Search Thermo Fisher Scientific
Carbonyl-reactive Crosslinker Chemistry
Introduction
Carbonyls (aldehydes) as crosslinking targets
Aldehydes (RCHO) and ketones (RCOR') are reactive varieties of the more general functional group called carbonyls, which have a carbon-oxygen double-bond (C=O). The polarity of this bond (especially in the context of aldehydes) makes the carbon atom electrophilic and reactive to nucleophiles such as primary amines.
Although aldehydes do not naturally occur in proteins or other macromolecules of interest in typical biological samples, they can be created wherever oxidizable sugar groups (also called reducing sugars) exist. Such sugars are common monomer-constituents of the polysaccharides or carbohydrates in post-translational modifications (glycosylation) of many proteins (i.e., glycoproteins). In addition, the ribose of RNA is a reducing sugar.
Periodic acid (HIO4) from dissolved sodium periodates (NaIO4) is a well-known mild agent for effectively oxidizing vicinal diols in carbohydrate sugars to yield reactive aldehyde groups. The carbon-carbon bond is cleaved between adjacent hydroxyl groups. By altering the amount of periodate used, aldehydes can be produced on a smaller or larger selection of sugar types. For example, treatment of glycoproteins with 1 mM periodate usually affects only sialic acid residues, which frequently occur at the ends of polysaccharide chains. At concentrations of 6 to 10 mM periodate, other sugar groups in proteins will be affected.
Carbohydrate modification is particularly useful for creating target sites for conjugation on polyclonal antibodies because the polysaccharides are located in the Fc region, away from the antigen-binding site. This results in labeling or crosslinking sites located away from antigen binding sites, ensuring that antibody function will not be adversely affected by the conjugation procedure.
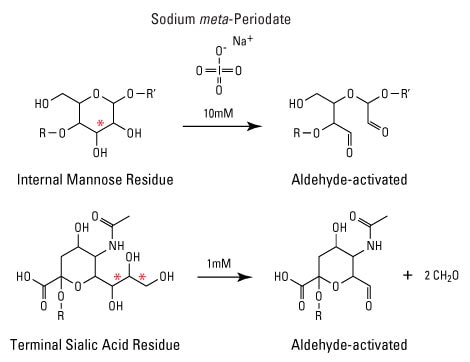
The reaction of sodium periodate with sugar residues yields aldehydes for conjugation reactions. R and R' represent connecting sugar monomers of the polysaccharide. Red asterisks indicate sites of diol cleavage. Sialic acid is also called N-acetyl-D-neuraminic acid.
Carbohydrate modification is particularly useful for creating target sites for conjugation on polyclonal antibodies because the polysaccharides are located in the Fc region. This results in labeling or crosslinking sites located away from antigen binding sites, ensuring that antibody function will not be adversely affected by the conjugation procedure.
Bioconjugation and crosslinking technical handbook
Learn how to optimize your bioconjugation strategies with our updated Bioconjugation and crosslinking technical handbook. This easy-to-use guide overviews our portfolio of reagents for bioconjugation, crosslinking, biotinylation, and modification of proteins and peptides.
Achieve the most efficient modification for your typical applications including:
- Protein and peptide biotinylation
- Antibody labeling with fluorophores and biotin
- Immobilizing biomolecules to surfaces
- Capturing protein interactions
Aldehyde-reactive crosslinker reactive groups
As already mentioned above, nucleophilic varieties of primary amines (–NH2) are the main class of compounds that are reactive with aldehydes. Since primary amines are abundant in proteins, it is important to remember that aldehydes represent an amine-reactive crosslinker chemistry just as much as primary amines constitute an aldehyde-reactive crosslinker chemistry (this page).
However, the natural primary amines of proteins (N-terminus of polypeptides and the side chain of lysines), being in the form R–C–NH2, are not strongly or permanently reactive with aldehydes, except in certain conditions and when additional compounds are added to stabilize the bond. The reaction, called reductive amination, is discussed at the end of this article, because its main applications do not involve the use of discrete reactive groups that can be incorporated into ready-to-use labeling or crosslinking reagents.
Instead, hydrazides and alkoxyamines are the most important aldehyde-reactive functional groups for incorporation into synthetic labeling reagents and crosslinkers. The terminal amino groups in these compounds are more strongly nucleophilic than protein amines, and they spontaneously react with aldehydes to form stable bonds.
Hydrazide reaction chemistry
Aldehydes created by periodate-oxidation of sugars in biological samples react with hydrazides at pH 5 to 7 to form hydrazone bonds. Although this bond to a hydrazide group is a type of Schiff base, it is considerably more stable than a Schiff base formed with a simple amine. The hydrazone bond is sufficiently stable for most protein-labeling applications. If desired, however, the double bond can be reduced to a more stable secondary amine bond using sodium cyanoborohydride (see section on reductive amination at the end of this page).

Hydrazide reaction scheme for chemical conjugation to an aldehyde. R represents a labeling reagent or one end of a crosslinker having the hydrazide reactive group; P represents a glycoprotein or other glycosylated molecule that contains the target functional group (e.g., an aldehyde formed by periodate oxidation of carbohydrate-sugar groups, such as sialic acid).
With low molecular weights (<1,000 Da) and bright fluorescent signals, hyrazides are used as polar tracers that cross gap junctions and follow neuronal projections. Therefore, hydrazides are often used to investigate neuronal communication in conjunction with labeled dextrans, which are retained within the cell.Hydrazides exist in a range of fluorescent colors and can be multiplexed with other probes. The following confocal image provides a representative example of a hydrazide used for neuronal tracing.

Neuron staining. Confocal image stack of a 10,000 MW calcium green dextran–labeled climbing fiber in a sagittal cerebellar slice, showing incoming axon and terminal arborization (in yellow). The Purkinje cell innervated by this fiber was labeled with Invitrogen Alexa Fluor 568 hydrazide.
Bioconjugate Techniques, 3rd Edition (2013) by Greg T. Hermanson is a major update to a book that is widely recognized as the definitive reference guide in the field of bioconjugation.
Bioconjugate Techniques is a complete textbook and protocols-manual for life scientists wishing to learn and master biomolecular crosslinking, labeling, and immobilization techniques that form the basis of many laboratory applications. The book is also an exhaustive and robust reference for researchers looking to develop novel conjugation strategies for entirely new applications. It also contains an extensive introduction to the field of bioconjugation that covers all of the major applications of the technology used in diverse scientific disciplines as well as containing tips for designing the optimal bioconjugate for any purpose.

Aniline catalysis of hydrazide conjugation
Recently, it was discovered that aniline acts as a catalyst for hydrazide-aldehyde reactions. The aromatic amine of aniline rapidly and efficiently forms a Schiff base with the aldehyde, effectively increasing the activation of the aldehyde. As a result, the aniline is easily replaced by the hydrazide. Thus, aniline (also called GlycoLink Coupling Catalyst) allows significantly greater total coupling yields with hydrazides and/or greater efficiency (i.e., equal yields with less hydrazide reagent). GlycoLink Coupling Catalyst decreases reaction times and increases aldehyde-hydrazide coupling efficiency, resulting in greater than 90% coupling of glycoproteins in 4 hours.
See the following references for additional information about aniline catalysis:
- Byeon, J.Y., et al. (2010). Efficient bioconjugation of protein capture agents to biosensor surfaces using aniline-catalyzed hydrazone ligation. Langmuir 26(19):15430-5.
- Dirksen, A., et al. (2006). Nucleophilic catalysis of hydrazone formation and transimination: implications for dynamic covalent chemistry. J. Am. Chem. Soc. 128(49):15602-3.
- Dirksen, A., Dawson, PE. (2008). Rapid oxime and hydrazone ligations with aromatic aldehydes for biomolecular labeling. Bioconjug. Chem. 19(12):2543-8.
To learn more about aniline, see the table below.
| Protein | Molecular Weight | # Glycosylation Sites | % Occupancy of Sites | % Coupled |
|---|---|---|---|---|
| Monoclonal Rat IgG1 | 150,000 | 2 | variable | 72.93 |
| Monoclonal Mouse IgG1 | 150,000 | 2 | variable | 84.74 |
| Rabbit Serum IgG | 150,000 | 2 | 100 | 89.78 |
| Polyclonal Chicken IgY | 170,000 | 4 | 100 | 90.62 |
| Human Serum IgG | 150,000 | 2 | 100 | 97.13 |
| Human Serum IgM | 970,000 | 10 | 100 | 97.18 |
| Ovalbumin | 45,000 | 1 | 100 | 98.50 |
Table 1. Glycosylation properties of polyclonal antibodies (and ovalbumin) and efficiency of glycoprotein coupling to Thermo Scientific GlycoLink Resin. For each experiment, 0.4 mg of protein was reacted with 0.1 mL of resin
Applications for hydrazide crosslinking
1. Using bis-hydrazide compounds
Commercially available homobifunctional hydrazide compounds (i.e., those which have a hydrazide group at each end) include carbohydrazide (MW 90.1) and adipic acid dihydrazide (ADH, MW 174.2). These bis-hydrazide compounds could be used in a single reaction to crosslink and polymerize prepared (aldehyde-containing) sugars or polysaccharides. However, they are more frequently used to modify and conjugate molecules in two stages, either homobifunctionally or heterobifunctionally.
For example, by reacting ADH in huge excess with periodate-oxidized dextran (polysaccharide), only one side of each individual ADH molecule will conjugate, resulting in hydrazide-activation of the dextran. After desalting to remove the excess, unreacted ADH, a small aldehyde-containing ligand could be added to conjugate at multiple locations onto the much larger dextran.
Because each hydrazide is also an amine, a bis-hydrazide compound can be used heterobifunctionally in a variety of ways to perform specialized conjugates. For example, ADH could be reacted in large excess with an RNA oligonucleotide whose ribose groups had been oxidized to create aldehydes. After desalting to remove excess ADH, EDC (Carbodiimide Reaction Chemistry) could be used to conjugate a carboxyl-containing label or carrier molecule to the amine-derivatized nucleic acid.
2. Prepare specific glycoprotein conjugates
Heterobifunctional hydrazide crosslinkers whose opposing end contains a sulfhydryl-reactive maleimide group are useful for conjugating glycoproteins to other proteins or molecules. See the page on Sulfhydryl-Reactive Crosslinker Chemistry for information on the maleimide group. Aldehyde-to-sulfhydryl crosslinkers are mentioned here, because maleimides are one of the few groups that can be paired opposite hydrazides in a single reagent. This is because the hydrazide group contains a primary amine and cannot be paired in a single molecule with an amine-reactive group, such as an NHS ester.
However, this limited reagent selection does not mean that applications for conjugation to carbohydrates are limited to proteins that have native sulfhydryl groups. It simply means that whatever macromolecule one wishes to attached to a carbohydrate must first be modified to contain a sulfhydryl group. Analogous to the bis-hydrazide scenarios described above, reagents exist to modify molecules to contain sulfhydryl groups. For example, Traut's Reagent (2-Iminothiolane, 2-IT) and SATA will add sulfhydryl groups onto primary amine sites.
When maleimide-hydrazide crosslinking is done sequentially, reaction to sulfhydryls is usually performed first followed by reaction to the molecule containing prepared aldehydes. The opposite sequence can be done, but initial steps must be performed quickly to prevent hydrolysis of the maleimide group.
The best crosslinking approach and strategy of modification to prepare respective molecular targets for conjugation depends on several factors. Although conditions can be adapted to use maleimide-hydrazide crosslinkers, many similar conjugation goals can be accomplished using different strategies. For example, see the discussion of reductive amination at the end of this page with respect to preparing antibody-HRP conjugates.
3. Glycoprotein labeling
Hydrazide-activated biotin compounds have a number of useful applications for biotinylating glycoproteins and other glycosylated molecules or polysaccharides. Biotin is a very specific tag through which biotinylated molecules can be affinity-purified or detected in plate-based assays or western blots using streptavidin resins or probes.
Example 1 (biotinylating a purified glycoprotein) – Custom antibodies (IgG) are most commonly biotinylated for use in ELISA and other immunodetection procedures by way of primary amines using NHS-ester reagents. However, that strategy can cause inactivation of the antibody if the antigen-binding sites contain primary amines that become labeled. An alternative is to biotinylate the antibody at glycosylation sites, which most antibodies (especially polyclonals) have at discrete locations away from the antigen-binding domains. Many purified enzymes and binding proteins of interest can be biotinylated and kept functional using this strategy if they are (1) glycosylated at sites away from functional domains and (2) not inactivated by treatment with periodate.
Example 2 (cell surface glycoprotein labeling) – In situ or complex mixtures of polysaccharides and glycoproteins can be labeled with biotin for later purification or detection using hydrazide-biotin reagents. When treatment of samples with periodate is not compatible with the biology being investigated, enzymatic methods of aldehyde-generation (e.g., neuraminidase, galactose oxidase) have been used (see Bayer, EA, et al., 1988 Anal. Biochem. 170:271-81).
4. Glycoprotein immobilization
In the same way that antibodies (or other purified glycoproteins) can be biotinylated through carbohydrate groups to retain unhindered and functional binding sites, they can be immobilized to affinity resins and other solid supports that are activated to contain hydrazide groups.
For example, GlycoLink Coupling Resin is hydrazide-activated Thermo Scientific UltraLink Resin, a durable, acrylamide-type porous resin for protein affinity purification methods. The resin and kit procedures are optimized for antibody immobilization using sodium periodate for carbohydrate preparation and aniline for catalysis (see above discussion of Aniline Catalysis). A small-scale immobilization kit and method are also available for using this technique to perform immunoprecipitation.
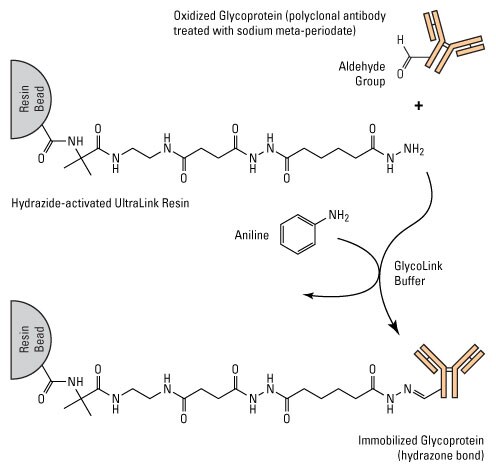
Chemistry of glycoprotein immobilization with GlycoLink Resin Proteins with oxidized sugars contain aldehyde groups which will conjugate to the hydrazide-activated beaded resin support
In principle, nearly any hard or porous surface material that can be modified to contain hydrazide groups could be used to immobilize molecules containing oxidizable carbohydrates or other aldehyde-containing moieties.
Alkoxyamine reaction chemistry
Although not currently as popular or common as hydrazide reagents, alkoxyamine compounds conjugate to carbonyls (aldehydes and ketones) in much the same manner as hydrazides. In this case, the reaction results in an oxime linkage. The similarity to hydrazides also includes the use of aniline as a catalyst. Alkoxyamine is also commonly called an "aminooxy" or "aminoxy" group.

Alkoxyamine reaction scheme for chemical conjugation to an aldehyde. R represents a labeling reagent or one end of a crosslinker having the alkoxyamine (aminooxy, aminoxy) reactive group; P represents a glycoprotein or other glycosylated molecule that contains the target functional group (i.e., an aldehyde formed by periodate oxidation of carbohydrate-sugar groups, such as sialic acid).
Applications for alkoxyamine crosslinking
Few alkoxyamine reagents are currently available. In principle, any of the hydrazide-based applications discussed above could be accomplished with alkoxyamine analogs.
Select products
Reductive amination reaction chemistry
As described in the introduction of this article, aldehyde conjugation is necessarily also amine conjugation when biological samples are concerned. In other words, this section applies as much to the article about Amine-reactive Crosslinking Chemistry as it does to the current page on carbonyl chemistry.
In reductive amination, the electrophilic carbon atom of an aldehyde attacks the nucleophilic nitrogen of a primary amine to yield a weak bond called a Schiff base. Unlike the bond formed with hydrazide or alkoxyamines (discussed above), the Schiff base formed with ordinary amines rapidly hydrolyzes (reverses) in aqueous solution and must be reduced to an alkylamine (secondary amine) linkage to stabilize it. Sodium cyanoborohydride (NaCNBH3) is a mild reducing agent that performs this function effectively without also reducing other chemical groups in biological samples.
Like Carbodiimide Crosslinking Chemistry (carboxyl-to-amine), reductive amination (aldehyde-to-amine) is a zero-length crosslinking method (no spacer arm is incorporated into the conjugate).
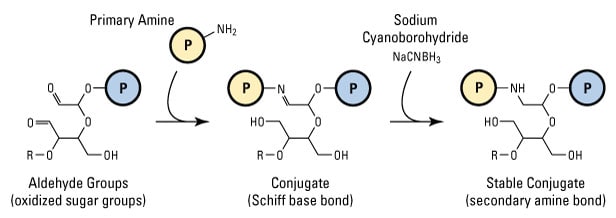
Reductive amination, the conjugation of aldehydes and primary amines. The initial reaction results in a weak, reversible Schiff base linkage. Reduction with sodium cyanoborohydride creates a stable, irreversible secondary amine bond.
Learn more
Select products
Applications for reductive amination crosslinking
1. Formaldehyde and glutaraldehyde crosslinking
These two reactive aldehyde compounds crosslink aggressively and somewhat indiscriminantly among amines and other groups via a combination of polymerization, Schiff base formation (amination) and Mannich reactions (active hydrogens). Together, these properties provide the basis for their widespread use as preservatives and fixatives with tissue and cell culture samples for microscopy and staining, immunohistochemistry (IHC) and high content screening.
Glutaraldehyde (a bis-aldehyde), if added in large excess, can be used to aldehyde-activate the amines of one purified protein for conjugation by reductive amination to another protein. This is the reverse of the hydrazide-activation strategy described above using ADH (a bis-hydrazide) with dextran.
Learn more
Select products
2. Protein to glycoprotein conjugation
The aldehyde-activation of horseradish peroxidase enzyme (HRP) and its conjugation to an antibody (IgG) serves to illustrate one application of reductive amination for protein crosslinking. HRP is a glycoprotein, and reactive aldehydes can be created by periodate oxidation without adversely affecting its functional activity. When the prepared HRP (40kDa) is combined in an appropriate ratio (usually 3- to 5-fold excess) with IgG (150 kDa), the two proteins will conjugate by reductive amination. Sodium cyanoborohydride is added to stabilize the bonds.
HRP is a commonly needed reporter enzyme, so it is commercially available in pre-activated (periodated-treated) form, ready for conjugation with purified antibodies.
Using periodate-activated HRP is more effective than the traditional method, which used glutaraldehyde, as described above. Most antibodies are also glycoproteins; therefore, the entire strategy could be reversed in this example. Thus, depending on the unique properties of the proteins needing to be conjugated, one or another conjugation method or orientation may be more effective. Often, empirical testing is the only way to determine which strategy is best.

Protein functional group targets located on a representative protein. This illustration depicts the generalized structure of an immunoglobulin (IgG) protein. Heavy and light chains are held together by a combination of non-covalent interactions and covalent interchain disulfide bonds, forming a bilaterally symmetric structure. The V regions of H and L chains comprise the antigen-binding sites of the immunoglobulin (Ig) molecules. Each Ig monomer contains two antigen-binding sites and is said to be bivalent.The hinge region is the area of the H chains between the first and second C region domains and is held together by disulfide bonds. This flexible hinge (found in IgG, IgA and IgD, but not IgM or IgE) region allows the distance between the two antigen-binding sites to vary. Also shown are several functional groups that are selectable targets for practical bioconjugation.
Select products
- Plus HRP Labeling Kit (periodate-activated)
- HRP Labeling Kit (glutaraldehyde-activated)
- Maleimide-activated HRP (sulfhydryl-reactive)
- Antibody Selection Tool
3. Protein immobilization through primary amines
One of the most reliable and popular methods (in manufacturing as well as research) for covalently attaching antibodies or other proteins to beaded agarose resin for affinity purification procedures involves the use of ordinary reductive amination. AminoLink Resin and AminoLink Plus Resin are carefully prepared varieties of aldehyde-activated agarose resin. As described in the article on Amine-reactive Crosslinker Chemistry, nearly all proteins have multiple primary amines available on their surfaces that provide numerous target sites for conjugation (in this case, immobilization).
In the appropriate amine-free, alkaline buffer with (or followed by the addition of) sodium cyanoborohydride, proteins conjugate to the agarose resin by permanent secondary amine bonds. High densities of stably immobilized protein are possible, enabling many repeated rounds (binding, washing, elution) of affinity purification without degradation or leaching. AminoLink Plus Resin is also the basis for the Direct IP Kit for immunoprecipitation. The process is outlined in the following diagram.

Antibody immobilization. This illustration depicts the chemistry of Thermo Scientific AminoLink Plus Coupling Resin. The resin is aldehyde-activated beaded agarose to which antibodies, other proteins and molecules can be covalently attached through primary amines. The result is a reusable affinity resin for use in a variety of purification methods.
The standard AminoLink Resin procedure uses near-neutral coupling buffer (PBS, pH 7.2) in which the sodium cyanoborohydride (AminoLink Reductant) is included for the duration of the reaction. An optional high-yield procedure with AminoLink Plus Resin involves initial incubation of protein and resin in a more alkaline coupling buffer (citrate-carbonate, pH 10). Schiff-base formation is much more efficient in this alkaline condition, but then the buffer must be changed to near-neutral PBS before adding the sodium cyanoborohydride for the reducing step. Proteins that are not compatible with the high-pH coupling condition can be immobilized using the near-neutral condition. Various additives and components can be included to make or keep proteins soluble for the reaction.
- Byeon, J.Y., et al. (2010). Efficient bioconjugation of protein capture agents to biosensor surfaces using aniline-catalyzed hydrazone ligation. Langmuir 26(19):15430-5.
- Dirksen, A., et al. (2006). Nucleophilic catalysis of hydrazone formation and transimination: implications for dynamic covalent chemistry. J. Am. Chem. Soc. 128(49):15602-3.
- Dirksen, A., Dawson, PE. (2008). Rapid oxime and hydrazone ligations with aromatic aldehydes for biomolecular labeling. Bioconjug. Chem. 19(12):2543-8.Bayer, EA, et al., 1988 Anal. Biochem. 170:271-81
For Research Use Only. Not for use in diagnostic procedures.
