Search
Page Contents
This section describes Molecular Probes reagents used in the synthesis of fluorophore- or hapten-labeled peptides and fluorogenic protease substrates, as well as in peptide and protein sequencing. Some of our fluorescent probes and research chemicals have also been used for N-terminal amino acid analysis and peptide sequencing, as well as for protein fragment modification prior to Edman sequencing.
Except when it is already blocked by formylation, acetylation, pyroglutamic acid formation or other chemistry, the N-terminal amino acid of proteins can be labeled with a variety of fluorescent and chromophoric reagents from Fluorophores and Their Amine-Reactive Derivatives—Chapter 1. However, only those functional groups that survive complete protein hydrolysis, such as sulfonamides, are useful for N-terminal amino acid analysis. Dansyl chloride (D21) is the most commonly employed reagent for such analyses.![]()
Nonacylated N-terminal serine and threonine residues of proteins can be periodate-oxidized to aldehydes ![]() (Figure 9.5.1) that can then be modified by a variety of hydrazine and hydroxylamine derivatives listed in Reagents for Modifying Aldehydes and Ketones—Section 3.3. Only peptides and proteins that contain these two terminal amino acids become fluorescent, although oxidation of the carbohydrate portion of glycoproteins to aldehydes may cause interference in this analysis.
(Figure 9.5.1) that can then be modified by a variety of hydrazine and hydroxylamine derivatives listed in Reagents for Modifying Aldehydes and Ketones—Section 3.3. Only peptides and proteins that contain these two terminal amino acids become fluorescent, although oxidation of the carbohydrate portion of glycoproteins to aldehydes may cause interference in this analysis.
N-Acetylated or N-formylated proteins have been detected by transfer of the acyl group to dansyl hydrazine (D100) and subsequent chromatographic separation of the fluorescent product.![]() The sensitivity of this method can likely be improved by the use of other fluorescent hydrazine and hydroxylamine derivatives described in Reagents for Modifying Aldehydes and Ketones—Section 3.3.
The sensitivity of this method can likely be improved by the use of other fluorescent hydrazine and hydroxylamine derivatives described in Reagents for Modifying Aldehydes and Ketones—Section 3.3.
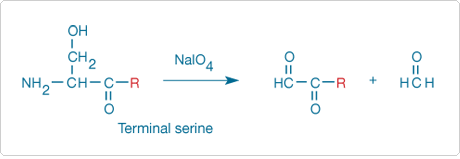
Figure 9.5.1 Sodium periodate oxidation of an N-terminal serine residue to an aldehyde, with the release of formaldehyde. The aldehyde thus formed from the protein can be subsequently modified with a variety of hydrazine, hydroxylamine or amine derivatives.
The dominant chemistry for sequencing peptides employs the nonfluorescent reagent phenyl isothiocyanate, which forms phenylthiohydantoins (PTH) in the sequencing reaction. As analogs of phenyl isothiocyanate, fluorescein-5-isothiocyanate (FITC; F143, F1906, F1907; Fluorescein, Oregon Green and Rhodamine Green Dyes—Section 1.5) and other fluorescent Isothiocyanates yield peptide conjugates that are also susceptible to Edman degradation via their thiohydantoins. Thus, these fluorescent reagents are potentially useful for ultrasensitive amino acid sequencing.![]()
Peptides specifically labeled with fluorescent dyes, haptens, photoactive groups or radioisotopes are important both as probes for receptors and as substrates for enzymes (Detecting Peptidases and Proteases—Section 10.4). Labeled peptides can be prepared by modifying isolated peptides or by incorporating the label during solid-phase synthesis. We offer several fluorescent neuropeptides, most of which are described in Probes for Neurotransmitter Receptors—Section 16.2.
Labeling Peptides in Solution
Appropriately substituted synthetic peptides can be labeled in solution by almost any of the reactive probes described in Chapters 1–5 (Labeling Small Peptides with Amine-Reactive Dyes in Organic Solvents—Note 9.2). Many peptides contain multiple residues that can be modified, potentially leading to complex mixtures of products, some of which may be biologically inactive. Modification of a peptide's thiol group by one of the thiol-reactive reagents described in Thiol-Reactive Probes—Chapter 2 is usually easy, selective and very efficient. If the peptide is synthetic, or can be modified by site-directed mutagenesis, incorporation of a cysteine residue at the desired site of labeling is recommended. The N-terminal α-amine of peptides, which has a lower pKa than the ε-amino group of lysine residues, can sometimes be labeled in the presence of other amines if the pH is kept near neutral. Conversion of tyrosine residues to o-aminotyrosines (Reagents for Modifying Alcohols—Section 3.2) can be used to provide selective sites for peptide modification, unless the tyrosine residues are essential for the biological activity of the peptide.
Solid-Phase Synthesis of Labeled Peptides
If specific labeling of peptides in solution is problematic, it may be more convenient to conjugate the fluorophore to the N-terminus of a resin-bound peptide before removal of other protecting groups and release of the labeled peptide from the resin. About five equivalents of an amine-reactive fluorophore are usually used per amine of the immobilized peptide. The fluorescein, Alexa Fluor, Oregon Green, Rhodamine Green, tetramethylrhodamine, Rhodamine Red, Texas Red, coumarin and NBD fluorophores, as well as the QSY, dabcyl and dabsyl chromophores and the biotin hapten, are all expected to be reasonably stable to hydrogen fluoride (HF) as well as to most other acids.![]() These fluorophores, chromophores and biotin are also expected to be stable to reagents used for deprotection of peptides synthesized using FMOC chemistry.
These fluorophores, chromophores and biotin are also expected to be stable to reagents used for deprotection of peptides synthesized using FMOC chemistry.![]() In contrast, the BODIPY fluorophore may be unstable to the conditions used to remove some protecting groups.
In contrast, the BODIPY fluorophore may be unstable to the conditions used to remove some protecting groups.
We prepare unique reagents for use in the automated synthesis of peptides that are specifically labeled with fluorophores, chromophores and haptens. These precursors permit the incorporation of a specific label at a specific site in the peptide's sequence. The α-FMOC derivative of ε-dabcyl-L-lysine (D6216) can be used to incorporate the dabcyl chromophore at selected sites in the peptide sequence. The dabcyl chromophore, which has broad visible absorption (Figure 9.5.2), has been extensively used as a quenching group in the automated synthesis of HIV protease and renin (H2930, R2931; Detecting Peptidases and Proteases—Section 10.4) and other fluorogenic peptidase substrates.![]() The dabcyl group can also be incorporated at the N-terminus by using dabcyl succinimidyl ester
The dabcyl group can also be incorporated at the N-terminus by using dabcyl succinimidyl ester ![]() (D2245). The aminonaphthalene derivative EDANS (A91) has been the most common fluorophore for pairing with the dabcyl quencher in fluorescence resonance energy transfer (FRET) experiments because its fluorescence emission spectrum overlaps the absorption spectrum of dabcyl (Figure 9.5.2) (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2). This fluorophore is conveniently introduced during automated synthesis of peptides by using γ-EDANS-α-FMOC-L-glutamic acid (F11831). The tetramethylrhodamine fluorophore can be incorporated during automated FMOC synthesis of peptides using our single-isomer α-(FMOC)-ε-TMR-L-lysine building block (F11830).
(D2245). The aminonaphthalene derivative EDANS (A91) has been the most common fluorophore for pairing with the dabcyl quencher in fluorescence resonance energy transfer (FRET) experiments because its fluorescence emission spectrum overlaps the absorption spectrum of dabcyl (Figure 9.5.2) (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2). This fluorophore is conveniently introduced during automated synthesis of peptides by using γ-EDANS-α-FMOC-L-glutamic acid (F11831). The tetramethylrhodamine fluorophore can be incorporated during automated FMOC synthesis of peptides using our single-isomer α-(FMOC)-ε-TMR-L-lysine building block (F11830).
QSY dyes (Long-Wavelength Rhodamines, Texas Red Dyes and QSY Quenchers—Section 1.6, Reagents for Analysis of Low Molecular Weight Amines—Section 1.8) have broad visible to near-infrared absorption (Figure 9.5.3). These dyes, which are essentially nonfluorescent, are particularly useful as energy acceptors from blue-, green-, orange- or red-fluorescent donor dyes (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2). The QSY 7, QSY 9, QSY 21 and QSY 35 chromophores can be conjugated to amines via their succinimidyl esters (Q10193, Q20131, Q20132, Q20133). The QSY 7 and QSY 35 chromophores can also be conjugated to peptide thiols or thiol-modified oligonucleotides via its maleimide ![]() (Q10257) or iodoacetamide (Q20348); additionally, peptide amides can be prepared from QSY 7 amine (Q10464). We have also prepared α-(FMOC)-ε-QSY 7-L-lysine and α-FMOC-β-QSY 35-L-alanine (Q21930, Q21931), which can be used in the automated synthesis of QSY 7 quencher– or QSY 35 quencher–containing peptides.
(Q10257) or iodoacetamide (Q20348); additionally, peptide amides can be prepared from QSY 7 amine (Q10464). We have also prepared α-(FMOC)-ε-QSY 7-L-lysine and α-FMOC-β-QSY 35-L-alanine (Q21930, Q21931), which can be used in the automated synthesis of QSY 7 quencher– or QSY 35 quencher–containing peptides.
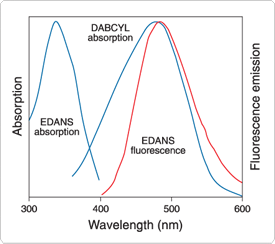
Figure 9.5.2 Spectral overlap between EDANS fluorescence and dabcyl absorption, which is required for efficient quenching of EDANS fluorescence by resonance energy transfer to the nonfluorescent dabcyl chromophore. Spectra are normalized to the same intensities.
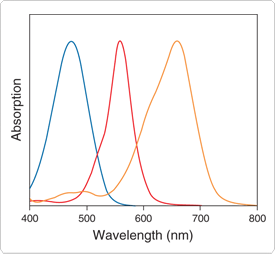
Figure 9.5.3 Normalized absorption spectra of the QSY 35 (blue), QSY 7 (red) and QSY 21 (orange) dyes. The QSY 7 and QSY 9 dyes have essentially identical spectra.
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Notes |
|---|---|---|---|---|---|---|---|---|
| A91 | 288.30 | L | pH >10, DMF | 335 | 5900 | 493 | pH 8 | |
| D21 | 269.75 | F,DD,L | DMF, MeCN | 372 | 3900 | none | CHCl3 | 1, 2 |
| D100 | 265.33 | L | EtOH | 336 | 4400 | 534 | MeOH | |
| D2245 | 366.38 | F,D,L | DMF, DMSO | 453 | 32,000 | none | MeOH | 3 |
| D6216 | 619.72 | F,D,L | DMF, MeCN | 427 | 30,000 | none | MeOH | |
| F11830 | 780.88 | F,D,L | DMF, MeCN | 543 | 92,000 | 570 | MeOH | |
| F11831 | 617.67 | F,D,L | DMF, MeCN | 341 | 5200 | 471 | MeOH | |
| Q10193 | 791.32 | F,D,L | DMSO | 560 | 90,000 | none | MeOH | |
| Q10257 | 858.45 | F,D,L | DMSO | 560 | 92,000 | none | MeOH | |
| Q10464 | 814.87 | L | DMSO | 560 | 92,000 | none | MeOH | |
| Q20131 | 951.43 | F,D,L | H2O, DMSO | 562 | 88,000 | none | MeOH | 4 |
| Q20132 | 815.34 | F,D,L | DMSO | 661 | 90,000 | none | MeOH | |
| Q20133 | 411.33 | F,D,L | DMSO | 475 | 23,000 | none | MeOH | |
| Q20348 | 453.20 | F,D,L | DMSO | 475 | 24,000 | none | MeOH | 5 |
| Q21930 | 1044.66 | F,D,L | DMF, MeCN | 560 | 90,000 | none | MeOH | |
| Q21931 | 565.54 | F,D,L | DMF, MeCN | 475 | 23,000 | none | MeOH | |
| ||||||||
For Research Use Only. Not for use in diagnostic procedures.