Search
Strategies for Detecting mRNA: Northern Blotting, Nuclease Protection Assays, In Situ Hybridization, and RT-PCR
Molecular characterization of any gene usually includes a thorough analysis of the temporal and spatial distribution of RNA expression. A number of widely used procedures exist for detecting and determining the abundance of a particular mRNA in a total or poly(A) RNA sample. Here, we review four popular methods: Northern blot analysis, nuclease protection assays (NPA), in situ hybridization, and reverse transcription-polymerase chain reaction (RT-PCR).
In theory, each of these techniques can be used to detect specific RNAs and to precisely determine their expression level. However, the methods each have inherent advantages and/or limitations. In general, Northern analysis is the only method that provides information about transcript size, whereas NPAs are the easiest way to simultaneously examine multiple messages (Figure 1). In situ hybridization is used to localize expression of a particular gene within a tissue or cell type, and RT-PCR is the most sensitive method for detecting and quantitating gene expression.
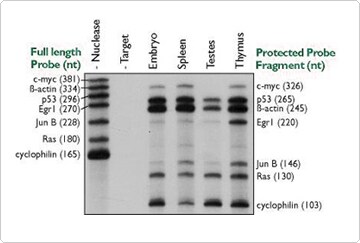
Figure 1. Simultaneous analysis of multiple mRNAs using RPA III. Ten micrograms of various mouse tissue total RNAs were hybridized overnight with approximately 50,000 cpm each of seven distinct probe transcripts. Nuclease digestion, product separation on a denaturing 6% acrylamide gel, and a four-hour exposure to film at -80°C were used to assess hybridization levels.
Northern Analysis
Northern analysis remains the standard for detection and quantitation of mRNA levels despite the advent of more sensitive techniques. Northern analysis presents several advantages over the other techniques. The most compelling of these is that it is the easiest method for determining transcript size, and for identifying alternatively spliced transcripts and multigene family members. It can also be used to directly compare the relative abundance of a given message between all the samples on a blot.
The Northern blotting procedure is straightforward and provides opportunities to evaluate progress at various points (e.g., intactness of the RNA sample and how efficiently it has transferred to the membrane). RNA samples are first separated by size via electrophoresis in an agarose gel under denaturing conditions. The RNA is then transferred to a membrane, crosslinked and hybridized with a labeled probe. Nonisotopic or high specific activity radiolabeled probes can be used including random-primed, nick-translated, or PCR-generated DNA probes, in vitro transcribed RNA probes, and oligonucleotides. Additionally, sequences with only partial homology (e.g., cDNA from a different species or genomic DNA fragments that might contain an exon) may be used as probes.
Despite these advantages, there are limitations associated with Northern analysis. First, if RNA samples are even slightly degraded, the quality of the data and the ability to quantitate expression are severely compromised. Second, a standard Northern procedure is, in general, the least sensitive of the reviewed techniques. However, substantial improvements can be made to increase detection of most mRNA species (see below). Another limitation of Northern blotting has been the difficulty associated with multiple probe analysis. To detect more than one message, it is usually necessary to strip the initial probe before hybridizing with a second probe. This process can be time consuming and problematic.
Making a Good Technique Even Better
Although established Northern blotting procedures are up and working in most molecular biology laboratories, Ambion has found ways to considerably improve on standard protocols, resulting in greatly increased Northern sensitivity. We have developed RNase-free reagents optimized for each step of the procedure to provide complete, high-sensitivity Northern blotting systems. The Invitrogen NorthernMax Kits provide everything needed to perform Northern analysis except for the membrane and probe. Each of the reagents is also available separately including the NorthernMax Rapid Transfer Buffer, that facilitates complete transfer in as little as an hour and a half. The new ULTRAhyb Ultrasensitive Hybridization Buffer increases sensitivity up to 100 fold (Figure 2) by pushing hybridization to completion. As few as 10,000 molecules can be detected.
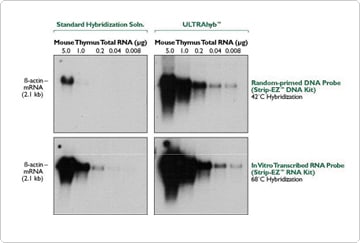
Figure 2. ULTRAhyb versus a Standard Hybridization Buffer Using DNA and RNA Probes. Identical Northern blots of indicated amounts of mouse thymus total RNA were hybridized overnight with radiolabeled Invitrogen StripAble DNA or RNA probe. The blots were incubated with probe in either ULTRAhyb or standard hybridization buffer as indicated. Standard low and high stringency SSC/SDS washes were used to remove non-specifically bound probe. Blots were exposed to film for 2.5 hours with a single intensifying screen.
Nuclease Protection Assays (NPAs)
The NPA (including both ribonuclease protection assays and S1 nuclease assays) is an extremely sensitive method for the detection and quantitation of specific mRNAs. The basis of the NPA is solution hybridization of an antisense probe (radiolabeled or nonisotopic) to an RNA sample. After hybridization, single-stranded, unhybridized probe and RNA are degraded by nucleases. The remaining protected fragments are separated on an acrylamide gel. Solution hybridization is typically more efficient than membrane-based hybridization, and it can accommodate up to 100 µg of sample RNA, compared with the 20-30 µg maximum of blot hybridizations. NPAs are also less sensitive to RNA sample degradation than Northern analysis since cleavage is only detected in the region of overlap with the probe (probes are usually about 100-400 bases in length).
NPAs are the method of choice for the simultaneous detection of several RNA species. Figure 1 shows how 6 different probes and an internal control can be used together for detection of mRNA transcripts each within 4 different RNA samples. During solution hybridization and subsequent analysis, individual probe/target interactions are completely independent of one another. Thus, several RNA targets and appropriate controls can be assayed simultaneously (up to twelve have been used in the same reaction), provided that the individual probes are of different lengths. NPAs are also commonly used to precisely map mRNA termini and intron/exon junctions.
The primary limitation of NPAs is the lack of information on transcript size. The portion of probe homologous to target RNA determines the size of the protected fragment. Another drawback to NPAs is the lack of probe flexibility. The most common type of NPA, the ribonuclease protection assay, requires the use of RNA probes. Oligonucleotides and other single-stranded DNA probes can only be used in assays containing S1 nuclease. The single-stranded, antisense probe must typically be completely homologous to target RNA to prevent cleavage of the probe:target hybrid by nuclease. This means that partially related sequences (i.e., cross species) usually cannot be used.
RNA Analysis in a Single Tube
Invitrogen Ambion released the first commercially available Ribonuclease Protection Assay Kit in 1990 and has since continued to refine and improve the procedure resulting in a number of kits with different advantages. The Invitrogen RPA III Kit is recommended for all new users and consists of a single-tube protocol. The Invitrogen HybSpeed RPA Kit utilizes a 10-minute hybridization step so that the entire assay can be completed in a single day.
In Situ Hybridization
The procedure begins by fixing samples in neutral-buffered formalin, and embedding the tissue in paraffin. The samples are then sliced into thin sections and mounted onto microscope slides. (Alternatively, tissue can be sectioned frozen and post-fixed in paraformaldehyde.) After a series of washes to dewax and rehydrate the sections, a Proteinase K digestion is performed to increase probe accessibility, and a labeled probe is then hybridized to the sample sections. Radiolabeled probes are visualized with liquid film dried onto the slides, while nonisotopically labeled probes are conveniently detected with colorimetric or fluorescent reagents.
The major drawback to ISH is the procedure itself. Standard protocols are time-consuming, laborious and may require specialized equipment for preparing samples and visualizing results of the experiment. Additionally, quantitation of gene expression is not as straightforward as with the other techniques.
RT-PCR
RT-PCR has revolutionized the study of gene expression. It is now theoretically possible to detect the RNA transcript of any gene, regardless of the scarcity of the starting material or relative abundance of the specific mRNA. In RT-PCR, an RNA template is copied into a complementary DNA (cDNA) using a retroviral reverse transcriptase. The cDNA is then amplified exponentially by PCR. As with NPAs, RT-PCR is somewhat tolerant of degraded RNA. As long as the RNA is intact within the region spanned by the primers, the target will be amplified.
Although RT-PCR is the most sensitive method of mRNA detection available, it does have drawbacks. It can be the most technically challenging method of detection and quantitation, often requiring substantial pre-experimental planning and design. Additionally, because of its extreme sensitivity, even minute amounts of contamination by genomic DNA or previously amplified PCR products can lead to aberrant results, so steps must be taken to avoid this pitfall.
Relative quantitative RT-PCR involves amplifying an internal control simultaneously with the gene of interest. The internal control is used to normalize the samples. Once normalized, direct comparisons of relative abundance of a specific mRNA can be made across the samples. It is crucial to choose an internal control with a constant level of expression across all experimental samples (i.e., not affected by experimental treatment). Commonly used internal controls (e.g., GAPDH, -actin, cyclophilin) often vary in expression and, therefore, may not be appropriate internal controls. Additionally, most common internal controls are expressed at much higher levels than the mRNA being studied. For relative RT-PCR results to be meaningful, all products of the PCR reaction must be analyzed in the linear range of amplification. This becomes difficult for transcripts of widely different levels of abundance.
Competitive RT-PCR is used for absolute quantitation. This technique involves designing, synthesizing, and accurately quantitating a competitor RNA that can be distinguished from the endogenous target by a small difference in size or sequence. Known amounts of the competitor RNA are added to experimental samples and RT-PCR is performed. Signals from the endogenous target are compared with signals from the competitor to determine the amount of target present in the sample.
Making RT-PCR Work for You
Invitrogen Ambion has a complete line of products to simplify RT-PCR. The Invitrogen RETROscript Kit provides a convenient and flexible system for synthesizing first-strand cDNA. The Invitrogen QuantumRNA 18S Internal Standards Kits utilize Invitrogen Ambion's Competimer technology (patent pending) to allow the use of the best internal control, 18S rRNA, for relative RT-PCR (Figure 3).
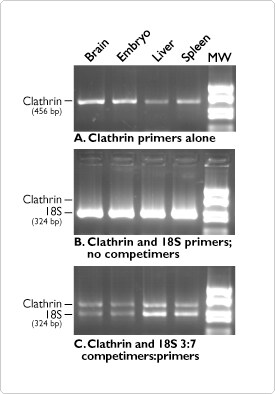
Figure 3. Invitrogen Ambion's QuantumRNA Technology in Multiplex Quantitative RT-PCR using 18S rRNA as an Internal Control. RT-PCR reactions on brain, embryo, liver, and spleen total RNA using A) primers for clathrin, B) primers for clathrin and 18S, or C) primers for clathrin, 18S rRNA primers and 18S rRNA Competimers. Note that without Competimers, 18S cannot be used as an internal control because of its high abundance (B). Addition of Competimers (C) makes multiplex PCR possible, providing sample-to-sample relative quantitation.
Summary
For Research Use Only. Not for use in diagnostic procedures.