Search
Simple and Accurate Monitoring of Adipogenesis
Introduction
During human development, pluripotent stem cells (PSCs) differentiate into the three distinct primary germ layers known as the mesoderm, ectoderm, and endoderm. Each of these germ layers is responsible for creating its own specific types of the over 220 different cell types in the body. Adipocytes, or fat cells, are one of the many cell types generated from the mesoderm.
The differentiation of mesoderm cells into adipocytes is called adipogenesis and is driven by the activation of specific transcription factors and proteins, including PPARγ, C/EBPα, GLUT4, and FABP4 [1]. Adipocyte function is primarily characterized by the storage of lipid reserves in adiposomes, organelles specifically found in this cell type. Adipogenesis is a widely studied area of cell biology and medicine, with implications in non-shivering thermogenesis, obesity, diabetes, cardiovascular disease, and a host of other diseases and cellular processes [2,3]. In a laboratory setting, adipocytes can be routinely differentiated from fibroblasts by a variety of methods [4].
Adipogenesis can be studied in a number of ways, with varying degrees of difficulty [4]. One simple method to determine how efficiently a population of fibroblasts has differentiated into adipocytes is to use a fluorescent neutral lipid stain. Invitrogen HCS LipidTOX neutral lipid stains label lipids in live and fixed cells and are available in green, red, and deep red versions. The availability of multiple stain colors allows for flexibility when multiplexing with other probes, enabling acquisition of additional details about proteins of interest and cell health in adipogenesis. The HCS LipidTOX stains are commonly used in toxicological profiling in hepatocyte models to detect excess accumulation of neutral lipids, known as steatosis. They also prominently label lipid-rich adiposomes in adipocytes. These reagents, combined with the appropriate instrumentation, enable simple and efficient examination of adipogenesis. Here we demonstrate the measurement of adipogenesis over the course of differentiation from mouse fibroblasts, using the HCS LipidTOX Green Neutral Lipid Stain in both live and fixed cells.
Adipocyte differentiation protocol
Materials
Methods
Adipocyte differentiation: 3T3-L1 mouse fibroblasts were grown in DMEM supplemented with 10% FBS for 17 days and were passaged every other day. At each passage, cell samples were harvested and divided into either Gibco StemPro Adipocyte Differentiation Medium or fresh DMEM with FBS. This staggered cell culture approach provided samples with and without exposure to differentiation media, in 2-day increments across the entire time course, that could be harvested, fixed, and analyzed on day 17. Alternatively, for analysis of adipogenesis in live cells over time, 3T3-L1 mouse fibroblasts can be stained with HCS LipidTOX neutral lipid stains and placed in StemPro Adipocyte Differentiation Medium for ongoing culture and differentiation.
Adipogenesis workflow
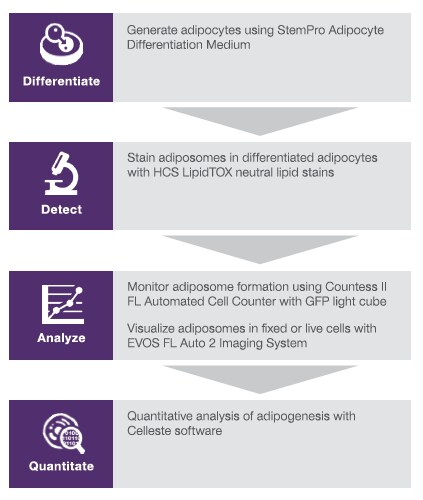
Tips and troubleshooting for adipocyte differentiation
- Expansion culture: Expand primary mesenchymal stem cells (MSCs) with Gibco MesenPRO RS Medium (reduced-serum medium), CTS StemPro MSC SFM (serum-free medium), or StemPro MSC SFM XenoFree (serum-free, xeno-free medium) in T-75 or T-150 fl asks. We have successfully used a standard growth medium of DMEM plus 10% MSC-qualifi ed FBS. We recommend refeeding the cultures every 2–3 days and passaging every 5–7 days.
- Passaging: We strongly recommend using low– passage number MSCs (<8–10 passages). Continually passaged MSCs will gradually lose their multipotency with increased passage numbers (>10 passages).
- Harvesting: We recommend using Gibco TrypLE Express Enzyme to enzymatically treat and harvest MSCs. TrypLE Express is a recombinant protease that has been demonstrated to be gentle on MSCs. Overexposure to trypsin will lead to reduced MSC viability and expansion.
- Timing of passaging: Do not let passaged MSCs become completely confl uent, as it can reduce the multipotency of MSCs. Passage cultures when they reach 60–80% confluency, cell viability is at least 90%, and growth is in the mid-logarithmic phase.
- Seeding density: For expansion, we recommend a seeding density of 3 x 103 to 5 x 103 viable cells/cm2 with MesenPRO RS Medium, or 1 x 104 viable cells/cm2 with StemPro MSC SFM.
- Confluency: Expanding MSCs in growth medium for 2–4 days before refeeding with complete StemPro Adipocyte Differentiation Medium can enhance adipogenesis.
Automated analysis of differentiated adipocytes
For rapid and reliable analysis of adipogenesis, an automated fluorescence cell counter such as the Invitrogen Countess II FL Automated Cell Counter can be used. The Countess II FL system is a benchtop cell counter with fluorescence detection capabilities, allowing for quick and accurate monitoring of adipogenesis when used with the HCS LipidTOX neutral lipid stains. This simple approach can be used to monitor differentiation for time-point honing during a standard cell culture workflow before proceeding with extensive analyses involving more complex instrumentation and multiplex labeling protocols. Here we demonstrate the measurement of adipogenesis over the course of differentiation from mouse fibroblasts, in fixed cells using the HCS LipidTOX Green Neutral Lipid Stain and the Countess II FL system.
Materials
- HCS LipidTOX Green Neutral Lipid Stain, for cellular imaging (Cat. No. H34475)
- Gibco DPBS, no calcium, no magnesium (Cat. No. 14190144)
- Countess II FL Automated Cell Counter (Cat. No. AMQAF1000)
- Invitrogen EVOS Light Cube, GFP (Cat. No. AMEP4651)
- Invitrogen Countess Cell Counting Chamber Slides (Cat. No. C10283)
Methods
After adipocyte differentiation, trypsin-harvested cells were pelleted at 180 x g for 5 min followed by replacement of the medium with 4% formaldehyde in DPBS for 15 min at room temperature. Samples were then washed twice with DPBS using the same centrifuge settings. Cells were labeled with HCS LipidTOX Green Neutral Lipid Stain (diluted 1:200 in DPBS) for 45 min at room temperature before analysis on the Countess II FL system using the GFP light cube.
Results
3T3-L1 cells cultured in differentiation medium showed a time-dependent increase in adipogenesis, as measured by fluorescently labeled adiposomes in adipocytes (Figure 1). After exposure to differentiation medium, the percentage of adipocytes in the population increased to approximately 60% of the cell population by day 15 (Figure 2).

Figure 1. Analysis of adipogenesis in fixed cells. Analysis of cells on the Countess II FL system on days 0, 7, 9, and 17 shows an increase in the number of adipocytes as the time spent by the cells in the differentiation medium increases.
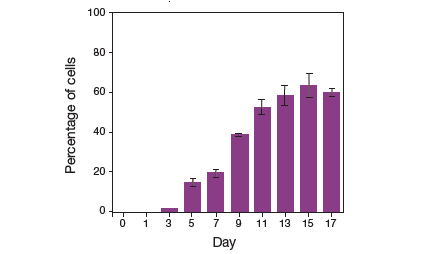
Figure 2. Quantitative comparison of adipocyte numbers during different stages of adipogenesis. Analysis of mouse fibroblasts undergoing adipogenesis shows an increase in the number of adipocytes to approximately 60% of the cell population by day 15.
Imaging and analysis of differentiated adipocytes
For more extensive analysis and quantitation or for multiplexing with other probes, fluorescence microscopy can be used. The Invitrogen EVOS FL Auto 2 Imaging System with Invitrogen Celleste Image Analysis Software is an ideal microscope and image analysis package to perform these types of experiments. Here we use simplified methods to obtain live- and fixed-cell images on the EVOS FL Auto 2 Imaging System to visualize adiposomes labeled with HCS LipidTOX Green Neutral Lipid Stain in mouse fibroblasts that have been differentiated into adipocytes. In addition, we show quantitative analysis of adipogenesis with Celleste software.
Materials
- EVOS FL Auto 2 Imaging System (Cat. No. AMAFD2000)
- Invitrogen NucBlue Live ReadyProbes Reagent (Cat. No. R37605)
- StemPro Adipogenesis Differentiation Kit (Cat. No. A1007001)
- Invitrogen Alpha Tubulin Monoclonal Antibody (Cat. No. 236-10501 and A11126)
- HCS LipidTOX Green Neutral Lipid Stain, for cellular imaging (Cat. No. H34475)
- Invitrogen HCS CellMask Blue Stain (Cat. No. H32720)
- Invitrogen Goat Anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 647 (Cat. No. A21236)
- Invitrogen EVOS 20x Objective, fluorite, coverslip-corrected (Cat. No. AMEP4698)
- Invitrogen Olympus 60x Oil Objective, apochromat, coverslip-corrected (Cat. No. AMEP4694)
- EVOS Light Cube, GFP (Cat. No. AMEP4651)
- EVOS Light Cube, DAPI (Cat. No. AMEP4650)
- EVOS Light Cube, Cy5 (Cat. No. AMEP4656)
- Invitrogen ProLong Diamond Antifade Mountant (Cat. No. P36970)
- Celleste Image Analysis Software (Cat. No. AMEP4816)
Methods
Visualization of adiposomes for multiplex imaging
After adipocyte differentiation, cells were fixed in paraformaldehyde and permeabilized with 0.2% Triton™ X-100 (Note: This fixation method may not be optimal for the type of experiment) in PBS for 15 min, followed by 3 washes in PBS, a 1 hr block with 3% BSA, and incubation with 1 μg/mL of anti–alpha tubulin antibody in blocking buffer for 1 hr. Cells were then washed 3 times with PBS and incubated for 45 min with 2 μg/mL of goat anti-mouse secondary antibody, Alexa Fluor 647 conjugate. After antibody staining, cells were washed 3 times with PBS, followed by incubation with HCS LipidTOX Green Neutral Lipid Stain diluted 1:200 in PBS for 30 min and 3 additional PBS washes. Samples were then incubated with 2 drops of NucBlue Live ReadyProbes Reagent per milliliter of PBS for 15 min, washed 3 times with PBS, and mounted in ProLong Diamond Antifade Mountant. Samples were imaged on the EVOS FL Auto 2 Imaging System in the DAPI, GFP, and Cy5 channels using an Olympus 60x oil objective.
Quantitative analysis of adipogenesis in fixed cells
After fixation, cells were washed 3 times with PBS, then labeled with 1:5,000 HCS CellMask Blue Stain and 1:200 HCS LipidTOX Green Neutral Lipid Stain in PBS for 30 min. Cells were washed 3 times with PBS, then mounted in ProLong Diamond Antifade Mountant. Samples were imaged on the EVOS FL Auto 2 Imaging System in the DAPI and GFP channels using an EVOS 20x objective. Quantitative analysis was performed using Celleste software.
Qualitative analysis of adipogenesis in live cells
3T3-L1 mouse fibroblasts were seeded at a density of 50,000 cells/well in a 6-well plate. After overnight incubation in normal growth serum, one well was labeled with 1:200 HCS LipidTOX Green Neutral Lipid Stain for 30 min at 37°C with 5% CO2. Cells were imaged in normal cell culture media on the EVOS FL Auto 2 Imaging System with GFP and transmitted light channels using an EVOS 20X Objective. After imaging, cells were placed in the StemPro Adipocyte Differentiation Medium for ongoing culture and differentiation. Cells were again labeled and imaged in the above-mentioned manner at 5-, 7-, 9-, 11-, and 14-day time points.
Results
High-resolution images of fixed cells that have been in differentiation medium for 22 days show formation of large adiposomes (Figure 3). In live-cell experiments, 3T3-L1 mouse fibroblasts show an increase in adiposome number and size as they differentiate into adipocytes in differentiation medium (Figure 4). Quantitative analysis of undifferentiated 3T3-L1 cells and 3T3-L1 cells that have been in differentiation medium for 22 days shows an increase in neutral lipid staining for cells in differentiation medium, compared to the cells left in standard growth medium (Figure 5).
Conclusion
This study demonstrates expected results in terms of fibroblast differentiation into adipocytes and shows that adipogenesis can be easily and accurately monitored using HCS LipidTOX neutral lipid stains. Automated fluorescence cell counting using the Countess II FL system is a simple approach for monitoring differentiation for time-point honing during a standard cell culture workflow, and a useful consideration before proceeding with extensive analyses involving more complex instrumentation and multiplex labeling protocols. For more extensive analysis, fluorescence microscopy and image analysis software are useful tools. In live and fixed cells, adipogenesis efficiency can easily and accurately be quantitatively or qualitatively analyzed using the HCS LipidTOX neutral lipid stains, EVOS FL Auto 2 Imaging System, and Celleste Image Analysis Software. Multiplexing with other probes allows for the investigation of other factors that affect adipogenesis, such as cell health, providing robust data on this important area of cell and developmental biology.
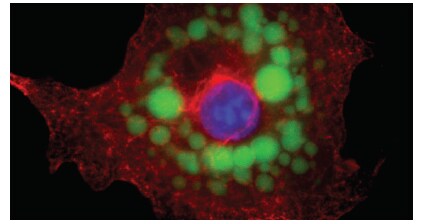
Figure 3. High-resolution spatial imaging of fixed adipocytes. 3T3-L1 mouse fibroblasts were cultured using the StemPro Adipogenesis Differentiation Kit, then fixed and labeled with the HCS LipidTOX Green Neutral Lipid Stain for adiposomes, NucBlue Live ReadyProbes Reagent for the nucleus, and an anti–alpha tubulin monoclonal primary antibody followed with an AlexaFluor 647–conjugated secondary antibody, for microtubules. An adipocyte imaged on the EVOS FL Auto 2 Imaging System with an Olympus 60x oil objective shows large adiposomes surrounding the nucleus.
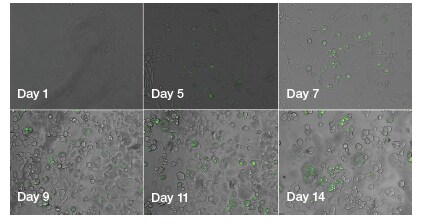
Figure 4. Qualitative time-course monitoring of adipogenesis in live cells by visualization of the adiposomes. Images of 3T3-L1 cells differentiating into adipocytes taken on the EVOS FL Auto 2 Imaging System every few days show an increase in the number of adiposomes over time.
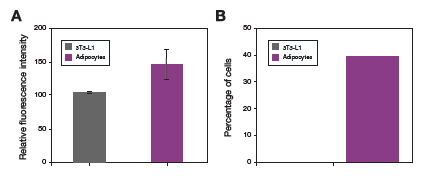
Figure 5. Quantitative analysis of adiposome formation in mouse fibroblasts. (A) Quantitative analysis of 3T3-L1 cells under normal cell culture conditions vs. 3T3-L1 cells cultured in StemPro Adipocyte Differentiation Medium for 22 days showed an increase in neutral lipid staining for the cells in differentiation medium. (B) Further analysis of the same cell populations showed that 40% of the cells grown in differentiation medium contained adiposomes, compared to 0% of the cells in standard culture medium. Analysis was performed on Celleste Image Analysis Software.
For Research Use Only. Not for use in diagnostic procedures.