Search
Pull-Down Assays
View and select products
Introduction to pull-down assays
The pull-down assay is an in vitro method used to determine a physical interaction between two or more proteins. Pull-down assays are useful for both confirming the existence of a protein–protein interaction predicted by other research techniques (e.g., co-immunoprecipitation) and as an initial screening assay for identifying previously unknown protein–protein interactions.
Pull-down assays are a form of affinity purification and are similar to immunoprecipitation, except that a "bait" protein is used instead of an antibody. Affinity chromatography (i.e., affinity purification) methodologies greatly enhance the speed and efficiency of protein purification and simultaneously provide the technology platform to perform a pull-down, or co-purification, of potential binding partners. In a pull-down assay, a bait protein is tagged and captured on an immobilized affinity ligand specific for the tag, thereby generating a "secondary affinity support"’ for purifying other proteins that interact with the bait protein. The secondary affinity support of immobilized bait is then incubated with a protein source that contains putative "prey" proteins, such as a cell lysate. The source of prey protein at this step depends on whether the researcher is confirming a previously suspected protein–protein interaction or identifying an unknown interaction. The method of protein elution depends on the affinity ligand and ranges from using competitive analytes to low pH or reducing buffers.
Besides investigating the interaction of two or more proteins, pull-down assays are a powerful tool to detect the activation status of specific proteins. For example, proteins that are activated in response to tyrosine phosphorylation can be pulled down using an immobilized SH2 domain that targets the phosphorylated tyrosine on a given protein. Additionally, GTPases, which act as molecular switches that regulate cell signaling by cycling between a GTP-bound (active) and GDP-bound (inactive) state, can be pulled down using an immobilized GTPase-binding domain of downstream proteins that are recruited to GTP-bound, activated GTPases. In both types of pull-down assays, because the specificity of the interaction is dependent on the sequence of the binding domain, these approaches are highly specific in detecting the activation of distinct proteins.
Protein Preparation Handbook
Learn more about how to desalt, buffer exchange, concentrate, and/or remove contaminants from protein samples, immunoprecipitation and other protein purification and clean up methods using various Thermo Scientific protein biology tools in this 32-page handbook.
- Immunoprecipitation (IP), co-IP, and chromatin-IP
- Recombinant protein purification tags
- Dialyze protein samples securely using Slide-A-Lyzer dialysis cassettes and devices
- Rapidly desalt samples with high protein recovery using Zeba spin desalting columns and plates
- Efficiently extract specific contaminants using resins optimized for detergent or endotoxin removal
- Concentrate dilute protein samples quickly using Pierce protein concentrators

General schematic of a pull-down assay. A pull-down assay is a small-scale affinity purification technique similar to immunoprecipitation, except that the antibody is replaced by some other affinity system. In this case, the affinity system consists of a glutathione S-transferase (GST)–, polyHis- or streptavidin-tagged protein or binding domain that is captured by glutathione-, metal chelate (cobalt or nickel) – or biotin-coated agarose beads, respectively. The immobilized fusion-tagged protein acts as the "bait" to capture a putative binding partner (i.e., the "prey"). In a typical pull-down assay, the immobilized bait protein is incubated with a cell lysate, and after the prescribed washing steps, the complexes are selectively eluted using competitive analytes or low pH or reducing buffers for in-gel or western blot analysis.
The pull-down assay as a confirmatory tool
The confirmation of previously suspected interactions typically utilizes a prey protein source that has been expressed in an artificial protein expression system. This allows the researcher to work with a larger quantity of the protein than is typically available under endogenous expression conditions and eliminates confusing results that could arise from interaction of the bait with other interacting proteins present in the endogenous system that are not under study. Protein expression system lysates (i.e., E. coli or baculovirus-infected insect cells), in vitro transcription/translation reactions, and previously purified proteins are appropriate prey protein sources for confirmatory studies.
The pull-down assay as a discovery tool
The discovery of unknown interactions contrasts with confirmatory studies because the research interest lies in discovering new proteins in the endogenous environment that interact with a given bait protein. The endogenous environment can entail a plethora of possible protein sources but is generally characterized as a complex protein mixture considered to be the native environment of the bait protein. Any cellular lysate in which the bait is normally expressed, or complex biological fluid (i.e., blood, intestinal secretions, etc.) where the bait would be functional, is an appropriate prey protein source for discovery studies.
Learn more
Critical components of pull-down assays
Bait protein criteria
Bait proteins for pull-down assays can be generated either by linking an affinity tag to proteins purified by traditional purification methods or by expressing recombinant fusion-tagged proteins. Researchers who have access to commercially available purified protein or frozen aliquots of purified protein from an earlier study can design a pull-down assay without the need for cloning the gene encoding the protein of interest. The purified protein can be tagged with a protein-reactive tag commonly used for such labeling applications. Alternatively, if a cloned gene is available, molecular biology methods can be employed to subclone the gene to an appropriate vector with a fusion tag. Recombinant clones can be overexpressed and easily purified, resulting in an abundance of bait protein for use in pull-down assays. In the representative example below, glutathione-S-transferase (GST) is utilized to perform a pull-down experiment.
Common fusion tags and their affinity binding ligands
| Fusion tag | Affinity ligand |
|---|---|
| Glutathione S-transferase (GST) | Glutathione |
| Poly-histidine (polyHis or 6xHis) | Nickel or cobalt chelate complexes |
| Biotin | Streptavidin |
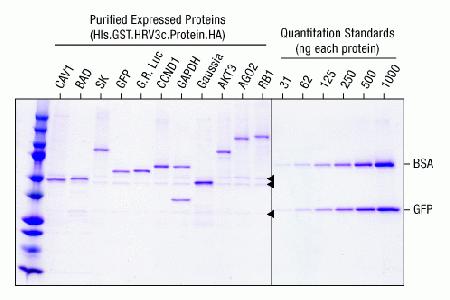
Purification of N-terminal GST fusion proteins with immobilized glutathione. Genes cloned into pT7CFE1-NHis-GST-CHA were used to express GST-fusion proteins for 6 hours. After binding to Thermo Scientific Pierce glutathione agarose, the GST-fused proteins were eluted with 50 mM glutathione. The additional bands (denoted with arrows) found with the purified proteins were previously identified by mass spectrometry as cellular proteins eEF1G and GSTM3, which are known to bind to the glutathione column and co-elute with GST tagged proteins. Approximately 500 ng of each of the purified proteins was separated by SDS-PAGE and stained with the Thermo Scientific Pierce Power Stainer (Cat. No. 22833). Abbreviations: His: repeat of 6X Histidines; GST: Glutathione S Transferase; HRV3c site: Human Rhinovirus 3C Protease site; GOI: gene of interest; HA: Haemaglutunnin sequence; BSA: Bovine serum albumin; GFP: Green fluorescent protein; CAV1: caveolin1; BAD: Bcl-2-associated death promoter; Green Renilla: Green Renilla luciferase; CCND1: cyclin D1; Guassia: Guassia luciferase; AKT3: protein kinase; AGO2: Argonaute RISC catalytic component 2; and RB1: Retinoblastoma 1.
Binding parameters: stable vs. transient interactions
The discovery and confirmation of protein–protein interactions using the pull-down technique depend heavily on the nature of the interaction under study. Interactions can be stable or transient, and this characteristic determines the conditions for optimizing binding between the bait and prey proteins. Transient interactions are usually associated with transport or enzymatic mechanisms. The ribosome illustrates both examples because the structure consists of many stable protein–protein interactions, but the enzymatic mechanism that translates mRNA to nascent protein requires transient interactions.
Stable protein–protein interactions are easiest to isolate by physical methods like pull-down assays because the protein complex does not disassemble over time. Strong, stable protein complexes can be washed extensively with high ionic strength buffers to eliminate any false positive results due to nonspecific interactions. If the complex interaction has a higher dissociation constant and a weaker interaction, the interaction strength and thus the protein complex recovery can be improved by optimizing the assay conditions related to pH, salt species and salt concentration. Problems of nonspecific interactions can be minimized with the careful design of appropriate control experiments. This example provides an example of an experiment designed to pull-down the active form of Cdc42 using a specialized kit.
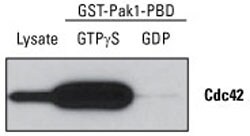
Detection of active Cdc42. The Active Cdc42 Pull-Down and Detection Kit was used to detect active protein present in NIH 3T3 cell lysate treated with GTPγS (activator) or GDP (inactivator). Active Cdc42 was enriched by pull-down assay. Half of each eluate (25 µL) and 40 µg of total lysate were analyzed by western blot using the anti-Cdc42 antibody supplied in the kit. Only active Cdc42 was detected, as indicated by the lower signal intensity in the GDP-treated sample.
Transient interactions are defined by their temporal interaction with other proteins and are the most challenging protein-protein interactions to isolate. These interactions are more difficult to identify using physical methods like pull-down assays because the complex may dissociate during the assay. Since transient interactions occur during transport or as part of enzymatic processes, they often require cofactors and energy via nucleotide triphosphates hydrolysis. Incorporating cofactors and nonhydrolyzable nucleoside triphosphate (NTP) analogs during assay optimization can serve to ‘trap’ interacting proteins in different stages of a functional complex that is dependent on the cofactor or NTP.
Weak or transient protein–protein interactions can be strengthened by covalently crosslinking the interacting proteins prior to pull-down. While this strategy is more advanced than performing the pull-down assay without crosslinking, freezing protein interactions by crosslinking may make or break the success of a pull-down assay.
Elution of the bait–prey complex
The identification of bait–prey interactions requires that the complex is removed from the affinity support and analyzed by standard protein detection methods. The entire complex can be eluted from the affinity support using sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) loading buffer or a competitive analyte specific for the tag on the bait protein. SDS-PAGE loading buffer is a harsh treatment that will denature all protein in the sample and restricts the sample to SDS-PAGE analysis only. This method may also strip excess protein off the affinity support that is nonspecifically bound to the matrix, and this material can interfere with analysis. Competitive analyte elution is much more specific for the bait–prey interaction, because it does not strip proteins that are nonspecifically bound to the affinity support. This method is non-denaturing; thus, it can elute a biologically functional protein complex, which could be useful for subsequent studies.
An alternative elution protocol is to use a step-wise gradient of increasing salt concentration or decreasing pH, which allows the selective elution of prey proteins while the bait remains immobilized. A gradient elution is not necessary once the critical salt concentration or pH has been optimized for efficient elution. These elution methods are also non-denaturing and can be informative in determining the relative interaction strength.
Gel detection of bait–prey complex
Protein complexes contained in eluted samples can be visualized by SDS-PAGE and associated detection methods, including gel staining, Western blotting detection and S-35 radioisotopic detection. The final determination of interacting proteins often entails protein band isolation from a polyacrylamide gel, tryptic digestion of the isolated protein and mass spectrometric identification of digested peptides.
Pull-down assay controls
Pull-down assays entail multiple steps often using more than one cell lysate, and therefore each experiment must be properly controlled to demonstrate that the final results are not artifactual. Each experiment should analyze the lysate(s) both before and after being passed through the support to identify any nonspecific binding to the support. Each wash should also be analyzed to observe any eluted protein, and bait- and prey-free controls should also be used to confirm that there are no bait–prey interactions in the bait lysate and that the prey protein does not bind to the immobilized support.
Learn more
- Pull-Down Assays
- Overview of Affinity Purification
- GST-tagged Proteins – Production and Purification
- His-tagged Proteins – Production and Purification
- Avidin-Biotin Interaction
- Overview of Protein-Protein Interaction Analysis
- Crosslinking Protein Interaction Analysis
- Tech Tip #27: Optimize elution conditions for immunoaffinity purification
- Overview of Western Blotting
- Workflow for Protein Mass Spectrometry
Pull-down methodologies
Homemade pull-down approaches for confirming or identifying protein–protein interactions are ubiquitous in contemporary scientific literature. The homemade pull-down assay represents a collection of reagents from multiple commercial vendors that cannot be validated together as a functional assembly except by extensive assay development by the researcher, and troubleshooting this combination of reagents can be tedious and time consuming. Commercial pull-down kits contain complete, validated sets of reagents specifically developed for performing pull-down assays. The buffers provided in each kit allow complete flexibility to determine the optimal conditions for isolating interacting proteins. The working solutions for washing and binding are physiologic in pH and ionic strength, providing a starting point from which specific buffer conditions for each unique interacting pair can be optimized. Many commercial kits also incorporate spin columns for efficient handling of small volumes of affinity support, complete retention of the affinity support during the pull-down assay and thorough washing of the protein complexes for minimal nonspecific protein pull-down, all of which are common sources of variability and high background using traditional pull-down assay formats.
Learn more
- Protein Interaction Analysis
- Tech Tip #64: Immunoprecipitation (IP) technical guide and protocols
- Application Note: Calculate the Number of Immobilized Proteins per Bead of Agarose Affinity Supports
- Protein Immunoprecipitation (IP), Co-Immunoprecipitation (Co-IP), and Pull-down Support
Select products
Recommended reading
- Einarson MB, Orlinick JR (2002) Identification of Protein-Protein Interactions with Glutathione S-Transferase Fusion Proteins. In: Protein-Protein Interactions: A Molecular Cloning Manual. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press. pp 37–57.
- Einarson MB (2001) Detection of Protein-Protein Interactions Using the GST Fusion Protein Pulldown Technique. In: Molecular Cloning: A Laboratory Manual, 3rd Edition. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press. pp 18.55–18.59.
- Vikis HG, Guan K-L (2004) Glutathione-S-Transferase-Fusion Based Assays for Studying Protein-Protein Interactions. In: Fu H (editor), Protein-Protein Interactions, Methods and Applications, Methods in Molecular Biology, 261. Totowa (NJ): Humana Press. pp 175–186.
For Research Use Only. Not for use in diagnostic procedures.
