Search
The Tissue Collection: RNA Stabilization Solution
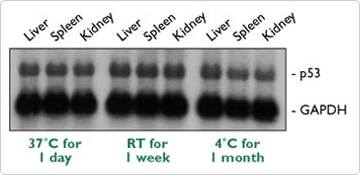
The Tissue Collection: RNA Stabilization Solution, RNAlater™ preserves and protects the RNA in fresh tissue samples from degradation prior to nucleic acid isolation. RNAlater rapidly penetrates samples and inactivates nucleases. Samples are simply placed in 5-10 volumes of RNAlater where they can then be stored refrigerated for weeks or months, before RNA isolation. Figure 1 demonstrates the quality of RNA obtained from tissue preserved in RNAlater. RNAlater provides the equivalent of quick-freezing a sample in liquid nitrogen without the inconvenience associated with processing frozen samples (i.e. grinding samples using a mortar and pestle). Although RNAlater can not preserve the RNA in previously frozen samples or rescue samples in which the RNA is already degraded, it brings a new level of standardization and reliability to sample collection by minimizing the time lag between sample procurement and RNase arrest, and simplifies the RNA extraction procedure.
Preserving RNA within tissues has many applications including the ability to collect and stabilize samples in the field or operating room (see "RNA later Around the World: Don't Leave Earth Without It"), and making it easier to use a single sample for both histology and RNA isolation (see "RNA later and Histology" below).
RNAlater™ and Histology
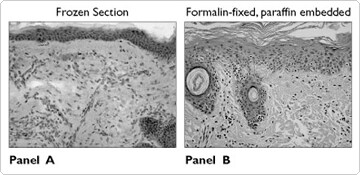
Many of our customers have inquired about the suitability of using RNAlater™ as a "holding solution" to preserve both the RNA and the histology of tissue samples collected from clinical sources. This would enable the recovery of intact RNA from samples held in reserve for pathologists. Drs. Scott Florell and Sancy Leachman at the Huntsman Cancer Institute carried out a blinded study in which two pathologists compared human tissue sections that were either immediately processed for histology (fixed in formalin and embedded in paraffin, or frozen sectioned) to samples stored in RNAlater, rinsed, and then processed for histology. Their results indicate that morphological detail and staining characteristics were identical for the 2 groups of samples. An example of their findings is presented in Figure 2. In addition to excellent staining and preservation of morphological detail, many immunohistological stains performed equally well in the RNAlater preserved samples, suggesting preservation in RNAlater caused no damage to cellular epitopes. Dr. Florell's paper is published in Modern Pathology (Mod Pathol 2001;14(2):116–128).