Search
Laser capture microdissection (LCM)
Laser capture microdissection (LCM) enables researchers to isolate single cells or cell groups to subsequently perform DNA, gene expression, or protein analyses. In order to identify and microdissect the cells of interest, an adequate tissue staining method is essential. This study by Roozen (B.Sc.) and Dr. Van Kempen (Department of Pathology at the Radboud University Nijmegen Medical Centre in the Netherlands) demonstrates that the cresyl violet staining procedure of the Ambion® LCM Staining Kit can be successfully used on formalin-fixed and paraffin-embedded (FFPE) tissues without compromising either the yield or the quality of RNA extracted.
Paul Roozen and Leon Van Kempen
Department of Pathology
Radboud University Nijmegen Medical Center, The Netherlands
Staining of Biopsies
FFPE skin biopsies were obtained from the tissue archive of the Department of Pathology (Radboud University Nijmegen Medical Centre, The Netherlands). In order to compare the efficacy of staining and subsequent RNA isolation, 8 µm sections mounted on LCM slides were stained with 10% Mayer’s hematoxylin or cresyl violet (from the Ambion® LCM Staining Kit). A third tissue section was not stained.
Figure 1 shows an 8 µm skin section with an unstained hair follicle (A, B), or stained with Mayer’s hematoxylin (C, D) or cresyl violet from the Ambion LCM Staining Kit (E, F); before LCM and after LCM. Although hair follicles can be observed in unstained and stained tissue, cresyl violet most strongly stains the cellular component without intense staining of the acellular component. The researchers also improved the morphological quality of the tissue sections without affecting nucleic acids by using the mounting solution described by van Dijk et al. [1].

Figure 1. Cresyl Violet Strongly Stains the Cellular Component of a Skin Biopsy. Scale bar: 200 µm
RT-PCR
Hair follicles were microdissected from the tissue specimens and total RNA was isolated using the Ambion RecoverAll™ Total Nucleic Acid Isolation Kit for FFPE. RNA concentration, quality, and purity could not be accurately determined using the NanoDrop Spectrophotometer because the RNA concentration was near the instrument’s lower limit of detection (2 ng/µL). However, there was sufficient RNA to perform quantitative real-time RT-PCR to determine if the staining process had any impact on the quality or performance of the RNA.
Total RNA was prepared from both 3 and 9 microdissected follicles from unstained, Mayer’s hematoxylin stained, or cresyl violet stained preparations, converted into cDNA and analyzed for GAPDH expression using quantitative real-time PCR (Figure 2). As expected, the CT value decreased appropriately with increasing input of follicles in unstained tissue (3-fold difference = 1.6 CTs). However, use of Mayer’s hematoxylin staining had a detrimental effect on RNA analysis as measured by the increased CT value of GAPDH expression at both 3 and 9 follicles. In contrast, staining the tissue with cresyl violet from the Ambion LCM Staining Kit did not have a negative effect on RNA amplification as compared to unstained tissue.
The data demonstrate that cresyl violet staining allows for higher contrast and more intense cellular staining compared to either 10% Mayer’s hematoxylin or unstained samples, and does not interfere with downstream RNA analysis.
The study concluded that the Ambion LCM Staining Kit could successfully stain FFPE tissue sections for LCM procedures. In combination with the Ambion RecoverAll Total Nucleic Acid Isolation Kit for FFPE, the quality of the subsequently isolated RNA is comparable to that of unstained control LCM tissue.
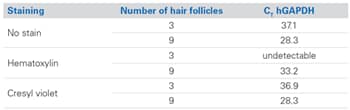
Figure 2. Real-Time RT-PCR Data from Total RNA Isolated from Unstained or Cresyl Violet-Stained FFPE Tissues. RNA (10 µL) was reverse transcribed with M-MLV reverse transcriptase and random primers. cDNA was used in real-time PCR (ABI PRISM® 7000 Sequence Detection System) to evaluate GAPDH expression using SYBR® Green and GAPDH-specific primers [2].
Next Steps
With the FFPE sample workflow validated, the laboratory has expanded the study to a larger group of tumor samples and genes. TaqMan Arrays, 384-well micro fluidic cards containing TaqMan Gene Expression Assays, are currently being used to validate microarray signatures from colon, breast, and pancreatic cancers, and translate them into qRT-PCR signatures using RNA derived from frozen and FFPE samples.
Scientific Contributor
Susan McCarthy • H. Lee Moffitt Cancer Center and Research Institute, Tampa, FL
References
- van Dijk MC, Rombout PD, Dijkman HB, Ruiter DJ, Bernsen MR (2003) Improved resolution by mounting of tissue sections for laser microdissection. Journal of Clinical Pathology: Molecular Pathology 56: 240–243.
- van Kempen LC, Rijntjes J, Mamor-Cornelissen I, Vincent-Naulleau S, Gerritsen MJ, Ruiter DJ, van Dijk MC, Geffrotin C, van Muijen GN (2008) Type I collagen expression contributes to angiogenesis and the development of deeply invasive cutaneous melanoma. International Journal of Cancer 122(5): 1019–1029