Search
Page Contents
We offer a large assortment of fluorogenic and chromogenic enzyme substrates, which are described in the following sections. We prepare substrates for detecting very low levels of enzymatic activity in fixed cells, tissues, cell extracts and purified preparations, as well as substrates for enzyme-linked immunosorbent assays (ELISAs). Our RediPlate product line includes enzyme substrates predispensed in 96-well plates for high-throughput applications, along with the appropriate reference standards and other reaction components. We have also developed effective methods for detecting some enzymes in live cells.
In this section, we describe the characteristics of our enzyme substrates and the fluorophores and chromophores from which they are derived, focusing primarily on the suitability of these substrates for different types of enzyme assays. The fluorophores that are available as reference standards—including a NIST-traceable fluorescein standard (F36915)—can be found in the data table and product list associated with this section. Substrates for specific enzymes are described in subsequent sections of this chapter.
Solution assays designed to quantitate enzymatic activity in cell extracts or other biological fluids typically employ substrates that yield highly fluorescent or intensely absorbing water-soluble products. ELISAs also rely on these substrates for indirect quantitation of analytes.![]() An ideal fluorogenic substrate for fluorescence-based solution assays yields a highly fluorescent, water-soluble product with optical properties significantly different from those of the substrate. If the fluorescence spectra of the substrate and product overlap significantly, analysis will likely require a separation step, especially when using excess substrate to obtain pseudo–first-order kinetics. Fortunately, many substrates have low intrinsic fluorescence or are metabolized to products that have longer-wavelength excitation or emission spectra (Figure 10.1.1). These fluorescent products can typically be quantitated in the presence of the unreacted substrate using a fluorometer or a fluorescence microplate reader. Microplate readers facilitate high-throughput analysis and require relatively small assay volumes, which usually reduces reagent costs. Moreover, the front-face optics in many microplate readers allows researchers to use more concentrated solutions, which may both improve the linearity of the kinetics and reduce inner-filter effects.
An ideal fluorogenic substrate for fluorescence-based solution assays yields a highly fluorescent, water-soluble product with optical properties significantly different from those of the substrate. If the fluorescence spectra of the substrate and product overlap significantly, analysis will likely require a separation step, especially when using excess substrate to obtain pseudo–first-order kinetics. Fortunately, many substrates have low intrinsic fluorescence or are metabolized to products that have longer-wavelength excitation or emission spectra (Figure 10.1.1). These fluorescent products can typically be quantitated in the presence of the unreacted substrate using a fluorometer or a fluorescence microplate reader. Microplate readers facilitate high-throughput analysis and require relatively small assay volumes, which usually reduces reagent costs. Moreover, the front-face optics in many microplate readers allows researchers to use more concentrated solutions, which may both improve the linearity of the kinetics and reduce inner-filter effects.
When the spectral characteristics of the substrate and its metabolic product are similar, techniques such as thin-layer chromatography (TLC), high-performance liquid chromatography (HPLC), capillary electrophoresis, solvent extraction or ion exchange can be used to separate the product from unconsumed substrate prior to analysis. For example, our FAST CAT Chloramphenicol Acetyltransferase Assay Kits (F2900, F6616, F6617; Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6) utilize chromatography to separate the intrinsically fluorescent substrates from their fluorescent products.
 | Figure 10.1.1 Normalized emission spectra of 1) 7-hydroxy-4-methylcoumarin (H189) and 6,8-difluoro-7-hydroxy-4-methylcoumarin (DiFMU, D6566), 2) fluorescein (F1300, F36915), 3) resorufin (R363) and 4) DDAO (H6482) in aqueous solution at pH 9. These fluorophores correspond to the hydrolysis, oxidation or reduction products of several of our fluorogenic enzyme substrates. |
Substrates Derived from Water-Soluble Coumarins
Hydroxy- and amino-substituted coumarins have been the most widely used fluorophores for preparing fluorogenic substrates. Phosphate, ester and ether derivatives of 7-hydroxy-4-methylcoumarin (β-methylumbelliferone, H189), typified by 4-methylumbelliferyl phosphate (MUP, M6491; Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3), are only weakly fluorescent in the optimal configuration for 7-hydroxy-4-methylcoumarin detection (excitation/emission = 360/460 nm, Figure 10.1.2). Thus, enzymatic cleavage of the phosphate, ester or ether results in an increase in fluorescence under these conditions. Because the fluorescence of 7-hydroxycoumarin dyes is pH dependent, this detection configuration is only optimal when the phenolate anion form is the predominant species (i.e., at pH >8 for 7-hydroxy-4-methylcoumarin). At lower pH values, the excitation wavelength must be switched to about 320 nm, which is optimal for the protonated phenol form. 6,8-Difluoro-7-hydroxy-4-methylcoumarin (DiFMU, D6566; Figure 10.1.2) has a much lower phenol/phenolate pKa![]() (Figure 10.1.3), allowing measurements to be made over a broader pH range without adjusting the excitation/emission wavelength settings to accommodate the pH-dependent spectral shift of the 7-hydroxycoumarin fluorophore. Furthermore, the electron-withdrawing effect of the fluorine substituents manifested in the lower pKa also facilitates cleavage of the phosphate, ester or ether substituent, resulting in accelerated enzyme kinetics.
(Figure 10.1.3), allowing measurements to be made over a broader pH range without adjusting the excitation/emission wavelength settings to accommodate the pH-dependent spectral shift of the 7-hydroxycoumarin fluorophore. Furthermore, the electron-withdrawing effect of the fluorine substituents manifested in the lower pKa also facilitates cleavage of the phosphate, ester or ether substituent, resulting in accelerated enzyme kinetics.![]() Consequently, the phosphate ester of 6,8-difluoro-7-hydroxy-4-methylcoumarin (DiFMUP, D6567, D22065, E12020; Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3) is one of the most sensitive fluorogenic substrates for continuous high-throughput assay of alkaline phosphatase and its bioconjugates.
Consequently, the phosphate ester of 6,8-difluoro-7-hydroxy-4-methylcoumarin (DiFMUP, D6567, D22065, E12020; Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3) is one of the most sensitive fluorogenic substrates for continuous high-throughput assay of alkaline phosphatase and its bioconjugates.
Aromatic amines, including the commonly used 7-amino-4-methylcoumarin (AMC, A191), are partially protonated at low pH (less than ~5) but fully deprotonated at physiological pH. Thus, their fluorescence spectra are not subject to variability due to pH-dependent protonation/deprotonation when assayed near or above physiological pH. AMC is widely used to prepare peptidase substrates in which the amide has shorter-wavelength absorption and emission spectra than the amine hydrolysis product.
 | Figure 10.1.2 Absorption and fluorescence emission spectra of 7-hydroxy-4-methylcoumarin (H189) in pH 9.0 buffer. The spectra of 6,8-difluoro-7-hydroxy-4-methylcoumarin (D6566) are essentially identical. |
 | Figure 10.1.3 Comparison of the pH-dependent fluorescence responses of 7-hydroxy-4-methylcoumarin (H189) and 6,8-difluoro-7-hydroxy-4-methylcoumarin (DiFMU, D6566). Fluorescence intensities were measured for equal concentrations of the two dyes using excitation/emission at 355/460 nm. Under these conditions, DiFMU (pKa = 4.9) provides superior detection sensitivity in the 5–8 pH range compared to 7-hydroxy-4-methylcoumarin (pKa = 7.8). |
Substrates Derived from Water-Soluble Green to Yellow Fluorophores
As compared with coumarin-based substrates, substrates derived from fluoresceins, rhodamines and resorufins often provide significantly greater sensitivity in fluorescence-based enzyme assays. In addition, most of these longer-wavelength dyes have extinction coefficients that are 5 to 25 times that of coumarins, nitrophenols or nitroanilines, making them additionally useful as sensitive chromogenic substrates.
Hydrolytic substrates based on the derivatives of fluorescein (fluorescein reference standard, F1300; fluorescein NIST-traceable standard, F36915) or rhodamine 110 (R110, R6479) usually incorporate two moieties, each of which serves as a substrate for the enzyme. Consequently, they are cleaved first to the monosubstituted analog and then to the free fluorophore. Because the monosubstituted analog often absorbs and emits light at the same wavelengths as the ultimate hydrolysis product, this initial hydrolysis complicates the interpretation of hydrolysis kinetics.![]() When highly purified, however, the disubstituted fluorescein- and rhodamine 110–based substrates have virtually no visible-wavelength absorbance or background fluorescence, making them extremely sensitive detection reagents. For example, researchers have reported that the activity of as few as 1.6 molecules of β-galactosidase can be detected with fluorescein di-β-D-galactopyranoside (FDG) and capillary electrophoresis.
When highly purified, however, the disubstituted fluorescein- and rhodamine 110–based substrates have virtually no visible-wavelength absorbance or background fluorescence, making them extremely sensitive detection reagents. For example, researchers have reported that the activity of as few as 1.6 molecules of β-galactosidase can be detected with fluorescein di-β-D-galactopyranoside (FDG) and capillary electrophoresis.![]() Fluorogenic substrates based on either the AMC and R110 fluorophore are used in our EnzChek Caspase Assay Kits (Assays for Apoptosis—Section 15.5) to detect apoptotic cells.
Fluorogenic substrates based on either the AMC and R110 fluorophore are used in our EnzChek Caspase Assay Kits (Assays for Apoptosis—Section 15.5) to detect apoptotic cells.
Chemical reduction of fluorescein- and rhodamine-based dyes yields colorless and nonfluorescent dihydrofluoresceins and dihydrorhodamines. Although extremely useful for detection of reactive oxygen species (ROS) in phagocytic and other cells (Generating and Detecting Reactive Oxygen Species—Section 18.2), these dyes tend to be insufficiently stable for solution assays.
Substrates Derived from Water-Soluble Red Fluorophores
Long-wavelength fluorophores are often preferred because background absorbance and autofluorescence are generally lower when longer excitation wavelengths are used. Substrates derived from the red-fluorescent resorufin (R363) and the dimethylacridinone derivative 7-hydroxy-9H-(1,3-dichloro-9,9-dimethylacridin-2-one) (DDAO, H6482; Figure 10.1.4) contain only a single hydrolysis-sensitive moiety, thereby avoiding the biphasic kinetics of both fluorescein- and rhodamine-based substrates.![]()
Resorufin is used to prepare several substrates for glycosidases, hydrolytic enzymes and dealkylases. In most cases, the relatively low pKa of resorufin (~6.0) permits continuous measurement of enzymatic activity. Thiols such as DTT or 2-mercaptoethanol should be avoided in assays utilizing resorufin-based substrates. Our Amplex Red peroxidase substrate (A12222, A22177; Substrates for Oxidases, Including Amplex Red Kits—Section 10.5) is a chemically reduced, colorless form of resorufin that is oxidized to resorufin by horseradish peroxidase (HRP) in combination with hydrogen peroxide. Resorufin is also the product of enzyme-catalyzed reduction of resazurin (R12204; Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6, Viability and Cytotoxicity Assay Reagents—Section 15.2)—also known as alamarBlue, a trademark of AccuMed International, Inc. Our Amplex UltraRed reagent (A36006, Substrates for Oxidases, Including Amplex Red Kits—Section 10.5) improves upon the performance of the Amplex Red reagent, offering brighter fluorescence and enhanced sensitivity on a per-mole basis in peroxidase or peroxidase-coupled enzyme assays.
Substrates derived from DDAO, a red He-Ne laser–excitable fluorophore, generally exhibit good water solubility, low Kms and high turnover rates. In addition, the difference between the excitation maximum of the DDAO-based substrates and that of the phenolic DDAO product is greater than 150 nm (Figure 10.1.4), which allows the two species to be easily distinguished.
 | Figure 10.1.4 Normalized absorption and fluorescence emission spectra of DDAO, which is formed by alkaline phosphatase–mediated hydrolysis of DDAO phosphate (D6487). |
We have developed a number of innovative strategies for investigating enzymatic activity in live cells.![]() For example, we offer a diverse set of probes that can passively enter the cell; once inside, they are processed by intracellular enzymes to generate products with improved cellular retention. We also offer kits and reagents for detecting the expression of several common reporter genes in cells and cell extracts. These include substrates for β-galactosidase and β-glucuronidase (Detecting Glycosidases—Section 10.2), secreted alkaline phosphatase (SEAP, Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3), chloramphenicol acetyltransferase (CAT) and luciferase (Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6). Some of our EnzChek and DQ Kits (Detecting Peptidases and Proteases—Section 10.4) are useful for study of the uptake and metabolism of proteins during phagocytosis (Probes for Following Receptor Binding and Phagocytosis—Section 16.1). Substrates for specific proteases are also useful for the detection of apoptosis (Assays for Apoptosis—Section 15.5).
For example, we offer a diverse set of probes that can passively enter the cell; once inside, they are processed by intracellular enzymes to generate products with improved cellular retention. We also offer kits and reagents for detecting the expression of several common reporter genes in cells and cell extracts. These include substrates for β-galactosidase and β-glucuronidase (Detecting Glycosidases—Section 10.2), secreted alkaline phosphatase (SEAP, Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3), chloramphenicol acetyltransferase (CAT) and luciferase (Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6). Some of our EnzChek and DQ Kits (Detecting Peptidases and Proteases—Section 10.4) are useful for study of the uptake and metabolism of proteins during phagocytosis (Probes for Following Receptor Binding and Phagocytosis—Section 16.1). Substrates for specific proteases are also useful for the detection of apoptosis (Assays for Apoptosis—Section 15.5).
Thiol-Reactive Fluorogenic Substrates
We prepare a number of enzyme substrates for live-cell assays that incorporate a mildly thiol-reactive chloromethyl moiety. Once inside the cell, this chloromethyl group undergoes what is believed to be a glutathione S-transferase–mediated reaction to produce a membrane-impermeant, glutathione–fluorescent dye adduct, although our experiments suggest that they may also react with other intracellular components. Regardless of the mechanism, many cell types loaded with these chloromethylated substrates are both fluorescent and viable for at least 24 hours after loading and often through several cell divisions. Furthermore, unlike the free dye, the peptide–fluorescent dye adducts contain amino groups and can therefore be covalently linked to surrounding biomolecules by fixation with formaldehyde or glutaraldehyde. This property permits long-term storage of the labeled cells or tissue and, in cases where the anti-dye antibody is available (Anti-Dye and Anti-Hapten Antibodies—Section 7.4), amplification of the conjugate by standard immunochemical techniques, including tyramide signal amplification (TSA, TSA and Other Peroxidase-Based Signal Amplification Techniques—Section 6.2). Chloromethyl analogs of fluorogenic substrates for glycosidases (for example, in the DetectaGene Green CMFDG Kit, (D2920); Detecting Glycosidases—Section 10.2), peptidases, dealkylases, peroxidases and esterases are available. Our CellTracker Blue CMAC and CellTracker Blue CMF2HC dyes (C2110, C12881) are precursors to peptidase and glycosidase substrates, respectively, and are also used for long-term cell tracing (Membrane-Permeant Reactive Tracers—Section 14.2).
Lipophilic Fluorophores
Lipophilic analogs of fluorescein and resorufin exhibit many of the same properties as the water-soluble fluorophores, including relatively high extinction coefficients and good quantum yields. In most cases, however, substrates based on these lipophilic analogs load more readily into cells, permitting use of much lower substrate concentrations in the loading medium, and their fluorescent products are better retained after cleavage than their water-soluble counterparts. Lipophilic substrates and their products probably also distribute differently in cells and likely associate with lipid regions of the cell. When passive cell loading or enhanced dye retention are critical parameters of the experiment, we recommend using our lipophilic substrates for glycosidases (such as our ImaGene Green and ImaGene Red products, Detecting Glycosidases—Section 10.2). Like resazurin (R12204, Viability and Cytotoxicity Assay Reagents—Section 15.2), dodecylresazurin—the substrate in our LIVE/DEAD Cell Vitality Assay Kit, Vybrant Cell Metabolic Assay Kit and Metabolic Activity/Annexin V/Dead Cell Apoptosis Kit (L34951, V23110, V35114; Viability and Cytotoxicity Assay Kits for Diverse Cell Types—Section 15.3, Assays for Apoptosis—Section 15.5)—is reduced to dodecylresorufin by metabolically active cells; however, this lipophilic substrate is more useful than resazurin for microplate assays of all metabolic activity and permits single-cell analysis of cell metabolism by flow cytometry and cell counting. Dodecylresorufin is also the hydrolysis product of the β-galactosidase substrate used in our ImaGene Red C12RG lacZ Gene Expression Kit (I2906, Detecting Glycosidases—Section 10.2).
Alkaline phosphatase, β-galactosidase and horseradish peroxidase (HRP) conjugates are widely used as secondary detection reagents for immunohistochemical analysis and in situ hybridization, as well as for protein and nucleic acid detection by Southern, northern and western blots. In order to precisely localize enzymatic activity in a tissue, cell, gel or blot, the substrate must yield a product that immediately precipitates or reacts at the site of enzymatic activity. In addition, various methods such as chromatography, isoelectric focusing and gel electrophoresis are commonly employed to separate enzymes preceding their detection. A review by Weder and Kaiser discusses the use of a wide variety of fluorogenic substrates for the detection of electrophoretically separated hydrolases.![]()
Substrates Yielding Insoluble Fluorescent Products
In addition to the commonly used chromogenic substrates, including BCIP, NBT and X-gal (see below), we have developed a fluorogenic ELF 97 phosphatase substrate that fluoresces only weakly in the blue range. Upon enzymatic cleavage, however, this substrate forms the intensely yellow-green–fluorescent ELF 97 alcohol (E6578), which precipitates immediately at the site of enzymatic activity. The fluorescent ELF alcohol precipitate is exceptionally photostable (Figure 10.1.5) and has a high Stokes shift (Figure 10.1.6). We offer several ELF Kits based on the ELF 97 phosphatase substrate; see Phosphatase-Based Signal Amplification Techniques—Section 6.3 for a complete discussion of ELF technology.
Tyramide signal amplification (TSA) technology utilizes fluorescent or biotin-labeled tyramide substrates that are activated by HRP to phenoxyl radicals. These phenoxy radicals are then trapped by reaction with nearby tyrosine residues, resulting in localized tyramide deposition. We offer a wide assortment of TSA Kits based on this detection technology; see TSA and Other Peroxidase-Based Signal Amplification Techniques—Section 6.2) for a complete discussion of TSA technology.
 | Figure 10.1.5 Photostability comparison for ELF 97 alcohol– and fluorescein-labeled tubulin preparations. Tubulin in acetone-fixed CRE BAG 2 mouse fibroblasts was labeled with an anti–β-tubulin monoclonal antibody and then detected using biotin-XX goat anti–mouse IgG antibody (B2763) in conjunction with either our ELF 97 Cytological Labeling Kit (E6603, |
 | Figure 10.1.6 The normalized fluorescence excitation and emission spectra of the ELF 97 alcohol precipitate (E6578), which is generated by enzymatic cleavage of the soluble ELF 97 phosphatase substrate (E6588, E6589). |
Substrates Yielding Insoluble Chromophoric Products
A number of chromogenic substrates for hydrolytic enzymes are derived from indolyl chromophores. These initially form a colorless—and sometimes blue-fluorescent—3-hydroxyindole ("indoxyl"), which spontaneously, or through mediation of an oxidizing agent such as nitro blue tetrazolium (NBT, N6495; Detecting Enzymes That Metabolize Phosphates and Polyphosphates—Section 10.3) or potassium ferricyanide,![]() is converted to an intensely colored indigo dye that typically precipitates from the medium (Figure 10.1.7). Halogenated indolyl derivatives, including 5-bromo-4-chloro-3-indolyl β-D-galactopyranoside (X-Gal, B1690; Detecting Glycosidases—Section 10.2) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP, N6547) are generally preferred because they produce finer precipitates that are less likely to diffuse from the site of formation, making them especially useful for detecting enzymatic activity in cells, tissues, gels and blots.
is converted to an intensely colored indigo dye that typically precipitates from the medium (Figure 10.1.7). Halogenated indolyl derivatives, including 5-bromo-4-chloro-3-indolyl β-D-galactopyranoside (X-Gal, B1690; Detecting Glycosidases—Section 10.2) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP, N6547) are generally preferred because they produce finer precipitates that are less likely to diffuse from the site of formation, making them especially useful for detecting enzymatic activity in cells, tissues, gels and blots.
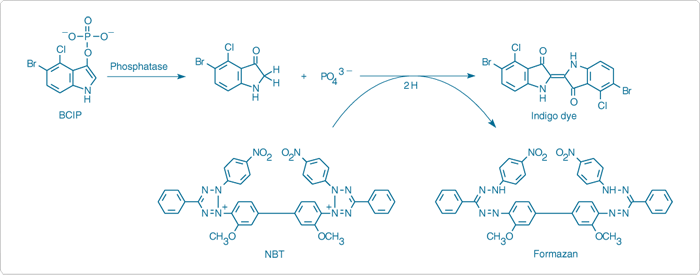
Figure 10.1.7 Principle of enzyme-linked detection using the reagents in our NBT/BCIP Reagent Kit (N6547). Phosphatase hydrolysis of BCIP is coupled to reduction of NBT, yielding a formazan and an indigo dye that together form a black-purple–colored precipitate.
The principle of excited-state energy transfer can also be used to generate fluorogenic substrates (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2). For example, the EDANS fluorophore in our HIV protease and renin substrates is effectively quenched by a nearby dabcyl acceptor chromophore (Figure 10.1.8). This chromophore has been carefully chosen for maximal overlap of its absorbance with the fluorophore's fluorescence, thus ensuring that the fluorescence is quenched through excited-state energy transfer. Proteolytic cleavage of the substrate results in spatial separation of the fluorophore and the acceptor chromophore, thereby restoring the fluorophore's fluorescence.![]()
Many of the dyes described in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1 have been used to form energy-transfer pairs, some of which can be introduced during automated synthesis of peptides using modified amino acids described in Peptide Analysis, Sequencing and Synthesis—Section 9.5. Molecular Probes nonfluorescent quenchers and photosensitizers—Table 1.10 (Long-Wavelength Rhodamines, Texas Red Dyes and QSY Quenchers—Section 1.6) lists our nonfluorescent quenching dyes and their spectral properties.
The protease substrates in three of our EnzChek Protease Assay Kits (Detecting Peptidases and Proteases—Section 10.4) are heavily labeled casein conjugates; the close proximity of dye molecules results in self-quenching. Hydrolysis of the protein to smaller fragments is accompanied by a dramatic increase in fluorescence, which forms the basis of a simple and sensitive continuous assay for a variety of proteases. In addition, we offer a phospholipase A substrate (bis-BODIPY FL C11-PC, B7701; Probes for Lipid Metabolism and Signaling—Section 17.4) that contains a BODIPY FL fluorophore on each phospholipid acyl chain. Proximity of the BODIPY FL fluorophores on adjacent phospholipid acyl chains causes fluorescence self-quenching that is relieved only when the fluorophores are separated by phospholipase A–mediated cleavage. PED6, a phospholipid with a green-fluorescent BODIPY fatty acid on the lipid portion of the molecule and a 2,4-dinitrophenyl quencher on the polar head group (PED6, D23739; Probes for Lipid Metabolism and Signaling—Section 17.4) is useful as a specific phospholipase-A2 substrate.![]()
 | Figure 10.1.8 Principle of the fluorogenic response to protease cleavage exhibited by HIV protease substrate 1 (H2930). Quenching of the EDANS fluorophore (F) by distance-dependent resonance energy transfer to the dabcyl quencher (Q) is eliminated upon cleavage of the intervening peptide linker. |
The mechanism of some enzymes makes it difficult to obtain a continuous optical change during reaction with an enzyme substrate. However, a discontinuous assay can often be developed by derivatizing the reaction products with one of the reagents described in Fluorophores and Their Amine-Reactive Derivatives—Chapter 1, Thiol-Reactive Probes—Chapter 2 and Click Chemistry and Other Functional Group Modifications—Chapter 3, usually followed by a separation step in order to generate a product-specific fluorescent signal.
For example, fluorescamine or o-phthaldialdehyde (OPA) (F2332, F20261, P2331MP; Reagents for Analysis of Low Molecular Weight Amines—Section 1.8) can detect the rate of any peptidase reaction by measuring the increase in the concentration of free amines in solution.![]() The activity of enzymes that produce free coenzyme A from its esters can be detected using thiol-reactive reagents such as 5,5'-dithiobis-(2-nitrobenzoic acid) (DTNB, D8451; Chemical Crosslinking Reagents—Section 5.2) or 7-fluorobenz-2-oxa-1,3-diazole-4-sulfonamide
The activity of enzymes that produce free coenzyme A from its esters can be detected using thiol-reactive reagents such as 5,5'-dithiobis-(2-nitrobenzoic acid) (DTNB, D8451; Chemical Crosslinking Reagents—Section 5.2) or 7-fluorobenz-2-oxa-1,3-diazole-4-sulfonamide ![]() (ABD-F, F6053; Thiol-Reactive Probes Excited with Visible Light—Section 2.2). The products of enzymes that metabolize low molecular weight substrates can frequently be detected by chromatographic or electrophoretic analysis. HPLC or capillary zone electrophoresis can also be used to enhance the sensitivity of reactions that yield fluorescent products.
(ABD-F, F6053; Thiol-Reactive Probes Excited with Visible Light—Section 2.2). The products of enzymes that metabolize low molecular weight substrates can frequently be detected by chromatographic or electrophoretic analysis. HPLC or capillary zone electrophoresis can also be used to enhance the sensitivity of reactions that yield fluorescent products.![]()
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Notes |
|---|---|---|---|---|---|---|---|---|
| A191 | 175.19 | L | DMF, DMSO | 351 | 18,000 | 430 | MeOH | 1 |
| C183 | 187.15 | L | pH >8, DMF | 408 | 43,000 | 450 | pH 9 | |
| C2110 | 209.63 | F,D,L | DMSO | 353 | 14,000 | 466 | pH 9 | 2 |
| C12881 | 246.60 | F,D,L | DMSO | 371 | 16,000 | 464 | pH 9 | 3 |
| D6566 | 212.15 | L | DMSO, DMF | 358 | 18,000 | 452 | pH 9 | 3, 4 |
| E6578 | 307.14 | L | DMSO | 345 | ND | 530 | pH 8 | 5, 6, 7 |
| F1300 | 332.31 | L | pH >6, DMF | 490 | 93,000 | 514 | pH 9 | 8 |
| F36915 | 332.31 | RO,L | see Notes | 490 | 93,000 | 514 | pH 9.5 | 8, 9 |
| H189 | 176.17 | L | DMSO, MeOH | 360 | 19,000 | 449 | pH 9 | 3 |
| H6482 | 308.16 | L | DMF | 646 | 41,000 | 659 | pH 10 | |
| R363 | 235.17 | L | pH >7, DMF | 571 | 62,000 | 585 | pH 9 | 10, 11, 12 |
| R6479 | 366.80 | L | DMSO | 499 | 92,000 | 521 | MeOH | 13 |
| ||||||||
For Research Use Only. Not for use in diagnostic procedures.