Search
Fluorescence Correlation Spectroscopy (FCS)—Note 1.3
Fluorescence correlation spectroscopy (FCS) is a technique in which spontaneous fluorescence intensity fluctuations are measured in a microscopic detection volume of about 10-15 L (1 femtoliter) defined by a tightly focused laser beam.![]() Renewed interest in FCS in recent years has been stimulated by the fact that it is inherently miniaturized and therefore applicable for high-throughput screening applications.
Renewed interest in FCS in recent years has been stimulated by the fact that it is inherently miniaturized and therefore applicable for high-throughput screening applications.![]() Fluorescence intensity fluctuations measured by FCS represent changes in either the number or the fluorescence quantum yield of molecules resident in the detection volume (Figure 1). Small, rapidly diffusing molecules produce rapidly fluctuating intensity patterns, whereas larger molecules produce more sustained bursts of fluorescence.
Fluorescence intensity fluctuations measured by FCS represent changes in either the number or the fluorescence quantum yield of molecules resident in the detection volume (Figure 1). Small, rapidly diffusing molecules produce rapidly fluctuating intensity patterns, whereas larger molecules produce more sustained bursts of fluorescence.
This situation is in marked contrast to conventional fluorescence photometry carried out in sample volumes of around 0.1–1.0 mL (~108 times larger than FCS measurement volumes) that report only the macroscopic average of diffusion-dependent intensity fluctuations. In a typical FCS measurement, fluorescence intensity is recorded for a small number of molecules in the detection volume (e.g., 3 molecules/femtoliter, equivalent to ~5 nM macroscopic concentration) over a time range from about 1 microsecond to 1 second. The time-dependent fluorescence intensity (F(t)) is then analyzed in terms of its temporal autocorrelation function (G (τ)), which compares the fluorescence intensity at time t with the intensity at (t + τ), where τ is a variable interval, averaged over all data points in the time series:
The autocorrelation function contains information about equilibrium concentrations, reaction kinetics and diffusion rates of molecules in the sample. The initial amplitude of the autocorrelation function is inversely proportional to the number of molecules in the detection volume. The autocorrelation function decays from its initial value with a time-dependence that is determined by molecular diffusion rates. For example, free fluorescent ligands exhibit faster autocorrelation decay than slower-moving complexed ligands (Figure 2).
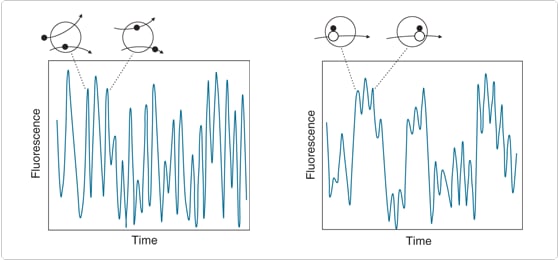
Figure 1. Physical origins of fluorescence correlation spectroscopy data. Free fluorescent ligands move in and out of the detection volume (open circle) and are detected as a series of short, randomized fluorescence bursts (left panel). Macromolecule-bound ligands are less mobile, producing a more slowly fluctuating (i.e., more highly autocorrelated) time-dependent fluorescence pattern (right panel).
 | Figure 2. Simulated FCS autocorrelation functions representing a low molecular weight ligand (left curve, blue), macromolecule-bound ligand (right curve, red) and a 1:1 mixture of free and bound ligand (middle curve, green). |
Probes and Applications for FCS
FCS is applicable for monitoring a multitude of biomolecular association and dissociation processes (Table 1). Because FCS is intrinsically sensitive to the mass changes occurring in these processes, probe design and selection is generally less critical than it is in assays based on macroscopic fluorescence intensity changes generated by dye–dye interactions (FRET, self-quenching etc.) or environment-dependent fluorescence enhancement. Dyes that perform well in confocal laser-scanning microscopy are usually among the best choices for FCS applications. Laser sources used for excitation in FCS include the 488 nm argon-ion spectral line and the 543 nm and 633 nm He-Ne laser spectral lines. Dyes with appreciable rates of triplet state population via intersystem crossing are generally not well suited for FCS measurement because this process results in an additional submillisecond autocorrelation decay component.![]()
Technical Developments in FCS
Two-photon excitation (TPE) has been applied to FCS for reasons similar to those that have motivated its use in fluorescence microscopy—inherent spatial confinement of excitation, diminished photobleaching and phototoxicity, less scattering and better optical penetration in turbid media.![]() Dual-color cross-correlation FCS
Dual-color cross-correlation FCS ![]() measures the cross-correlation of the time-dependent fluorescence intensities of two spectrally distinct dyes, instead of the conventional autocorrelation for a single dye. This approach has the advantage that cross-correlated fluorescence is only generated by molecules or complexes labeled with both dyes, allowing quantitation of interacting molecules without reference to their diffusion characteristics. In practice, discrimination based on mass in conventional FCS requires that the interacting components should have a molecular weight ratio of at least 1:7. FCS measurements using TPE in combination with dual-color cross-correlation have been reported.
measures the cross-correlation of the time-dependent fluorescence intensities of two spectrally distinct dyes, instead of the conventional autocorrelation for a single dye. This approach has the advantage that cross-correlated fluorescence is only generated by molecules or complexes labeled with both dyes, allowing quantitation of interacting molecules without reference to their diffusion characteristics. In practice, discrimination based on mass in conventional FCS requires that the interacting components should have a molecular weight ratio of at least 1:7. FCS measurements using TPE in combination with dual-color cross-correlation have been reported.![]()
Table 1. Applications of fluorescence correlation spectroscopy
For Research Use Only. Not for use in diagnostic procedures.
