Search
Page Contents
Related Tables
Get Chapter Downloads from The Molecular Probes Handbook, 11th edition |
Carboxylic acids can be converted to esters, amides, acyl hydrazides or hydroxamic acids, all of which are discussed in this section. Alternatively, the half-protected tert-butyloxycarbonyl (t-BOC) propylenediamine derivative is useful for converting organic solvent–soluble carboxylic acids into aliphatic amines. Following coupling of the half-protected aliphatic diamine to an activated carboxylic acid, the t-BOC group can be quantitatively removed with trifluoroacetic acid (Figure 3.4.1). The resultant aliphatic amine can then be modified with any of the amine-reactive reagents described in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1 or coupled to solid-phase matrices for affinity chromatography.
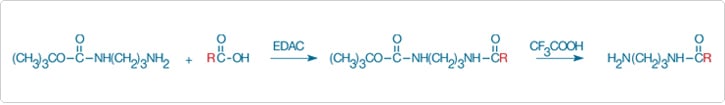
Figure 3.4.1 Conversion of a carboxylic acid group into an aliphatic amine. The activated carboxylic acid is derivatized with a half-protected aliphatic diamine (mono-N-(t-BOC)-propylenediamine), usually in an organic solvent, followed by removal of the t-BOC–protecting group with trifluoroacetic acid.
Modification in Aqueous Solutions
The carboxylic acids of water-soluble biopolymers such as proteins can be coupled to hydrazines, hydroxylamines and amines (Molecular Probes hydrazine, hydroxylamine and amine derivatives—Table 3.2) in aqueous solution using water-soluble carbodiimides such as 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDAC, E2247). Including N-hydroxysulfosuccinimide in the reaction mixture has been shown to improve the coupling efficiency of EDAC-mediated protein–carboxylic acid conjugations ![]() (Figure 3.4.2). To reduce intra- and inter-protein coupling to lysine residues,
(Figure 3.4.2). To reduce intra- and inter-protein coupling to lysine residues,![]() which is a common side reaction, carbodiimide-mediated coupling should be performed in a concentrated protein solution at a low pH, using a large excess of the nucleophile. EDAC-mediated coupling has been used to functionalize Qdot 605 ITK carboxyl quantum dots (Q21301MP, Qdot Nanocrystals—Section 6.6) with the GABA receptor agonist muscimol.
which is a common side reaction, carbodiimide-mediated coupling should be performed in a concentrated protein solution at a low pH, using a large excess of the nucleophile. EDAC-mediated coupling has been used to functionalize Qdot 605 ITK carboxyl quantum dots (Q21301MP, Qdot Nanocrystals—Section 6.6) with the GABA receptor agonist muscimol.![]() EDAC has been shown to be impermeable to membranes of live cells, which permits its use to distinguish between cytoplasmic and lumenal sites of reaction.
EDAC has been shown to be impermeable to membranes of live cells, which permits its use to distinguish between cytoplasmic and lumenal sites of reaction.![]()
Fluoresceinyl glycine amide (5-(aminoacetamido)fluorescein, A1363) and various hydrazines and hydroxylamines may be the best probes for this application because they are more likely to remain reactive at a lower pH than are aliphatic amines such as the cadaverines.![]() Fluoresceinyl glycine amide has been coupled to the carboxylic acid of a cyclosporin derivative by EDAC.
Fluoresceinyl glycine amide has been coupled to the carboxylic acid of a cyclosporin derivative by EDAC.![]()
ANTS (8-aminonaphthalene-1,3,6-trisulfonic acid, A350; Reagents for Modifying Aldehydes and Ketones—Section 3.3) has a high ionic charge, which permits electrophoretic separation of its products with complex oligosaccharides.![]() Several of the fluorescent hydrazine and hydroxylamine derivatives described in Reagents for Modifying Aldehydes and Ketones—Section 3.3 should have similar utility for carbodiimide-mediated derivatization of carboxylic acids.
Several of the fluorescent hydrazine and hydroxylamine derivatives described in Reagents for Modifying Aldehydes and Ketones—Section 3.3 should have similar utility for carbodiimide-mediated derivatization of carboxylic acids.
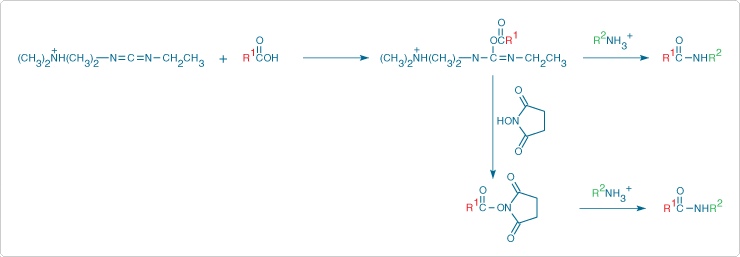
Figure 3.4.2 Stabilization of an unstable O-acylisourea intermediate by N-hydroxysuccinimide in a carbodiimide-mediated (EDAC) modification of a carboxylic acid with a primary amine.
Modification in Organic Solvents
Peptide synthesis research has led to the development of numerous methods for coupling carboxylic acids to amines in organic solution. One such method involves the conversion of carboxylic acids to succinimidyl esters or mixed anhydrides. Dicyclohexylcarbodiimide and diisopropylcarbodiimide are widely used to promote amide formation in organic solution. Another recommended derivatization method for coupling organic solvent–soluble carboxylic acids, including peptides, to aliphatic amines without racemization is the combination of 2,2'-dipyridyldisulfide and triphenylphosphine.![]() Unlike fluorescent aliphatic amines, fluorescent aromatic amines such as those derived from 7-amino-4-methylcoumarin (A191) and 2-aminoacridone (A6289, Reagents for Modifying Aldehydes and Ketones—Section 3.3) exhibit a shift in their absorption and emission (if any) to much shorter wavelengths upon forming carboxamides. This property makes these aromatic amines preferred reagents for preparing peptidase substrates (Detecting Peptidases and Proteases—Section 10.4). Aromatic amines can generally be coupled to acid halides and anhydrides, with organic solvents usually required for efficient reaction.
Unlike fluorescent aliphatic amines, fluorescent aromatic amines such as those derived from 7-amino-4-methylcoumarin (A191) and 2-aminoacridone (A6289, Reagents for Modifying Aldehydes and Ketones—Section 3.3) exhibit a shift in their absorption and emission (if any) to much shorter wavelengths upon forming carboxamides. This property makes these aromatic amines preferred reagents for preparing peptidase substrates (Detecting Peptidases and Proteases—Section 10.4). Aromatic amines can generally be coupled to acid halides and anhydrides, with organic solvents usually required for efficient reaction.
Hydrazine, Hydroxylamine and Aliphatic Amine Derivatives
We provide a wide selection of carboxylic acid–reactive reagents (Molecular Probes hydrazine, hydroxylamine and amine derivatives—Table 3.2), including several different Dapoxyl, Alexa Fluor, BODIPY, fluorescein, Oregon Green, rhodamine, Texas Red and QSY Hydrazine Derivatives, Hydroxylamine Derivatives and Amine Derivatives, all of which are particularly useful for synthesizing drug analogs and as probes for fluorescence polarization immunoassays ![]() (Fluorescence Polarization (FP)—Note 1.4). These probes all require a coupling agent such as a carbodiimide to react with carboxylic acids; they do not spontaneously react with carboxylic acids in solution. They do, however, react spontaneously with the common amine-reactive functional groups described in Introduction to Amine Modification—Section 1.1, including succinimidyl esters and isothiocyanates. Some of the more important probes and their applications include:
(Fluorescence Polarization (FP)—Note 1.4). These probes all require a coupling agent such as a carbodiimide to react with carboxylic acids; they do not spontaneously react with carboxylic acids in solution. They do, however, react spontaneously with the common amine-reactive functional groups described in Introduction to Amine Modification—Section 1.1, including succinimidyl esters and isothiocyanates. Some of the more important probes and their applications include:
- Alexa Fluor hydrazides (A10436, A10437, A10438, A10439, A30634, A20501MP, A20502; Reagents for Modifying Aldehydes and Ketones—Section 3.3), Alexa Fluor hydroxylamines (A30629, A30632; Reagents for Modifying Aldehydes and Ketones—Section 3.3) and Alexa Fluor cadaverines (A30675, A30676, A30677, A30678, A30679), our brightest and most photostable carboxylic acid–reactive probes
- BODIPY aliphatic amines (D2390, D6251), for preparing pH-insensitive probes, such as BODIPY FL etoposide,
 from carboxylic acid derivatives
from carboxylic acid derivatives - 5-(aminomethy)lfluorescein (A1353), which is readily coupled to activated carboxylic acids

- Dapoxyl (2-aminoethyl)sulfonamide for preparing conjugates with strong UV absorption and a Stokes shift of ~200 nm
- Dansyl ethylenediamine (D112) and Lissamine rhodamine B ethylenediamine (L2424), for carboxylic acid derivatization
 and glutamine transamidation reactions
and glutamine transamidation reactions  (Figure 3.4.3)
(Figure 3.4.3) - Bimane amine, a small blue-fluorescent dye for carboxylic acid derivatization
- EDANS for preparing radioactive IAEDANS,
 energy transfer–quenched substrates for endopeptidases
energy transfer–quenched substrates for endopeptidases  (Detecting Peptidases and Proteases—Section 10.4) and an ATP substrate analog for DNA-dependent RNA polymerase
(Detecting Peptidases and Proteases—Section 10.4) and an ATP substrate analog for DNA-dependent RNA polymerase 
- QSY 7 amine, which is an essentially nonfluorescent dye with strong visible absorption for preparing highly efficient quenchers for bioassays based on fluorescence resonance energy transfer (FRET) (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2)
- Hydrazine (Reagents for Modifying Aldehydes and Ketones—Section 3.3) and amine derivatives of Alexa Fluor dyes, which are precursors of highly fluorescent, water-soluble probes
- Hydrazine and amine derivatives of biotin and desthiobiotin (Biotinylation and Haptenylation Reagents—Section 4.2), which are versatile intermediates for synthesizing biotin- and desthiobiotin-containing probes

Enzyme-Catalyzed Transamidation
A transglutaminase-catalyzed transamidation reaction of glutamine residues in some proteins and peptides enables their selective modification by amine-containing probes ![]() (Figure 3.4.3). This unique method for selective protein modification requires formation of a complex consisting of the glutamine residue, the aliphatic amine probe and the enzyme. It has been found that a short aliphatic spacer in the amine probe enhances the reaction. The cadaverine (–NH(CH2)5NH–) spacer is usually optimal. Although dansyl cadaverine has been probably the most widely used reagent,
(Figure 3.4.3). This unique method for selective protein modification requires formation of a complex consisting of the glutamine residue, the aliphatic amine probe and the enzyme. It has been found that a short aliphatic spacer in the amine probe enhances the reaction. The cadaverine (–NH(CH2)5NH–) spacer is usually optimal. Although dansyl cadaverine has been probably the most widely used reagent,![]() Alexa Fluor cadaverines
Alexa Fluor cadaverines ![]() (A30675, A30676, A30677, A30678, A30679), Oregon Green 488 cadaverine (O10465), fluorescein cadaverine
(A30675, A30676, A30677, A30678, A30679), Oregon Green 488 cadaverine (O10465), fluorescein cadaverine ![]() (A10466), tetramethylrhodamine cadaverine
(A10466), tetramethylrhodamine cadaverine ![]() (A1318), Texas Red cadaverine (T2425) and BODIPY TR cadaverine (D6251) are among the most fluorescent transglutaminase substrates available. The intrinsic transglutaminase activity in sea urchin eggs has been used to covalently incorporate dansyl cadaverine during embryonic development.
(A1318), Texas Red cadaverine (T2425) and BODIPY TR cadaverine (D6251) are among the most fluorescent transglutaminase substrates available. The intrinsic transglutaminase activity in sea urchin eggs has been used to covalently incorporate dansyl cadaverine during embryonic development.![]() Two biotin cadaverines (A1594, B1596; Biotinylation and Haptenylation Reagents—Section 4.2) are also available for transglutaminase-mediated reactions.
Two biotin cadaverines (A1594, B1596; Biotinylation and Haptenylation Reagents—Section 4.2) are also available for transglutaminase-mediated reactions.![]() Amine-terminated peptides and fluorescent and biotin hydrazides, including Cascade Blue hydrazide, have been successfully incorporated into protein fragments by transamidation during enzyme-catalyzed proteolysis.
Amine-terminated peptides and fluorescent and biotin hydrazides, including Cascade Blue hydrazide, have been successfully incorporated into protein fragments by transamidation during enzyme-catalyzed proteolysis.![]()
Transamidation of cell-surface glutamine residues by the combination of a transglutaminase enzyme and a fluorescent or biotinylated aliphatic amine can form stable amides.![]() Impermeability of the enzyme restricts this reaction to a limited number of proteins on the cell surface. This technique was used to selectively label erythrocyte band 3 protein with dansyl cadaverine and proteins of the extracellular matrix with fluorescein cadaverine
Impermeability of the enzyme restricts this reaction to a limited number of proteins on the cell surface. This technique was used to selectively label erythrocyte band 3 protein with dansyl cadaverine and proteins of the extracellular matrix with fluorescein cadaverine ![]() (A10466). Following protease treatment, the dansylated peptides were isolated using an anti-dansyl affinity column.
(A10466). Following protease treatment, the dansylated peptides were isolated using an anti-dansyl affinity column.![]()

Figure 3.4.3 Transglutaminase-mediated labeling of a protein using dansyl cadaverine.
Biologically important molecules, especially the nonchromophoric fatty acids, bile acids and prostaglandins, are typically esterified by carboxylic acid–reactive reagents in organic solvents. Esterification of carboxylic acids in aqueous solution is usually not possible, and esters tend to be unstable in water. Fluorescent derivatization reagents for biomedical chromatography have been extensively discussed in reviews.![]()
HPLC derivatization reagents for carboxylic acids include two fluorescent analogs of the common esterification reagent diazomethane. Diazoalkanes react without the addition of catalysts and may be useful for direct carboxylic acid modification of proteins and synthetic polymers. Fluorescent diazoalkanes also react with phosphates ![]() and potentially with lipid-associated carboxylic acids in membrane-bound proteins or with free fatty acids.
and potentially with lipid-associated carboxylic acids in membrane-bound proteins or with free fatty acids.
The fluorescent diazomethyl derivative 9-anthryldiazomethane (ADAM) has been commonly used to derivatize biomolecules. Unfortunately, ADAM is not very stable and may decompose during storage. 1-Pyrenyldiazomethane ![]() (PDAM, P1405) is recommended as a replacement for ADAM because it has much better chemical stability. Moreover, the detection limit for PDAM conjugates is reported to be about 20–30 femtomoles, which is five times better than reported for detection of ADAM conjugates.
(PDAM, P1405) is recommended as a replacement for ADAM because it has much better chemical stability. Moreover, the detection limit for PDAM conjugates is reported to be about 20–30 femtomoles, which is five times better than reported for detection of ADAM conjugates.![]() In addition, fatty acids derivatized with these reagents have been used to measure phospholipase A2 activity
In addition, fatty acids derivatized with these reagents have been used to measure phospholipase A2 activity ![]() (Probes for Lipid Metabolism and Signaling—Section 17.4). It has been reported that photolysis of pyrenemethyl esters liberates the free carboxylic acid,
(Probes for Lipid Metabolism and Signaling—Section 17.4). It has been reported that photolysis of pyrenemethyl esters liberates the free carboxylic acid,![]() making PDAM a potential protecting group for carboxylic acids.
making PDAM a potential protecting group for carboxylic acids.
The low nucleophilicity of carboxylic acids requires that they be converted to anions (typically cesium or quaternary ammonium are used as counterions) before they can be esterified with alkyl halides in organic solvents. Conjugates of 6-bromoacetyl-2-dimethylaminonaphthalene (badan, B6057) have a high Stokes shift, as well as spectral properties that are very sensitive to their environment. 5-(Bromomethyl)fluorescein and BODIPY 493/503 methyl bromide have the strongest absorptivity and fluorescence of the currently available carboxylic acid–derivatization reagents.![]() An analytical method for estimating the degree of EDAC crosslinking of collagen has been developed based on derivatization of residual carboxyl groups by 5-bromomethylfluorescein.
An analytical method for estimating the degree of EDAC crosslinking of collagen has been developed based on derivatization of residual carboxyl groups by 5-bromomethylfluorescein.![]()
All of the alkyl halides in this section also react with thiol groups, including those in proteins.![]() Although more commonly used as thiol-reactive reagents, the monobromobimanes (M1378, M20381; Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3) have been reported to react with carboxylic acids in organic solvents.
Although more commonly used as thiol-reactive reagents, the monobromobimanes (M1378, M20381; Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3) have been reported to react with carboxylic acids in organic solvents.![]() The coumarin iodoacetamide DCIA (Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3) has also been used to derivatize carboxylic acids;
The coumarin iodoacetamide DCIA (Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3) has also been used to derivatize carboxylic acids;![]() other iodoacetamides described in Thiol-Reactive Probes—Chapter 2 will probably react similarly.
other iodoacetamides described in Thiol-Reactive Probes—Chapter 2 will probably react similarly.
2-(2,3-Naphthalimino)ethyl trifluoromethanesulfonate reacts rapidly with the anions of carboxylic acids in acetonitrile to give adducts that are reported to be detectable by absorption at 259 nm down to 100 femtomoles and by fluorescence at 394 nm down to 4 femtomoles.![]() This naphthalimide sulfonate ester will likely react with other nucleophiles too, including thiols, amines, phenols (e.g., tyrosine) and probably histidine. 2-(2,3-Naphthalimino)ethyl trifluoromethanesulfonate has been used for the sensitive reverse-phase HPLC detection of eicosanoids in brain tissue.
This naphthalimide sulfonate ester will likely react with other nucleophiles too, including thiols, amines, phenols (e.g., tyrosine) and probably histidine. 2-(2,3-Naphthalimino)ethyl trifluoromethanesulfonate has been used for the sensitive reverse-phase HPLC detection of eicosanoids in brain tissue.![]()
4-Sulfo-2,3,5,6-tetrafluorophenol (STP) and N-hydroxysulfosuccinimide (NHSS) can be used to prepare water-soluble activated esters from various carboxylic acids (Figure 3.4.4). Coupling typically involves a carbodiimide such as EDAC (E2247) and is performed in an organic solvent. We have found that the resulting STP esters are much easier to purify and more stable than activated esters prepared from N-hydroxysulfosuccinimide.![]() NHSS esters of biotin and other derivatives considerably increase the aqueous solubility of the reagents.
NHSS esters of biotin and other derivatives considerably increase the aqueous solubility of the reagents.![]() We offer a variety of amine-reactive STP esters, which are discussed in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1.
We offer a variety of amine-reactive STP esters, which are discussed in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1.
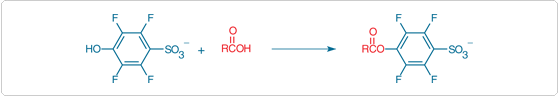
Figure 3.4.4 4-Sulfo-2,3,5,6-tetrafluorophenol (STP) can be used to prepare water-soluble activated esters from various carboxylic acids.
For a detailed explanation of column headings, see Definitions of Data Table Contents
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Notes |
|---|---|---|---|---|---|---|---|---|
| EDANS | 288.30 | L | pH >10, DMF | 335 | 5900 | 493 | pH 8 | |
| A191 7-amino-4-methylcoumarin | 175.19 | L | DMF, DMSO | 351 | 18,000 | 430 | MeOH | 1 |
| A1318 tetramethylrhodamine cadaverine | 514.62 | F,D,L | DMF, EtOH | 544 | 78,000 | 571 | MeOH | |
| lucifer yellow ethylenediamine | 491.57 | L | H2O | 425 | 12,000 | 532 | H2O | |
| lucifer yellow cadaverine | 533.65 | L | H2O | 426 | 11,000 | 531 | H2O | |
| 4'-(aminoethyl)fluorescein | 397.81 | L | pH >6, DMF | 492 | 80,000 | 516 | pH 9 | 2 |
| A1353 5-(aminomethyl)fluorescein | 397.81 | L | pH >6, DMF | 492 | 68,000 | 516 | pH 9 | 2 |
| A1363 fluoresceinyl glycine amide | 404.38 | L | pH >6, DMF | 491 | 80,000 | 515 | pH 9 | 2 |
| ADAM | 218.26 | FF,D,L | DMF, MeCN | 364 | 6100 | 411 | MeOH | |
| A10466 fluorescein cadaverine | 653.38 | D,L | pH >6, DMF | 493 | 82,000 | 517 | pH 9 | 2 |
| Alexa Fluor 350 cadaverine | 397.45 | F,D,L | H2O | 353 | 20,000 | 437 | MeOH | |
| A30675 Alexa Fluor 405 cadaverine | 666.58 | F,D,L | H2O | 399 | 29,000 | 422 | H2O | 3 |
| A30676 Alexa Fluor 488 cadaverine | 640.61 | F,D,L | H2O | 493 | 73,000 | 516 | pH 7 | 4, 5 |
| A30677 Alexa Fluor 555 cadaverine | ~950 | F,D,L | H2O | 555 | 155,000 | 572 | MeOH | |
| A30678 Alexa Fluor 594 cadaverine | 806.94 | F,D,L | H2O | 588 | 105,000 | 612 | pH 7 | |
| A30679 Alexa Fluor 647 cadaverine | ~1000 | F,D,L | H2O | 651 | 245,000 | 672 | MeOH | |
| Alexa Fluor 568 cadaverine | 812.95 | F,D,L | H2O | 578 | 93,000 | 602 | pH 7 | |
| 5-(bromomethyl)fluorescein | 425.23 | F,D,L | pH >6, DMF | 492 | 81,000 | 515 | pH 9 | |
| BODiPY 493/503 methyl bromide | 341.00 | F,D,L | DMSO, MeCN | 533 | 62,000 | 561 | CHCl3 | |
| B6057 badan | 292.17 | F,L | DMF, MeCN | 387 | 21,000 | 520 | MeOH | |
| bimane amine | 207.23 | F,D,L | DMSO | 375 | 6000 | 458 | MeOH | |
| Cascade Blue ethylenediamine | 624.49 | L | H2O | 399 | 30,000 | 423 | H2O | 3 |
| D112 dansyl ethylenediamine | 293.38 | L | EtOH, DMF | 335 | 4600 | 526 | MeOH | |
| dansyl cadaverine | 335.46 | L | EtOH, DMF | 335 | 4600 | 518 | MeOH | |
| D2390 BODIPY FL EDA | 370.64 | F,D,L | DMSO, MeCN | 503 | 76,000 | 510 | MeOH | 6 |
| D6251 BODIPY TR cadaverine | 544.85 | F,D,L | DMSO, MeCN | 588 | 64,000 | 616 | MeOH | 6 |
| Dapoxyl (2-aminoethyl)sulfonamide | 386.47 | L | DMF, DMSO | 373 | 23,000 | 571 | MeOH | 7 |
| E2247 EDAC | 191.70 | F,D | H2O | <300 | none | |||
| NHSS | 217.13 | D | H2O | <300 | none | |||
| L2424 Lissamine rhodamine B ethylenediamine | 600.75 | L | DMF, DMSO | 561 | 122,000 | 581 | MeOH | |
| mono-N-(t-BOC)-propylenediamine | 174.24 | D,A | DMF, MeCN | <300 | none | |||
| 2-(2,3-naphthalimino)ethyl triflluoromethanesulfonate | 373.30 | FF,DD,L | DMF, CHCl3 | 260 | 59,000 | 395 | MeOH | |
| O10465 Oregon Green 488 cadaverine | 496.47 | F,D,L | pH >6, DMF | 494 | 75,000 | 521 | pH 9 | 8 |
| P1405 PDAM | 242.28 | FF,L | DMF, MeCN | 340 | 41,000 | 375 | MeOH | |
| QSY 7 amine | 814.87 | L | DMSO | 560 | 92,000 | none | MeOH | |
| STP | 268.11 | D | H2O | <300 | none | |||
| T2425 Texas Red C5 | 690.87 | L | DMF | 591 | 85,000 | 612 | pH 9 | |
| ||||||||
For Research Use Only. Not for use in diagnostic procedures.