Search
Streamline Your siRNA Transfections siPORT™ NeoFX™ Transfection Agent Promotes High Reproducibility and Efficiency
- Fast--transfect cells as they are plated (reverse transfection), saving a day
- Reproducible--produces consistent results, lot-to-lot, plate-to-plate, and well-to-well
- Versatile--works with a broad range of cell lines
- Efficient--performs with high siRNA transfection efficiency, allowing use of low siRNA concentrations to minimize nonspecific effects
- Powerful--performs with low cytotoxicity in the presence or absence of serum
Streamlined Transfection Protocol
siRNA transfection is traditionally a two-day procedure, making optimization a relatively slow process. siPORT NeoFX Transfection Agent was developed to refine siRNA transfection protocols. siPORT NeoFX lipid-based formulation can be used to efficiently transfect adherent cells just after trypsinization--without increased cytotoxicity. Just add siPORT NeoFX to your diluted siRNA, incubate to form transfection complexes, add the complexes to the culture wells, and overlay with cells (Figure 1). This new streamlined protocol, named "neofection," will save researchers a full day of valuable time when compared to traditional pre-plated transfection procedures. The transfection complexes are active and stable even in the presence of serum, so there is no need to remove or replace media following transfection. From start to finish, successful gene silencing experiments can be completed in less than 24 hours. This streamlined protocol can be adapted to a wide range of cells and experimental designs, including high-throughput applications.
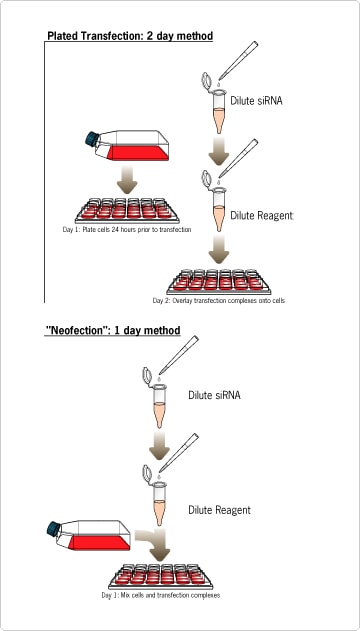
Figure 1. Traditional Plated Transfection vs. Neofection with siPORT™ NeoFX™

Figure 1. Traditional Plated Transfection vs. Neofection with siPORT™ NeoFX™
Optimized for Lower siRNA Concentrations
Recent reports indicate that siRNA concentrations of 100 nM or higher can lead to nonspecific changes in gene expression (off-target effects) in mammalian cultured cells [1-3]. Reducing the amount of siRNA used for transfections to 1-20 nM minimizes these nonspecific effects, while still providing extensive silencing of the target gene [3]. Ambion scientists specifically developed the siPORT
NeoFX Transfection Agent for efficient transfection of low concentrations of siRNA. For example, high levels of siPORT
NeoFX-mediated gene silencing (>80%) were achieved by transfection of as little as 0.1 nM siRNA (targeting GAPDH) into HeLa cells (Figure 2). Using 3 and 10 nM siRNA in siPORT
NeoFX transfection complexes, substantial reduction of target gene expression was observed in many adherent, human cell lines, including MCF-7 (breast adenocarcinoma cells), SKOV3 (ovarian cancer cells), A549 (lung carcinoma cells), SKNAS (bone marrow neuroblastoma cells), and BJ cells (primary foreskin fibroblast cells) (data not shown).
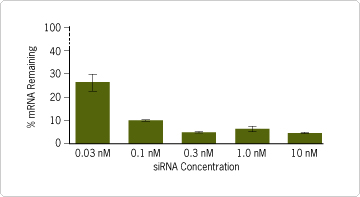
Figure 2. Efficient Transfection with Low siRNA Concentration. HeLa cells were trypsinized and resuspended in growth media at a concentration of 4x10 4 cells/ml. Transfection complexes were prepared using chemically synthesized GAPDH siRNA (0.03 nM to 10 nM) or negative control siRNA (data not shown) and 0.5 µl siPORT™ NeoFX™ Transfection Agent. 48 hours after transfection, cells were harvested and analyzed by real-time RT-PCR for both GAPDH mRNA and 18S rRNA levels. Percent mRNA remaining was calculated as the amount of GAPDH mRNA in cells transfected with GAPDH siRNA compared to that of cells transfected with the negative control siRNA. Data were normalized to the 18S rRNA signal. These results demonstrate that siPORT NeoFX performs well even with reduced levels of siRNA, which may offer significant benefits for eliminating off-target effects in gene-silencing applications.

Figure 2. Efficient Transfection with Low siRNA Concentration. HeLa cells were trypsinized and resuspended in growth media at a concentration of 4x10 4 cells/ml. Transfection complexes were prepared using chemically synthesized GAPDH siRNA (0.03 nM to 10 nM) or negative control siRNA (data not shown) and 0.5 µl siPORT™ NeoFX™ Transfection Agent. 48 hours after transfection, cells were harvested and analyzed by real-time RT-PCR for both GAPDH mRNA and 18S rRNA levels. Percent mRNA remaining was calculated as the amount of GAPDH mRNA in cells transfected with GAPDH siRNA compared to that of cells transfected with the negative control siRNA. Data were normalized to the 18S rRNA signal. These results demonstrate that siPORT NeoFX performs well even with reduced levels of siRNA, which may offer significant benefits for eliminating off-target effects in gene-silencing applications.
High siRNA Transfection Efficiency with Low Cytotoxicity
siPORT NeoFX can achieve a high level of siRNA transfection and knockdown efficiency while maintaining a high level of cell viability. In a representative transfection experiment, Cy™3-labeled siRNA was successfully delivered into a high percentage of HeLa cells (Figure 3A). Furthermore, in comparison with other commercially available transfection agents, siPORT NeoFX maintained the highest level of cell viability while exhibiting excellent gene silencing activity in HepG2 cells (Figure 3B).
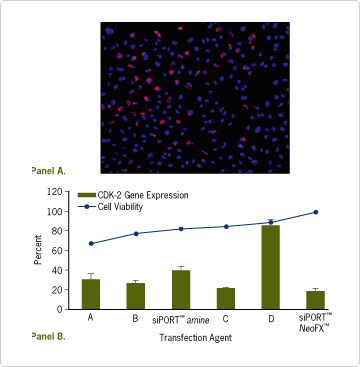
Figure 3. High Efficiency and Low Cytotoxicity of siPORT™ NeoFX™ Transfection Agent. (A) HeLa cells were transfected with 30 nM Cy™3-labeled siRNA using siPORT NeoFX Transfection Agent. Cells were subsequently fixed, stained with DAPI for visualization of nuclei, and analyzed by fluorescence microscopy. Blue: DAPI stained nuclei; Red: Cy3 fluorescent siRNA. (B) CDK2 siRNA or Silencer™ Negative Control siRNA #1 was complexed with six different transfection agents according to manufacturer's instructions and added to empty wells of a 96 well dish (final siRNA concentration of 30 nM). HepG2 cells were added to the wells and cultured for 48 hours in complete media containing serum. Cell viability was analyzed by microflow cytometry using PCA 96 instrument and Viacount™ assay (Guava Technologies). RNA was isolated, and target gene expression was quantified by real time RT-PCR. Relative reduction in mRNA levels is expressed as a percentage of expression in cells transfected with a negative control siRNA. Duplicate samples were normalized by measuring 18S rRNA levels by real time RT-PCR. The range of normalized gene expression is shown with error bars.

Figure 3. High Efficiency and Low Cytotoxicity of siPORT™ NeoFX™ Transfection Agent. (A) HeLa cells were transfected with 30 nM Cy™3-labeled siRNA using siPORT NeoFX Transfection Agent. Cells were subsequently fixed, stained with DAPI for visualization of nuclei, and analyzed by fluorescence microscopy. Blue: DAPI stained nuclei; Red: Cy3 fluorescent siRNA. (B) CDK2 siRNA or Silencer™ Negative Control siRNA #1 was complexed with six different transfection agents according to manufacturer's instructions and added to empty wells of a 96 well dish (final siRNA concentration of 30 nM). HepG2 cells were added to the wells and cultured for 48 hours in complete media containing serum. Cell viability was analyzed by microflow cytometry using PCA 96 instrument and Viacount™ assay (Guava Technologies). RNA was isolated, and target gene expression was quantified by real time RT-PCR. Relative reduction in mRNA levels is expressed as a percentage of expression in cells transfected with a negative control siRNA. Duplicate samples were normalized by measuring 18S rRNA levels by real time RT-PCR. The range of normalized gene expression is shown with error bars.
Lot-to-lot, Day-to-day, Plate-to-plate, and Well-to-well Consistency in Performance
In addition to efficient siRNA transfection with low cytotoxicity, a good transfection agent is also expected to reliably deliver high performance among multiple wells for replicate samples and between different lots of the transfection agent. Such characteristics are especially important in high throughput screening of siRNA libraries arrayed in 96 well plates. As shown in Figure 4, there was negligible variability in gene silencing efficiency between multiple wells and multiple plates when siPORT
NeoFX was used in experiments on two different days. Further, three different lots of siPORT
NeoFX were found to give consistent knockdown efficiency (data not shown).

Figure 4. Consistent Day-to-day, Plate-to-plate, and Well-to-well Performance. 8,000 HeLa cells were transfected in 96 well plates using siPORT™ NeoFX™ (0.3 µl/100 µl reaction volume) and either GAPDH siRNA (10 nM) or a scrambled negative control siRNA. Remaining GAPDH expression was quantified as described in Figure 2. Each bar represents the mean of eight replicate wells. siPORT NeoFX demonstrates consistent transfection performance across multiple wells, among multiple plates, and on multiple days.

Figure 4. Consistent Day-to-day, Plate-to-plate, and Well-to-well Performance. 8,000 HeLa cells were transfected in 96 well plates using siPORT™ NeoFX™ (0.3 µl/100 µl reaction volume) and either GAPDH siRNA (10 nM) or a scrambled negative control siRNA. Remaining GAPDH expression was quantified as described in Figure 2. Each bar represents the mean of eight replicate wells. siPORT NeoFX demonstrates consistent transfection performance across multiple wells, among multiple plates, and on multiple days.
Solutions for siRNA Delivery
Although the RNAi field is still young, the use of RNAi has already been demonstrated to have enormous potential for understanding gene function, elucidating biological pathways, and identifying and validating potential drug targets. Ambion is committed to bringing researchers the best solutions for siRNA delivery. Ambion's line of transfection and electroporation products offers consistent and reliable performance. siPORT NeoFX Transfection Agent is unique in that it can be used during seeding of cells into culture dishes, thereby streamlining the transfection protocol and shortening the procedure by an entire day.
References1. Jackson AL, Bartz SR, Schelter J, Kobayashi SV, Burchard J, Mao M, Li B, Cavet G, Linsley PS (2003) Expression profiling reveals off-target gene regulation by RNAi. Nat Biotechnol 21: 635-7.
2. Semizarov D, Frost L, Sarthy A, Kroeger P, Halbert DN, Fesik SW (2003) Specificity of short interfering RNA determined through gene expression signatures. Proc Natl Acad Sci USA 100: 6347-52.
3. Persengiev SP, Zhu X, Green MR (2004) Nonspecific, concentration-dependent stimulation and repression of mammalian gene expression by small interfering RNAs (siRNAs) RNA 10: 12-18.
2. Semizarov D, Frost L, Sarthy A, Kroeger P, Halbert DN, Fesik SW (2003) Specificity of short interfering RNA determined through gene expression signatures. Proc Natl Acad Sci USA 100: 6347-52.
3. Persengiev SP, Zhu X, Green MR (2004) Nonspecific, concentration-dependent stimulation and repression of mammalian gene expression by small interfering RNAs (siRNAs) RNA 10: 12-18.