Search
Obtaining high quality, intact RNA is the first and often the most critical step in performing gene expression analysis.
Typically, in order to isolate high quality RNA, the tissue has to be processed immediately after harvest. Ambion's patented RNAlater™ Tissue Collection: RNA Stabilization Solution makes it possible for researchers to postpone RNAlater® for days, weeks, or even months after tissue collection without sacrificing RNA integrity (Figure 1). Since RNAlater inactivates all cellular enzymes, including RNases, RNA expression profiles can be "frozen" without immediate RNA isolation.
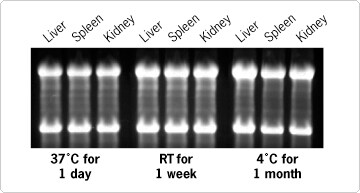
Figure 1. Quality of RNA Isolated From Tissue Stored in RNA later™ Solution. Fresh mouse tissues were dissected and stored in RNA later at 37°C for 1 day, room temperature for 1 week, or 4°C for 1 month. RNA was isolated using TRI Reagent® (MRC) and analyzed using denaturing agarose gel electrophoresis.
How do you use RNAlater?
Dissected tissue (< 0.5 cm in one dimension) is simply submerged in 5-10 volumes of RNA
later at room temperature. RNA
later is compatible with dissected tissue samples, eukaryotic and bacterial cells, and even plasma. The solution permeates the cells, stabilizing the RNA and "freezing" the RNA expression profile. The sample can then be stored in RNA
later at room temperature for up to a week, stored at 4°C for a month, or stored indefinitely at -20°C without nucleic acid degradation. For RNA isolation, the tissue is simply removed from RNAlater and treated as though it had just been harvested. Most tissues can be transferred directly to lysis buffer and homogenized, although some hard tissues such as bone may require a more rigorous method of disruption. RNA can be isolated from RNA
later-treated tissues using any of Ambion's RNA isolation kits.
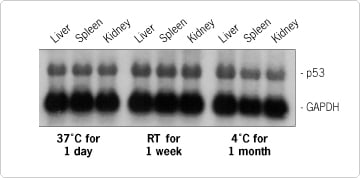
Figure 2. Northern Blot of RNA Isolated From RNA later™-Preserved Tissue. Mouse tissues were dissected and stored in RNA later as indicated. RNA was purified from equal mass amounts of tissue using TRI Reagent® (MRC). Five µg of each RNA sample was Northern blotted. The blot was hybridized with 10 6 cpm/ml of a high specific activity probe for p53 and 10 6 cpm/ml of a low specific activity probe for GAPDH.

Figure 2. Northern Blot of RNA Isolated From RNA later™-Preserved Tissue. Mouse tissues were dissected and stored in RNA later as indicated. RNA was purified from equal mass amounts of tissue using TRI Reagent® (MRC). Five µg of each RNA sample was Northern blotted. The blot was hybridized with 10 6 cpm/ml of a high specific activity probe for p53 and 10 6 cpm/ml of a low specific activity probe for GAPDH.
What can RNAlater do for you?
RNA
later is an aqueous, nontoxic tissue and cell collection reagent that stabilizes and protects cellular RNA in intact, unfrozen tissue and cell samples. RNA
later eliminates the need to immediately process samples or to freeze samples in liquid nitrogen for RNA preservation. Tissue pieces can be harvested and submerged in RNA
later for storage without jeopardizing the quality or quantity of RNA obtained after subsequent RNA isolation (1-4). RNA
later can be added to tissue, cell pellets, cells in media, or bacteria. The samples can then be stored frozen or at 4°C.
Treating tissue with RNA later can also preserve the tissue architecture, which is essential for histopathological analysis. Ellis et al (2002) found that treating breast tissue cores with RNA later prior to fixation allows optimal pathological interpretation and preservation of important diagnostic information.
Treating tissue with RNA later can also preserve the tissue architecture, which is essential for histopathological analysis. Ellis et al (2002) found that treating breast tissue cores with RNA later prior to fixation allows optimal pathological interpretation and preservation of important diagnostic information.
References
- Dunmire V, Wu C, Symmans WF, Zhang W. (2002) Increased yield of total RNA from fine-needle aspirates for use in expression microarray analysis. Biotechniques 33(4): 890-6.
- Ellis M, Davis N, Coop A, Liu M, Schumaker L, Lee RY, Srikanchana R, Russell CG, Singh B, Miller WR, Stearns V, Pennanen M, Tsangaris T, Gallagher A, Liu A, Zwart A, Hayes DF, Lippman ME, Wang Y, Clarke R. (2002) Development and validation of a method for using breast core needle biopsies for gene expression microarray analyses. Clin Cancer Res 8(5): 1155-66.
- Florell SR, Coffin CM, Holden JA, Zimmermann JW, Gerwels JW, Summers BK, Jones DA, Leachman SA. (2001) Preservation of RNA for functional genomic studies: a multidisciplinary tumor bank protocol. Mod Pathol 14(2): 116-28.
- Bachoon DS, Chen F, Hodson RE. (2001) RNA recovery and detection of mRNA by RT-PCR from preserved prokaryotic samples. FEMS Microbiol Lett 201(2): 127-32.
RNAlater™ and Western Analysis
In a recent study, Monique De Paepe et al. (1) compared mRNA and protein stability in human perinatal lung tissue samples treated with RNA
later or snap frozen in liquid nitrogen. They found that RNA
later treated tissue yielded more total RNA and protein than those treated by liquid nitrogen freezing. In addition, the protein integrity of RNA
later treated tissues was equivalent to that of liquid nitrogen frozen tissues, as determined by Western blot analysis of ß-actin protein expression. These authors concluded that, "RNA
later is equivalent if not superior to liquid nitrogen for preservation of postmortem RNA and protein in developing human lungs".
Stabilizing Viral RNA/DNA in Plasma
Nucleic Acid Testing of plasma samples to determine the presence of viral RNA/DNA is widely used with donated blood/plasma samples. This technique may require the transportation of plasma from blood banks to institutes where testing can be carried out. Commonly used techniques to ship plasma involve freezing the sample or making dried plasma spots. The former method relies on shipment of samples on dry ice and the latter is a long procedure requiring access to incubators and laminar flow hoods.
Recently, Dong-Hun Lee and colleagues at the Lindsley F. Kimball Research Institute of the New York Blood Center compared the above two techniques with the use of RNAlater to stabilize plasma samples during transportation (1). Their investigation showed that use of RNA later greatly improved the nucleic acid testing results for RNA viruses such as Hepatitis C Virus (HCV) and Human Immunodeficiency Virus (HIV). These samples were stable for up to 14 days at room temperature or 37ºC. RNA later was not found to be necessary for shipment of samples where only the HBV DNA virus is analyzed.
Recently, Dong-Hun Lee and colleagues at the Lindsley F. Kimball Research Institute of the New York Blood Center compared the above two techniques with the use of RNAlater to stabilize plasma samples during transportation (1). Their investigation showed that use of RNA later greatly improved the nucleic acid testing results for RNA viruses such as Hepatitis C Virus (HCV) and Human Immunodeficiency Virus (HIV). These samples were stable for up to 14 days at room temperature or 37ºC. RNA later was not found to be necessary for shipment of samples where only the HBV DNA virus is analyzed.