fetal bovine serum environment. The classic growth systems utilize high-quality GIBCO® DMEM supplemented with highly qualified fetal bovine serum. Dermal fibroblasts can be isolated and serially propagated in either of these systems utilizing the TrypLE™ subculture reagent, which maintains high viability.In addition to these serum-containing fibroblast growth systems, a xeno-free fibroblast growth system is in development. Cryopreserved dermal fibroblasts from either neonatal foreskin or adult skin are also available and are ready to use for reprogramming experiments.
Save Time with Ready-to-Use Somatic Cells
Methods for Reprogramming
Retroviruses and lentiviruses are randomly integrating viruses that are extremely efficient at transfecting multiple cell types and provide high levels of short-term expression of the required transgenes. While retroviruses are only able to infect dividing cells, lentiviruses can infect both dividing and nondividing cells [6], making them a preferred choice for reprogramming experiments.
Deliver Reprogramming Factors with Maximum Efficiency
Drug-inducible lentiviral particles allow temporal control of reprogramming factor expression, providing efficient tools for reprogramming factor screens and optimization of reprogramming conditions. ViraPower™ HiPerform™ iPSC lentiviral particles were constructed with the pLenti6.3 tetO DEST vector, allowing inducible expression of the reprogramming factor(s) of interest and precise control of expression level. Cells cotransduced with Lenti6.3/TetR virus particles and ViraPower™ HiPerform™ iPSC lentivirus particles do not express the reprogramming factors until the addition of tetracycline.
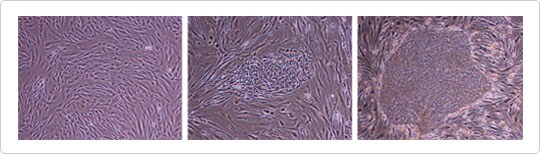 |
Figure 2. Human dermal fibroblasts reprogrammed with ViraPower™ HiPerform™ iPSC lentiviral particles. (Left) Human dermal fibroblasts (HDFs) from a 34-year old female were transduced at day 0 with ViraPower™ HiPerform™ iPSC lentiviral particles containing Oct4, Sox2, Klf4, and c-Myc. (Center) iPSC colony surrounded by HDFs at day 14. (Right) Large iPSC colony at day 75. Data provided by Yasuyuki Asai, ReproCELL Inc., Tokyo, Japan. |
- Aasen T, Raya A, Barrero MJ et al. (2008) Nat Biotechnol 26:1276–1284.
- Takahashi K, Yamanaka S (2006) Cell 126:663–676.
- Takahashi K, Tanabe K, Ohnuki M et al. (2007) Cell 131:861–872.
- Yu J, Vodyanik MA, Smuga-Otto K et al. (2007) Science 318:1917–1920.
- Maherali N, Hochedlinger K (2008) Cell Stem Cell 3:595–605.
- Naldini L, Blömer U, Gallay P et al. (1996) Science 272:263–267.
Find more information on our stem cell research reagents.
- Learn More about Tools for Induced Pluripotent Stem Cell Research
> Get a copy of this article as it appears in the print version of BioProbes 62.
See a complete listing of the products discussed in this article.
View products
