Search
Fluorescence Resonance Energy Transfer (FRET)—Note 1.2
Fluorescence resonance energy transfer (FRET) is a distance-dependent interaction between the electronic excited states of two dye molecules in which excitation is transferred from a donor molecule to an acceptor molecule without emission of a photon. The efficiency of FRET is dependent on the inverse sixth power of the intermolecular separation,![]() making it useful over distances comparable to the dimensions of biological macromolecules. Thus, FRET is an important technique for investigating a variety of biological phenomena that produce changes in molecular proximity.
making it useful over distances comparable to the dimensions of biological macromolecules. Thus, FRET is an important technique for investigating a variety of biological phenomena that produce changes in molecular proximity.![]() When FRET is used as a contrast mechanism, colocalization of proteins and other molecules can be imaged with spatial resolution beyond the limits of conventional optical microscopy.
When FRET is used as a contrast mechanism, colocalization of proteins and other molecules can be imaged with spatial resolution beyond the limits of conventional optical microscopy.![]()
Primary Conditions for FRET
- Donor and acceptor molecules must be in close proximity (typically 10–100 Å).
- The absorption spectrum of the acceptor must overlap the fluorescence emission spectrum of the donor (Figure 1).
- Donor and acceptor transition dipole orientations must be approximately parallel.

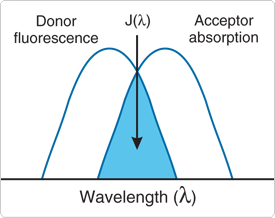
Figure 1. Schematic representation of the FRET spectral overlap integral.
Förster Radius
The distance at which energy transfer is 50% efficient (i.e., 50% of excited donors are deactivated by FRET) is defined by the Förster radius (R0). The magnitude of R0 is dependent on the spectral properties of the donor and acceptor dyes (Table 1):
Table 1. Typical Values of R0
| Donor | Acceptor | R0 (Å) |
|---|---|---|
| Fluorescein | Tetramethylrhodamine | 55 |
| IAEDANS | Fluorescein | 46 |
| EDANS | Dabcyl | 33 |
| Fluorescein | Fluorescein | 44 |
| BODIPY FL | BODIPY FL | 57 |
| Fluorescein | QSY 7 and QSY 9 dyes | 61 |
Donor/Acceptor Pairs
In most applications, the donor and acceptor dyes are different, in which case FRET can be detected by the appearance of sensitized fluorescence of the acceptor or by quenching of donor fluorescence. When the donor and acceptor are the same, FRET can be detected by the resulting fluorescence depolarization.![]() Typical values of R0 for some dye pairs are listed in Table 1 and more extensive compilations are in R<0> values for some Alexa Fluor dyes—Table 1.6 and R<0> values for QSY and dabcyl quenchers—Table 1.11. Note that because the component factors of R0 (see above) are dependent on the environment, the actual value observed in a specific experimental situation is somewhat variable. Extensive compilations of R0 values can be found in the literature.
Typical values of R0 for some dye pairs are listed in Table 1 and more extensive compilations are in R<0> values for some Alexa Fluor dyes—Table 1.6 and R<0> values for QSY and dabcyl quenchers—Table 1.11. Note that because the component factors of R0 (see above) are dependent on the environment, the actual value observed in a specific experimental situation is somewhat variable. Extensive compilations of R0 values can be found in the literature.![]() Nonfluorescent acceptors such as dabcyl and QSY dyes (Molecular Probes nonfluorescent quenchers and photosensitizers—Table 1.10) have the particular advantage of eliminating the potential problem of background fluorescence resulting from direct (i.e., nonsensitized) acceptor excitation.
Nonfluorescent acceptors such as dabcyl and QSY dyes (Molecular Probes nonfluorescent quenchers and photosensitizers—Table 1.10) have the particular advantage of eliminating the potential problem of background fluorescence resulting from direct (i.e., nonsensitized) acceptor excitation.![]() FRET efficiencies from several donor dyes to the QSY 7 quencher in molecular beacon hybridization probes have been calculated.
FRET efficiencies from several donor dyes to the QSY 7 quencher in molecular beacon hybridization probes have been calculated.![]() Probes incorporating fluorescent donor–nonfluorescent acceptor combinations have been developed primarily for detecting proteolysis
Probes incorporating fluorescent donor–nonfluorescent acceptor combinations have been developed primarily for detecting proteolysis ![]() (Figure 2) and nucleic acid hybridization.
(Figure 2) and nucleic acid hybridization.
 | Figure 2. Principle of the fluorogenic response to protease cleavage exhibited by HIV protease substrate 1 (H2930). Quenching of the EDANS fluorophore (F) by distance-dependent resonance energy transfer to the dabcyl quencher (Q) is eliminated upon cleavage of the intervening peptide linker. |
Selected Applications of FRET
- Structure and conformation of proteins

- Spatial distribution and assembly of protein complexes

- Receptor/ligand interactions

- Immunoassays

- Probing interactions of single molecules

- Structure and conformation of nucleic acids

- Real-time PCR assays and SNP detection

- Detection of nucleic acid hybridization

- Primer-extension assays for detecting mutations

- Automated DNA sequencing

- Distribution and transport of lipids

- Membrane fusion assays
 (Lipid-Mixing Assays of Membrane Fusion—Note 13.1)
(Lipid-Mixing Assays of Membrane Fusion—Note 13.1) - Membrane potential sensing

- Fluorogenic protease substrates

- Indicators for cyclic AMP
 and calcium
and calcium 
For Research Use Only. Not for use in diagnostic procedures.