Search
Page Contents
- Fluorescent Fatty Acid Analogs
- ADIFAB Fatty Acid Indicator
- Phospholipids with BODIPY Dye–Labeled Acyl Chains
- Phospholipid with DPH-Labeled Acyl Chain
- Phospholipids with NBD-Labeled Acyl Chains
- Phospholipids with Pyrene-Labeled Acyl Chains
- Phospholipids with a Fluorescent or Biotinylated Head Group
- LipidTOX Phospholipid and Neutral Lipid Stains for High-Content Screening
- Data Table
- Ordering Information
Related TablesRelated Technical Notes
Get Chapter Downloads from The Molecular Probes Handbook, 11th edition |
The probes in this section and in Sphingolipids, Steroids, Lipopolysaccharides and Related Probes—Section 13.3 bear some structural resemblance to natural lipids. Included in this section are fluorescent fatty acid analogs, as well as phospholipids wherein one or both fatty acid esters are replaced by fluorescent fatty acid esters. The fluorophores in these probes tend to remain buried in the hydrophobic interior of lipid bilayer membranes.![]() In this location, they are sensitive to membrane properties such as lipid fluidity, lateral domain formation and structural perturbation by proteins, drugs and other additives. Also included in this section are several head group–modified phospholipid analogs incorporating a fluorophore or biotin (Phospholipids with labeled head groups—Table 13.1).
In this location, they are sensitive to membrane properties such as lipid fluidity, lateral domain formation and structural perturbation by proteins, drugs and other additives. Also included in this section are several head group–modified phospholipid analogs incorporating a fluorophore or biotin (Phospholipids with labeled head groups—Table 13.1).
Sphingolipids, steroids and lipopolysaccharides are discussed in Sphingolipids, Steroids, Lipopolysaccharides and Related Probes—Section 13.3. Important applications of the fluorescent phosphatidylinositol derivatives as probes for signal transduction and various fluorescent phospholipids as phospholipase substrates are further described in Probes for Lipid Metabolism and Signaling—Section 17.4. A review of fluorescent lipid probes and their use in biological and biophysical research has been published.![]()
Our fluorescent fatty acid analogs have a fluorophore linked within the fatty acid chain or, more commonly, at the terminal (omega) carbon atom that is furthest from the carboxylate moiety. Although fluorescent fatty acid analogs are sometimes used as direct probes for membranes and liposomes, their most common applications have been for synthesis of fluorescent phospholipids and for metabolic incorporation by live cells. Our fluorescent fatty acids currently include derivatives based on the BODIPY, nitrobenzoxadiazole (NBD), pyrene and dansyl fluorophores, as well as the naturally fluorescent polyunsaturated fatty acid, cis-parinaric acid.
BODIPY Fatty Acids
BODIPY fatty acids are, by far, the most fluorescent fatty acid analogs that we have available.![]() The lack of ionic charge on the BODIPY fluorophore is unusual for long-wavelength fluorescent dyes and results in exclusive localization of the fluorophore within the membrane (Figure 13.2.1F). BODIPY derivatives typically have extinction coefficients greater than 90,000 cm-1M-1 with absorption maxima beyond 500 nm. A useful spectroscopic property of BODIPY dyes is the concentration-dependent formation of excited-state dimers ("excimers") with red-shifted emission. We have observed this phenomenon particularly with our green-fluorescent BODIPY fatty acid derivatives, which undergo a considerable red shift in their emission when metabolically incorporated into lipophilic products
The lack of ionic charge on the BODIPY fluorophore is unusual for long-wavelength fluorescent dyes and results in exclusive localization of the fluorophore within the membrane (Figure 13.2.1F). BODIPY derivatives typically have extinction coefficients greater than 90,000 cm-1M-1 with absorption maxima beyond 500 nm. A useful spectroscopic property of BODIPY dyes is the concentration-dependent formation of excited-state dimers ("excimers") with red-shifted emission. We have observed this phenomenon particularly with our green-fluorescent BODIPY fatty acid derivatives, which undergo a considerable red shift in their emission when metabolically incorporated into lipophilic products ![]() (
(![]() ). Pyrene fatty acids (see below) also exhibit excimer formation but their emission is at much shorter wavelengths than that of the BODIPY dyes and they are therefore less suitable for the study of live cells. The fluorophores in our current selection of BODIPY fatty acids and their approximate absorption/emission maxima (in nm) are:
). Pyrene fatty acids (see below) also exhibit excimer formation but their emission is at much shorter wavelengths than that of the BODIPY dyes and they are therefore less suitable for the study of live cells. The fluorophores in our current selection of BODIPY fatty acids and their approximate absorption/emission maxima (in nm) are:
- BODIPY 503/512 (BODIPY FL; D3821, D3822, D3834)
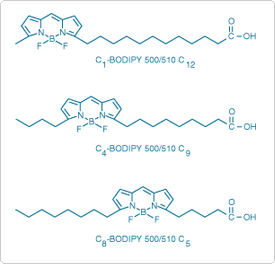
- BODIPY 500/510 (D3823, Figure 13.2.2)
- BODIPY 558/568 (D3835)
- BODIPY 581/591 (D3861)
BODIPY fatty acids are synthetic precursors to a wide variety of fluorescent phospholipids (described below), as well as several important sphingolipid probes described in Sphingolipids, Steroids, Lipopolysaccharides and Related Probes—Section 13.3. Some BODIPY fatty acids are readily metabolized by live cells to phospholipids, di- and triacylglycerols, cholesteryl esters and other natural lipids.![]() Analysis of cellular lipid extracts by HPLC has shown that glycerophosphocholines constitute more than 90% of the products of biosynthetic incorporation of BODIPY 500/510 dodecanoic acid (D3823) by BHK cells.
Analysis of cellular lipid extracts by HPLC has shown that glycerophosphocholines constitute more than 90% of the products of biosynthetic incorporation of BODIPY 500/510 dodecanoic acid (D3823) by BHK cells.![]()
The three BODIPY 500/510 probes form a unique series in which the green-fluorescent fluorophore is located within the fatty acid chain at different distances from the terminal carboxylate group.![]() The overall length of the probe is constant and, including the fluorophore, is about equivalent to that of an 18-carbon fatty acid (Figure 13.2.2).
The overall length of the probe is constant and, including the fluorophore, is about equivalent to that of an 18-carbon fatty acid (Figure 13.2.2).
BODIPY 581/591 undecanoic acid (D3861) is particularly useful for detecting reactive oxygen species in cells and membranes.![]() Oxidation of the polyunsaturated butadienyl portion of the BODIPY 581/591 dye truncates the conjugated π-electron system, resulting in a shift of the fluorescence emission peak from ~590 nm to ~510 nm.
Oxidation of the polyunsaturated butadienyl portion of the BODIPY 581/591 dye truncates the conjugated π-electron system, resulting in a shift of the fluorescence emission peak from ~590 nm to ~510 nm.![]() This oxidation response mechanism is similar to that of the naturally occurring polyunsaturated fatty acid cis-parinaric acid (see below). In comparison to cis-parinaric acid, advantages of BODIPY 581/591 undecanoic acid include:
This oxidation response mechanism is similar to that of the naturally occurring polyunsaturated fatty acid cis-parinaric acid (see below). In comparison to cis-parinaric acid, advantages of BODIPY 581/591 undecanoic acid include:
- Long-wavelength excitation, compatible with confocal laser-scanning microscopes and flow cytometers
- Avoidance of photooxidation effects induced by ultraviolet excitation
- Less interference by colored oxidant and antioxidant additives when detecting probe fluorescence

- Greater resistance to spontaneous oxidation
- Red-to-green fluorescence shift, allowing the use of fluorescence ratio detection methods

An alternative technique for detecting lipid peroxidation utilizes the oxidation-induced decrease of concentration-dependent excimer formation by BODIPY FL dye–labeled fatty acids.![]()
NBD Fatty Acids
Fluorescence of the nitrobenzoxadiazole (NBD) fluorophore is highly sensitive to its environment. Although it is moderately fluorescent in aprotic solvents, in aqueous solvents it is almost nonfluorescent.![]() The NBD fluorophore is moderately polar and both its homologous 6-carbon and 12-carbon fatty acid analogs and the phospholipids derived from these probes tend to sense the lipid–water interface region of membranes instead of the hydrophobic interior
The NBD fluorophore is moderately polar and both its homologous 6-carbon and 12-carbon fatty acid analogs and the phospholipids derived from these probes tend to sense the lipid–water interface region of membranes instead of the hydrophobic interior ![]() (Figure 13.2.1B). The environmental sensitivity of NBD fatty acids can be usefully exploited to probe the ligand-binding sites of fatty acid and sterol carrier proteins.
(Figure 13.2.1B). The environmental sensitivity of NBD fatty acids can be usefully exploited to probe the ligand-binding sites of fatty acid and sterol carrier proteins.![]() NBD fatty acids are not well metabolized by live cells.
NBD fatty acids are not well metabolized by live cells.![]()
Pyrene Fatty Acids
The hydrophobic pyrene fluorophore is readily accommodated within the membrane.![]() ω-Pyrene derivatives of longer-chain fatty acids were first described by Galla and Sackmann in 1975.
ω-Pyrene derivatives of longer-chain fatty acids were first described by Galla and Sackmann in 1975.![]() Pyrene derivatives of the 4-, 10-, 12- and 16-carbon fatty acids have been synthesized. Pyrenebutanoic acid—frequently called pyrenebutyric acid—has rarely been used as a membrane probe; however, its conjugates have exceptionally long excited-state lifetimes (τ >100 nanoseconds) and are consequently useful for time-resolved fluorescence immunoassays and nucleic acid detection.
Pyrene derivatives of the 4-, 10-, 12- and 16-carbon fatty acids have been synthesized. Pyrenebutanoic acid—frequently called pyrenebutyric acid—has rarely been used as a membrane probe; however, its conjugates have exceptionally long excited-state lifetimes (τ >100 nanoseconds) and are consequently useful for time-resolved fluorescence immunoassays and nucleic acid detection.![]() The long excited-state lifetime of pyrenebutyric acid also makes it useful as a probe for oxygen in cells
The long excited-state lifetime of pyrenebutyric acid also makes it useful as a probe for oxygen in cells ![]() and lipid vesicles.
and lipid vesicles.![]()
Pyrene derivatives form excited-state dimers (excimers) with red-shifted fluorescence emission ![]() (Figure 13.2.3). Pyrene excimers can even form when two pyrenes are tethered by a short trimethine spacer, as in 1,3-bis-(1-pyrenyl)propane (Other Nonpolar and Amphiphilic Probes—Section 13.5). Pyrene excimer formation is commonly exploited for assaying membrane fusion
(Figure 13.2.3). Pyrene excimers can even form when two pyrenes are tethered by a short trimethine spacer, as in 1,3-bis-(1-pyrenyl)propane (Other Nonpolar and Amphiphilic Probes—Section 13.5). Pyrene excimer formation is commonly exploited for assaying membrane fusion ![]() (Lipid-Mixing Assays of Membrane Fusion—Note 13.1) and for detecting lipid domain formation.
(Lipid-Mixing Assays of Membrane Fusion—Note 13.1) and for detecting lipid domain formation.![]() Pyrene fatty acids are metabolically incorporated into phospholipids, di- and tri-acylglycerols and cholesteryl esters by live cells.
Pyrene fatty acids are metabolically incorporated into phospholipids, di- and tri-acylglycerols and cholesteryl esters by live cells.![]() Other uses of pyrene fatty acids include:
Other uses of pyrene fatty acids include:
- Detecting lipid–protein interactions

- Inducing photodynamic damage

- Investigating phospholipase A2 action on lipid assemblies

- Studying lipid transport mechanisms and transfer proteins

- Synthesizing fluorescent sphingolipid probes

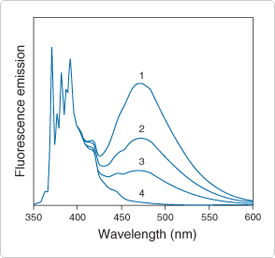
Figure 13.2.3 Excimer formation by pyrene in ethanol. Spectra are normalized to the 371.5 nm peak of the monomer. All spectra are essentially identical below 400 nm after normalization. Spectra are as follows: 1) 2 mM pyrene, purged with argon to remove oxygen; 2) 2 mM pyrene, air-equilibrated; 3) 0.5 mM pyrene (argon-purged); and 4) 2 µM pyrene (argon-purged). The monomer-to-excimer ratio (371.5 nm/470 nm) is dependent on both pyrene concentration and the excited-state lifetime, which is variable because of quenching by oxygen.
Dansyl Undecanoic Acid
Dansyl undecanoic acid (DAUDA) incorporates a polar, environment-sensitive dansyl fluorophore that preferentially locates in the polar headgroup region of lipid bilayer membranes.![]() DAUDA exhibits a 60-fold fluorescence enhancement and a large emission spectral shift to shorter wavelengths on binding to certain proteins.
DAUDA exhibits a 60-fold fluorescence enhancement and a large emission spectral shift to shorter wavelengths on binding to certain proteins.![]() This property has been exploited to analyze fatty acid–binding proteins
This property has been exploited to analyze fatty acid–binding proteins ![]() and also to develop a fluorometric phospholipase A2 assay (Probes for Lipid Metabolism and Signaling—Section 17.4) based on competitive fatty acid displacement.
and also to develop a fluorometric phospholipase A2 assay (Probes for Lipid Metabolism and Signaling—Section 17.4) based on competitive fatty acid displacement.![]()
cis-Parinaric Acid
The naturally occurring polyunsaturated fatty acid cis-parinaric acid was initially developed as a membrane probe by Hudson and co-workers and published in 1975.![]() cis-Parinaric acid is the closest structural analog of intrinsic membrane lipids among currently available fluorescent probes (Figure 13.2.1E). The chemical and physical properties of cis-parinaric acid have been well characterized. The lowest absorption band of cis-parinaric acid has two main peaks around 300 nm and 320 nm, with a high extinction coefficient. cis-Parinaric acid offers several experimentally advantageous optical properties, including a very large fluorescence Stokes shift (~100 nm) and an almost complete lack of fluorescence in water. In addition, the fluorescence decay lifetime of cis-parinaric acid varies from 1 to ~40 nanoseconds, depending on the molecular packing density in phospholipid bilayers. Consequently, minutely detailed information on lipid-bilayer dynamics can be obtained.
cis-Parinaric acid is the closest structural analog of intrinsic membrane lipids among currently available fluorescent probes (Figure 13.2.1E). The chemical and physical properties of cis-parinaric acid have been well characterized. The lowest absorption band of cis-parinaric acid has two main peaks around 300 nm and 320 nm, with a high extinction coefficient. cis-Parinaric acid offers several experimentally advantageous optical properties, including a very large fluorescence Stokes shift (~100 nm) and an almost complete lack of fluorescence in water. In addition, the fluorescence decay lifetime of cis-parinaric acid varies from 1 to ~40 nanoseconds, depending on the molecular packing density in phospholipid bilayers. Consequently, minutely detailed information on lipid-bilayer dynamics can be obtained.
Selected applications of cis-parinaric acid include:
- Measurement of peroxidation in lipoproteins
 and the relationship of peroxidation to cytotoxicity
and the relationship of peroxidation to cytotoxicity  and apoptosis
and apoptosis 
- Evaluation of antioxidants

- Detection of lipoproteins following chromatographic separation
 and structural characterization of lipoproteins
and structural characterization of lipoproteins 
- Detection of lipid–protein interactions
 and lipid clustering
and lipid clustering 
- High-affinity binding to a hydrophobic pocket between the heavy chain of myosin subfragment-1 and its essential light chain

- Investigation of the mechanism of fatty acid–binding proteins
 and phospholipid-transfer proteins
and phospholipid-transfer proteins
The extensive unsaturation of cis-parinaric acid makes it quite susceptible to oxidation. Consequently, a3 mM cis-parinaric acid solution in deoxygenated ethanol should be stable for at least six months if stored protected from light under an inert argon atmosphere at -20°C. During experiments, we strongly advise handling cis-parinaric acid samples under inert gas and preparing solutions using degassed buffers and solvents. cis-Parinaric acid is also somewhat photolabile, undergoing photodimerization under intense illumination, resulting in loss of fluorescence.![]()
Fatty acid–binding proteins are small cytosolic proteins found in a variety of mammalian tissues, and studies of their physiological function frequently involve fluorescent fatty acid probes.![]() ADIFAB, a dual-wavelength fluorescent indicator of free fatty acids, facilitates these studies
ADIFAB, a dual-wavelength fluorescent indicator of free fatty acids, facilitates these studies ![]() (Figure 13.2.4, Figure 13.2.5; contact Custom Services for more information). ADIFAB is a conjugate of I-FABP, a rat intestinal fatty acid–binding protein with a low molecular weight (15,000 daltons) and a high binding affinity for free fatty acids,
(Figure 13.2.4, Figure 13.2.5; contact Custom Services for more information). ADIFAB is a conjugate of I-FABP, a rat intestinal fatty acid–binding protein with a low molecular weight (15,000 daltons) and a high binding affinity for free fatty acids, ![]() and the polarity-sensitive acrylodan fluorophore (A433, Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3). It is designed to provide quantitative monitoring of free fatty acids without resorting to separative biochemical methods.
and the polarity-sensitive acrylodan fluorophore (A433, Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3). It is designed to provide quantitative monitoring of free fatty acids without resorting to separative biochemical methods.![]() With appropriate precautions, which are described in the product information sheet accompanying this product, ADIFAB can be used to determine free fatty acid concentrations between 1 nM and >20 µM.
With appropriate precautions, which are described in the product information sheet accompanying this product, ADIFAB can be used to determine free fatty acid concentrations between 1 nM and >20 µM.
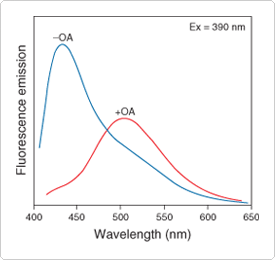
Figure 13.2.5 The free fatty acid–dependent spectral shift of ADIFAB. Spectra shown represent 0.2 µM ADIFAB in pH 8.0 buffer with (+OA) and without (–OA) addition of 4.7 µM cis-9-octadecenoic (oleic) acid (OA). The ratio of fluorescence emission intensities at 505 nm and 432 nm can be quantitatively related to free fatty acid concentrations.
BODIPY Glycerophospholipids
We offer a glycerophospholipid analog labeled with a single green-fluorescent BODIPY FL fluorophore on the sn-2 acyl chain: BODIPY FL dye–labeled glycerophosphocholine (D3792). In addition, we prepare a glycerophosphocholine analog with a single nonhydrolyzable ether-linked BODIPY FL fluorophore on the sn-1 position (contact Custom Services for more information), as well as several doubly labeled glycerophospholipids. These doubly labeled glycerophospholipids, which are discussed in greater detail in Probes for Lipid Metabolism and Signaling—Section 17.4, are designed primarily for detection of phospholipase A1 and phospholipase A2 and include:
- Glycerophosphoethanolamine with a BODIPY FL dye–labeled sn-1 acyl chain and a dinitrophenyl quencher–modified headgroup (PED-A1, A10070)
- Glycerophosphoethanolamine with a BODIPY FL dye–labeled sn-2 acyl chain and a dinitrophenyl quencher–modified headgroup
 (PED6, D23739; Figure 13.2.9)
(PED6, D23739; Figure 13.2.9) - Glycerophosphocholine with two BODIPY FL dye–labeled acyl chains (bis-BODIPY FL C11-PC, B7701; Figure 13.2.9)
- Glycerophosphocholine with a BODIPY 558/568 dye–labeled sn-1 alkyl chain and a BODIPY FL dye–labeled sn-2 acyl chain (Red/Green BODIPY PC-A2, A10072)
The spectral properties of BODIPY FL dye–labeled phospholipids are summarized in Spectral properties of some lipid probes—Table 13.2. Unlike the nitrobenzoxadiazole (NBD) fluorophore, the BODIPY FL and BODIPY 500/510 fluorophores are intrinsically lipophilic and readily localize in the membrane's interior.![]() The fluorophore is completely inaccessible to the membrane-impermeant anti–BODIPY FL antibody (A5770, Anti-Dye and Anti-Hapten Antibodies—Section 7.4), which also recognizes the BODIPY 500/510 derivative. As shown in Figure 13.2.6, the emission spectrum of the BODIPY 500/510 fluorophore is much narrower than that of the NBD fluorophore. Because both the extinction coefficient of the BODIPY 500/510 fluorophore and its quantum yield in a lipophilic environment (EC ~90,000 cm-1M-1 and QY ~0.9) are much higher than those of the NBD fluorophore (EC ~20,000 cm-1M-1 and QY ~0.3), much less BODIPY 500/510 dye–labeled phospholipid is required for labeling membranes.
The fluorophore is completely inaccessible to the membrane-impermeant anti–BODIPY FL antibody (A5770, Anti-Dye and Anti-Hapten Antibodies—Section 7.4), which also recognizes the BODIPY 500/510 derivative. As shown in Figure 13.2.6, the emission spectrum of the BODIPY 500/510 fluorophore is much narrower than that of the NBD fluorophore. Because both the extinction coefficient of the BODIPY 500/510 fluorophore and its quantum yield in a lipophilic environment (EC ~90,000 cm-1M-1 and QY ~0.9) are much higher than those of the NBD fluorophore (EC ~20,000 cm-1M-1 and QY ~0.3), much less BODIPY 500/510 dye–labeled phospholipid is required for labeling membranes.![]()
Incorporation of high molar ratios (>10 mole %) of the BODIPY 500/510 dye–labeled phospholipids into membranes results in a dramatic spectral shift of the fluorescence emission spectrum to longer wavelengths (Figure 13.2.7). We have also observed this spectral shift in the Golgi of live cells that have been labeled with our BODIPY dye–labeled ceramides (Probes for the Endoplasmic Reticulum and Golgi Apparatus—Section 12.4) and with BODIPY fatty acids that have been metabolically incorporated by cells (![]() ). In fluorescence resonance energy transfer (FRET) measurements, the green-fluorescent BODIPY 500/510 dye is an excellent donor to longer-wavelength BODIPY probes
). In fluorescence resonance energy transfer (FRET) measurements, the green-fluorescent BODIPY 500/510 dye is an excellent donor to longer-wavelength BODIPY probes ![]() (Figure 13.2.8) and acceptor from dansyl-, DPH- or pyrene-labeled phospholipids.
(Figure 13.2.8) and acceptor from dansyl-, DPH- or pyrene-labeled phospholipids.![]() These probe combinations offer several alternatives to the widely used NBD–rhodamine fluorophore pair for researchers using FRET techniques to study lipid transfer and membrane fusion.
These probe combinations offer several alternatives to the widely used NBD–rhodamine fluorophore pair for researchers using FRET techniques to study lipid transfer and membrane fusion.![]()

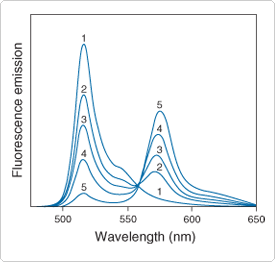
Figure 13.2.8 Fluorescence resonance energy transfer from β-BODIPY 500/510 C12-HPC (peak at 516 nm) to BODIPY 558/568 C12 (peak at 572 nm, D3835) in DOPC (dioctadecenoylglycerophosphocholine) lipid bilayers using 475 nm excitation. Ratio of acceptors to donors is: 1) 0; 2) 0.2; 3) 0.4; 4) 0.8; and 5) 2.0.
Applications
Once cells are labeled with a BODIPY phospholipid, the probe shows little tendency to spontaneously transfer between cells.![]() Consequently, BODIPY dye–labeled phospholipids have been used in a number of studies of cell membrane structure and properties:
Consequently, BODIPY dye–labeled phospholipids have been used in a number of studies of cell membrane structure and properties:
- Despite their good photostability, BODIPY lipids are useful for fluorescence recovery after photobleaching (FRAP) measurements of lipid diffusion.

- Researchers have used BODIPY fatty acids and phospholipids to visualize compartmentalization of specific lipid classes in Schistosoma mansoni
 and fungi.
and fungi.
- β-BODIPY FL C12-HPC (D3792) has been used to examine lipid–protein interactions involved in bacterial protein secretion via fluorescence resonance energy transfer (FRET) measurements
 (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2).
(Fluorescence Resonance Energy Transfer (FRET)—Note 1.2). - β-BODIPY FL C5-HPC
 has been used to characterize lipid domains by fluorescence correlation spectroscopy
has been used to characterize lipid domains by fluorescence correlation spectroscopy  (Fluorescence Correlation Spectroscopy (FCS)—Note 1.3), confocal laser-scanning microscopy
(Fluorescence Correlation Spectroscopy (FCS)—Note 1.3), confocal laser-scanning microscopy  (
( ) and near-field scanning optical microscopy.
) and near-field scanning optical microscopy.
- β-BODIPY FL C5-HPC has been used to investigate the cellular uptake of antineoplastic ether lipids.

- bis-BODIPY FL C11-PC (B7701) has BODIPY FL dye–labeled sn-1 and sn-2 acyl groups, resulting in partially quenched fluorescence that increases when one of the acyl groups is hydrolyzed by phospholipase A1 or A2. The hydrolysis products are BODIPY FL undecanoic acid and BODIPY FL dye–labeled lysophosphatidylcholine (Figure 13.2.9). The probe has been used successfully in human neutrophils, plants and zebrafish to detect phospholipase A activity
 (Probes for Lipid Metabolism and Signaling—Section 17.4).
(Probes for Lipid Metabolism and Signaling—Section 17.4).
Properties
The fluorescent phospholipid analog β-DPH HPC comprises diphenylhexatriene propionic acid coupled to glycerophosphocholine at the sn-2 position. It is therefore related to the neutral membrane probe DPH and the cationic derivative TMA-DPH (T204, Other Nonpolar and Amphiphilic Probes—Section 13.5). DPH and its derivatives exhibit strong fluorescence enhancement when incorporated into membranes, as well as sensitive fluorescence polarization (anisotropy) responses to lipid ordering (Fluorescence Polarization (FP)—Note 1.4). β-DPH HPC was originally devised to improve the localization of DPH in membranes.![]() Unlike underivatized DPH, it can be used to specifically label one leaflet of a lipid bilayer, facilitating analysis of membrane asymmetry.
Unlike underivatized DPH, it can be used to specifically label one leaflet of a lipid bilayer, facilitating analysis of membrane asymmetry.![]()
Applications
DPH derivatives are predominately used to investigate the structure and dynamics of the membrane interior either by fluorescence polarization or lifetime measurements. Researchers have used β-DPH HPC as a probe for lipid–protein interactions,![]() alcohol-induced perturbations of membrane structure,
alcohol-induced perturbations of membrane structure,![]() molecular organization and dynamics of lipid bilayers
molecular organization and dynamics of lipid bilayers ![]() and lipid peroxidation.
and lipid peroxidation.![]() Fluorescence lifetime measurements of β-DPH HPC provide a sensitive indicator of membrane fusion.
Fluorescence lifetime measurements of β-DPH HPC provide a sensitive indicator of membrane fusion.![]() In addition to membrane fusion, β-DPH HPC has been used to monitor various other lipid-transfer processes.
In addition to membrane fusion, β-DPH HPC has been used to monitor various other lipid-transfer processes.![]()
Properties
Spectral properties of some lipid probes—Table 13.2 compares the spectral properties of acyl-modified nitrobenzoxadiazole (NBD) phospholipid probes NBD C6-HPC and NBC C12-HPC with those of the BODIPY, DPH and pyrene lipid probes. Unlike the BODIPY phospholipids, the location of the relatively polar NBD fluorophore of NBD C12-HPC in phospholipid bilayers does not appear to conform to expectations based on the probe structure. A variety of physical evidence indicates that the NBD moiety "loops back" to the head-group region ![]() (Figure 13.2.1B). In fact, the fluorophore in this acyl-modified phospholipid appears to probe the same location as does the head group–labeled glycerophosphoethanolamine derivative NBD-PE
(Figure 13.2.1B). In fact, the fluorophore in this acyl-modified phospholipid appears to probe the same location as does the head group–labeled glycerophosphoethanolamine derivative NBD-PE ![]() (N360, see below).
(N360, see below).
These NBD probes transfer spontaneously between membranes, with NBD C6-HPC transferring more rapidly than its more lipophilic C12 analog.![]() NBD C6-HPC can be readily removed (back-exchanged) from the plasma membrane by incubating the labeled cells either with unlabeled lipid vesicles
NBD C6-HPC can be readily removed (back-exchanged) from the plasma membrane by incubating the labeled cells either with unlabeled lipid vesicles ![]() or with bovine serum albumin.
or with bovine serum albumin.![]() This property is useful for quantitating lipid transfer and for studying phospholipid distribution asymmetry and transmembrane "flip-flop" rates in lipid bilayers.
This property is useful for quantitating lipid transfer and for studying phospholipid distribution asymmetry and transmembrane "flip-flop" rates in lipid bilayers.![]()
Applications
NBD acyl–modified probes are used for investigating lipid traffic, either by directly visualizing NBD fluorescence,![]() by exploiting NBD self-quenching
by exploiting NBD self-quenching ![]() or by fluorescence resonance energy transfer methods.
or by fluorescence resonance energy transfer methods.![]() Lateral domains in model monolayers, bilayers and cell membranes have been characterized using NBD phospholipids in conjunction with fluorescence recovery after photobleaching
Lateral domains in model monolayers, bilayers and cell membranes have been characterized using NBD phospholipids in conjunction with fluorescence recovery after photobleaching ![]() (FRAP), fluorescence resonance energy transfer
(FRAP), fluorescence resonance energy transfer ![]() (FRET) (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2) and direct microscopy techniques.
(FRET) (Fluorescence Resonance Energy Transfer (FRET)—Note 1.2) and direct microscopy techniques.![]() Transmembrane lipid distribution (Lipid-Mixing Assays of Membrane Fusion—Note 13.1) has been assessed using fluorescence resonance energy transfer from NBD HPC to rhodamine DHPE
Transmembrane lipid distribution (Lipid-Mixing Assays of Membrane Fusion—Note 13.1) has been assessed using fluorescence resonance energy transfer from NBD HPC to rhodamine DHPE ![]() (L1392) or alternatively by selective dithionite (S2O42–) reduction of NBD phospholipids in the outer membrane monolayer
(L1392) or alternatively by selective dithionite (S2O42–) reduction of NBD phospholipids in the outer membrane monolayer ![]() (Figure 13.2.10).
(Figure 13.2.10).

Properties
Phospholipid analogs with pyrene-labeled sn-2 acyl chains are among the most popular fluorescent membrane probes.![]() The spectral properties of the pyrene lipid probes are summarized in Spectral properties of some lipid probes—Table 13.2. Of primary importance in terms of practical applications is the concentration-dependent formation of excited-state pyrene dimers (excimers), which exhibit a distinctive red-shifted emission (peak ~470 nm) (Figure 13.2.3).
The spectral properties of the pyrene lipid probes are summarized in Spectral properties of some lipid probes—Table 13.2. Of primary importance in terms of practical applications is the concentration-dependent formation of excited-state pyrene dimers (excimers), which exhibit a distinctive red-shifted emission (peak ~470 nm) (Figure 13.2.3).
Applications
The excimer-forming properties of pyrene are well suited for monitoring membrane fusion (Lipid-Mixing Assays of Membrane Fusion—Note 13.1) and phospholipid transfer processes.![]() The monomer/excimer emission ratio can also be used to characterize membrane structural domains and their dependence on temperature, lipid composition and other external factors.
The monomer/excimer emission ratio can also be used to characterize membrane structural domains and their dependence on temperature, lipid composition and other external factors.![]() Pyrenedecanoyl glycerophosphocholine (β-py-C10-HPC) has been used to elucidate the effect of extrinsic species such as Ca2+,
Pyrenedecanoyl glycerophosphocholine (β-py-C10-HPC) has been used to elucidate the effect of extrinsic species such as Ca2+,![]() platelet-activating factor,
platelet-activating factor,![]() drugs,
drugs,![]() membrane-associated proteins
membrane-associated proteins ![]() and ethanol
and ethanol ![]() on lipid bilayer structure and dynamics. The anionic phosphoglycerol analog is preferred as a substrate for secretory phospholipases A2 relative to other phospholipid classes.
on lipid bilayer structure and dynamics. The anionic phosphoglycerol analog is preferred as a substrate for secretory phospholipases A2 relative to other phospholipid classes.![]() The long excited-state lifetime of pyrene (Spectral properties of some lipid probes—Table 13.2) renders the fluorescence of its conjugates very susceptible to oxygen quenching, and consequently these probes can be used to measure oxygen concentrations in solutions,
The long excited-state lifetime of pyrene (Spectral properties of some lipid probes—Table 13.2) renders the fluorescence of its conjugates very susceptible to oxygen quenching, and consequently these probes can be used to measure oxygen concentrations in solutions,![]() lipid bilayers
lipid bilayers ![]() and cells.
and cells.![]()
Glycerophospholipids in which both alcohols are esterified to pyrene fatty acids (Figure 13.2.1C), as in bis-(1-pyrenebutanoyl)glycerophosphocholine and bis-(1-pyrenedecanoyl)glycerophosphocholine, show strong excimer fluorescence, with maximum emission near 470 nm.![]() Hydrolysis of either fatty acid ester by a phospholipase results in liberation of a pyrene fatty acid and an emission shift to shorter wavelengths, making these probes useful as phospholipase substrates
Hydrolysis of either fatty acid ester by a phospholipase results in liberation of a pyrene fatty acid and an emission shift to shorter wavelengths, making these probes useful as phospholipase substrates ![]() (Probes for Lipid Metabolism and Signaling—Section 17.4).
(Probes for Lipid Metabolism and Signaling—Section 17.4).
Phospholipid with a Dansyl-Labeled Head Group
The phospholipid analog incorporating the environment-sensitive ![]() dansyl fluorophore (dansyl DHPE) is a useful probe of lipid–water interfaces.
dansyl fluorophore (dansyl DHPE) is a useful probe of lipid–water interfaces.![]() It is sensitive to the interactions of a number of proteins, including protein kinase C,
It is sensitive to the interactions of a number of proteins, including protein kinase C,![]() annexins
annexins ![]() and phospholipase A2,
and phospholipase A2,![]() with membrane surfaces. Dansyl DHPE has also been used to examine the effects of cholesterol on the accessibility of the dansyl hapten to antibodies
with membrane surfaces. Dansyl DHPE has also been used to examine the effects of cholesterol on the accessibility of the dansyl hapten to antibodies ![]() (Antibodies for Detecting Membrane-Surface Labels—Note 13.2).
(Antibodies for Detecting Membrane-Surface Labels—Note 13.2).
Phospholipid with a Marina Blue Dye–Labeled Head Group
Marina Blue DHPE (M12652) is optimally excited by the intense 365 nm spectral line of the mercury-arc lamp and exhibits bright blue fluorescence emission near 460 nm. Significantly, the pKa value of this 6,8-difluoro-7-hydroxycoumarin derivative is 2–3 log units lower than that of nonfluorinated 7-hydroxycoumarin analogs; consequently, Marina Blue DHPE is expected to be strongly fluorescent in membranes, even at neutral pH.
Phospholipid with a Pacific Blue Dye–Labeled Head Group
The Pacific Blue dye–labeled phospholipid (Pacific Blue DMPE) is our only head group–labeled phospholipid with tetradecanoyl (myristoyl) esters rather than hexadecanoyl (palmitoyl) esters. This blue-fluorescent phospholipid is structurally similar to a phospholipid described by Gonzalez and Tsien for use in a FRET-based measurement of membrane potential.![]()
Phospholipid with an NBD-Labeled Head Group
The widely used membrane probe nitrobenzoxadiazolyldihexadecanoylglycerophosphoethanolamine ![]() (NBD-PE, N360) has three important optical properties: photolability, which makes it suitable for photobleaching recovery measurements; concentration-dependent self-quenching; and fluorescence resonance energy transfer to rhodamine acceptors (usually rhodamine DHPE, L1392). Spectroscopic characteristics of NBD-PE are generally similar to those described for phospholipids with NBD-labeled acyl chains. NBD-PE is frequently used in NBD–rhodamine fluorescence energy transfer experiments to monitor membrane fusion (Lipid-Mixing Assays of Membrane Fusion—Note 13.1). In addition, this method can be used to detect lipid domain formation
(NBD-PE, N360) has three important optical properties: photolability, which makes it suitable for photobleaching recovery measurements; concentration-dependent self-quenching; and fluorescence resonance energy transfer to rhodamine acceptors (usually rhodamine DHPE, L1392). Spectroscopic characteristics of NBD-PE are generally similar to those described for phospholipids with NBD-labeled acyl chains. NBD-PE is frequently used in NBD–rhodamine fluorescence energy transfer experiments to monitor membrane fusion (Lipid-Mixing Assays of Membrane Fusion—Note 13.1). In addition, this method can be used to detect lipid domain formation ![]() and intermembrane lipid transfer
and intermembrane lipid transfer ![]() and to determine the transbilayer distribution of phospholipids.
and to determine the transbilayer distribution of phospholipids.![]() Attachment of the NBD fluorophore to the head group makes NBD-PE resistant to transfer between vesicles.
Attachment of the NBD fluorophore to the head group makes NBD-PE resistant to transfer between vesicles.![]() NBD-PE has been used in combination with either rhodamine DHPE (L1392) or Texas Red DHPE (T1395MP) for visualizing the spatial relationships of lipid populations by fluorescence resonance energy transfer microscopy.
NBD-PE has been used in combination with either rhodamine DHPE (L1392) or Texas Red DHPE (T1395MP) for visualizing the spatial relationships of lipid populations by fluorescence resonance energy transfer microscopy.![]() The nitro group of NBD can be reduced with sodium dithionite, irreversibly eliminating the dye's fluorescence (Figure 13.2.10). This technique can be employed to determine whether the probe is localized on the outer or inner leaflet of the cell membrane.
The nitro group of NBD can be reduced with sodium dithionite, irreversibly eliminating the dye's fluorescence (Figure 13.2.10). This technique can be employed to determine whether the probe is localized on the outer or inner leaflet of the cell membrane.![]() The argon-ion laser–excitable NBD-PE is also a frequent choice for fluorescence recovery after photobleaching (FRAP) measurements of lateral diffusion in membranes.
The argon-ion laser–excitable NBD-PE is also a frequent choice for fluorescence recovery after photobleaching (FRAP) measurements of lateral diffusion in membranes.![]() In addition, NBD-PE is of particular value for monitoring bilayer-to-hexagonal phase transitions, because these transitions cause an increase in NBD-PE's fluorescence intensity.
In addition, NBD-PE is of particular value for monitoring bilayer-to-hexagonal phase transitions, because these transitions cause an increase in NBD-PE's fluorescence intensity.![]()
Phospholipid with a Fluorescein-Labeled Head Group
Fluorescein-derivatized dihexadecanoylglycerophosphoethanolamine (fluorescein DHPE, F362) is a membrane-surface probe that is sensitive to both the local electrostatic potential and pH.![]() An anti–fluorescein/Oregon Green dye antibody (A889, Anti-Dye and Anti-Hapten Antibodies—Section 7.4) has been employed in combination with fluorescein DHPE to investigate specific recognition interactions at membrane surfaces
An anti–fluorescein/Oregon Green dye antibody (A889, Anti-Dye and Anti-Hapten Antibodies—Section 7.4) has been employed in combination with fluorescein DHPE to investigate specific recognition interactions at membrane surfaces ![]() (Antibodies for Detecting Membrane-Surface Labels—Note 13.2).
(Antibodies for Detecting Membrane-Surface Labels—Note 13.2).
Because of fluorescein's photolability, fluorescein DHPE is a useful reagent for measuring lateral diffusion in membranes using fluorescence photobleaching recovery methods.![]() Another technique, single-particle tracking (SPT), provides direct measurements of diffusion rates by calculating the trajectories of fluorescent polystyrene beads or colloidal gold particles from time-sequential images.
Another technique, single-particle tracking (SPT), provides direct measurements of diffusion rates by calculating the trajectories of fluorescent polystyrene beads or colloidal gold particles from time-sequential images.![]() FluoSpheres fluorescent microspheres (Microspheres—Section 6.5) were labeled with streptavidin and then coupled to fluorescein DHPE using a biotinylated conjugate of anti–fluorescein/Oregon Green monoclonal 4-4-20 (A6421, Anti-Dye and Anti-Hapten Antibodies—Section 7.4). Diffusion rates measured with this bridged conjugate in glass-supported phospholipid bilayers were the same as those determined with streptavidin beads coupled directly to biotin-X DHPE. Fluorescein DHPE has also been used in conjunction with polyclonal anti–fluorescein/Oregon Green dye antibody (A889, Anti-Dye and Anti-Hapten Antibodies—Section 7.4) to prepare colloidal gold probes for SPT diffusion measurements in supported phospholipid bilayers and in keratocyte plasma membranes.
FluoSpheres fluorescent microspheres (Microspheres—Section 6.5) were labeled with streptavidin and then coupled to fluorescein DHPE using a biotinylated conjugate of anti–fluorescein/Oregon Green monoclonal 4-4-20 (A6421, Anti-Dye and Anti-Hapten Antibodies—Section 7.4). Diffusion rates measured with this bridged conjugate in glass-supported phospholipid bilayers were the same as those determined with streptavidin beads coupled directly to biotin-X DHPE. Fluorescein DHPE has also been used in conjunction with polyclonal anti–fluorescein/Oregon Green dye antibody (A889, Anti-Dye and Anti-Hapten Antibodies—Section 7.4) to prepare colloidal gold probes for SPT diffusion measurements in supported phospholipid bilayers and in keratocyte plasma membranes.![]()
Phospholipid with an Oregon Green 488 Dye–Labeled Head Group
With absorption and emission spectra that are virtually superimposable on those of fluorescein, our Oregon Green 488 DHPE (O12650) provides an important alternative to fluorescein DHPE in its many applications. When compared with the fluorescein derivative, Oregon Green 488 DHPE exhibits greater photostability and a lower pKa (pKa = 4.7 versus 6.4 for fluorescein); however, these pKa values may differ when the probes are bound to membranes.
Phospholipid with a BODIPY FL Dye–Labeled Head Group
Our phospholipid with the green-fluorescent BODIPY FL dye attached to the head group (BODIPY FL DHPE, D3800) has significant potential for studies of molecular recognition interactions at membrane surfaces (Antibodies for Detecting Membrane-Surface Labels—Note 13.2). Spectral properties of this BODIPY probe is generally the same as those described above for phospholipids with BODIPY FL dye–labeled acyl chains.
Phospholipids with a Rhodamine or Texas Red Dye–Labeled Head Group
The rhodamine-labeled phospholipids TRITC DHPE and rhodamine DHPE (often referred to as N-Rh-PE, L1392; Figure 13.2.1G) do not readily transfer between separated lipid bilayers.![]() This property has led to the extensive use of rhodamine DHPE for membrane fusion assays based on fluorescence resonance energy transfer from NBD-PE (Lipid-Mixing Assays of Membrane Fusion—Note 13.1). In addition, these probes are good resonance energy transfer acceptors from fluorescent lipid analogs such as the BODIPY and NBD phospholipids
This property has led to the extensive use of rhodamine DHPE for membrane fusion assays based on fluorescence resonance energy transfer from NBD-PE (Lipid-Mixing Assays of Membrane Fusion—Note 13.1). In addition, these probes are good resonance energy transfer acceptors from fluorescent lipid analogs such as the BODIPY and NBD phospholipids ![]() and from protein labels such as 5-iodoacetamidofluorescein 5-IAF, I30451; Thiol-Reactive Probes Excited with Visible Light—Section 2.2) and IAEDANS
and from protein labels such as 5-iodoacetamidofluorescein 5-IAF, I30451; Thiol-Reactive Probes Excited with Visible Light—Section 2.2) and IAEDANS ![]() (I14, Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3). Rhodamine-labeled phospholipids have also been used as tracers for membrane traffic during endocytosis
(I14, Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3). Rhodamine-labeled phospholipids have also been used as tracers for membrane traffic during endocytosis ![]() and for lipid processing in hepatocytes.
and for lipid processing in hepatocytes.![]() Texas Red DHPE (T1395MP) is principally employed as an energy transfer acceptor from NBD, BODIPY and fluorescein lipid probes. The longer emission wavelength of the Texas Red dye provides superior separation of the donor and acceptor emission signals in resonance energy transfer microscopy.
Texas Red DHPE (T1395MP) is principally employed as an energy transfer acceptor from NBD, BODIPY and fluorescein lipid probes. The longer emission wavelength of the Texas Red dye provides superior separation of the donor and acceptor emission signals in resonance energy transfer microscopy.![]() This technique has enabled visualization of ATP-dependent fusion of liposomes with the Golgi apparatus.
This technique has enabled visualization of ATP-dependent fusion of liposomes with the Golgi apparatus.![]() Membrane flux during hemagglutinin-mediated cell–cell fusion has been visualized using Texas Red DHPE and the lipophilic carbocyanine DiI (D282, D3911; Dialkylcarbocyanine and Dialkylaminostyryl Probes—Section 13.4) as membrane labels.
Membrane flux during hemagglutinin-mediated cell–cell fusion has been visualized using Texas Red DHPE and the lipophilic carbocyanine DiI (D282, D3911; Dialkylcarbocyanine and Dialkylaminostyryl Probes—Section 13.4) as membrane labels.![]()
Phospholipids with a Biotinylated Head Group
Phospholipids labeled with a biotin at the head group facilitate binding of labeled membranes to other biomolecules. The biotinylated phospholipids (biotin DHPE, biotin-X DHPE) can be used to couple avidin or streptavidin (Molecular Probes avidin, streptavidin, NeutrAvidin and CaptAvidin conjugates—Table 7.10) to cell membranes, liposomes and lipid monolayers.![]() Avidin can then be employed as a bridge for antibody coupling or for assembling liposomes into multiplex structures.
Avidin can then be employed as a bridge for antibody coupling or for assembling liposomes into multiplex structures.![]() Liposomes incorporating biotinylated phospholipids can also be used to immobilize membrane-bound proteins for analysis by affinity chromatography.
Liposomes incorporating biotinylated phospholipids can also be used to immobilize membrane-bound proteins for analysis by affinity chromatography.![]() Interactions of biotinylated lipids with streptavidin provide a model for molecular recognition processes at membrane surfaces.
Interactions of biotinylated lipids with streptavidin provide a model for molecular recognition processes at membrane surfaces.![]() The phase structure of lipid assemblies incorporating biotinylated phospholipids has been studied by X-ray diffraction,
The phase structure of lipid assemblies incorporating biotinylated phospholipids has been studied by X-ray diffraction,![]() 31P NMR and differential scanning calorimetry.
31P NMR and differential scanning calorimetry.![]()
With the resolution inherent in an image-based methodology and the productivity of high-throughput assays, high-content screening (HCS) or automated imaging provides a powerful tool for studying biology in a spatial and temporal context. Using HCS technology, researchers can examine multiple cellular targets and parameters in a large number of individually imaged cells and quantitatively assess the data. While many Molecular Probes products can be directly applied to HCS protocols, we have developed validated tools and assays specifically for HCS platforms. These HCS products are:
- Validated on multiple imaging platforms
- Packaged in automation-compatible formulations
- Compatible with multiplex applications
Although designed for HCS platforms, HCS products and kits can also be used with conventional fluorescence microscopes equipped with standard optical filter sets.
HCS LipidTOX Phospholipidosis Detection Reagents
Phospholipidosis is often triggered by cationic amphiphilic drugs, which become enriched in lysosomes to high concentrations and inhibit normal metabolism of phospholipids. The subsequent intracellular accumulation of phospholipids and formation of lamellar bodies—phospholipidosis—can be detected in cells incubated in the presence of phospholipids conjugated to fluorescent dyes.
HCS LipidTOX Green and HCS LipidTOX Red phospholipidosis detection reagents (H34350, H34351), also called LipidTOX phospholipid stains, were specifically developed to characterize the potentially toxic side effects of compounds on lipid metabolism in mammalian cell lines using image-based HCS assays.![]() Key advantages of this series of phospholipidosis detection reagents over conventional stains such as NBD-PE (N360) include their ready-to-use aqueous formulation, their narrow emission profiles (excitation/emission maxima ~495/525 nm for HCS LipidTOX Green phospholipidosis detection reagent and ~595/615 nm for HCS LipidTOX Red phospholipidosis detection reagent) and their compatibility with HCS LipidTOX neutral lipid stains (see below).
Key advantages of this series of phospholipidosis detection reagents over conventional stains such as NBD-PE (N360) include their ready-to-use aqueous formulation, their narrow emission profiles (excitation/emission maxima ~495/525 nm for HCS LipidTOX Green phospholipidosis detection reagent and ~595/615 nm for HCS LipidTOX Red phospholipidosis detection reagent) and their compatibility with HCS LipidTOX neutral lipid stains (see below).
HCS LipidTOX phospholipidosis detection reagents have not been observed to affect the normal growth of cells, and their live-cell staining patterns are maintained after formaldehyde fixation. These reagents are designed for fixed–end point workflows in which formaldehyde-fixed cells in microplates are processed, imaged and analyzed. HCS LipidTOX phospholipidosis detection reagents can easily be detected with fluorescence microscopes or HCS readers equipped with standard filter sets.
HCS LipidTOX Neutral Lipid Stains
As with phospholipidosis, steatosis or the intracellular accumulation of neutral lipids as lipid droplets or globules is often triggered by drugs that affect the metabolism of fatty acids or neutral lipids. HCS LipidTOX neutral lipid stains were developed to characterize the effects of drugs and other compounds on lipid metabolism in mammalian cell lines. HCS LipidTOX neutral lipid stains have an extremely high affinity for neutral lipid droplets. These reagents are added after cell fixation and do not require subsequent wash steps after incubation with the sample. Key advantages of this series of neutral lipid stains over conventional stains such as nile red (N1142, Other Nonpolar and Amphiphilic Probes—Section 13.5) include their ready-to-use formulations, their flexibility for multiplexing protocols and their compatibility with HCS LipidTOX phospholipidosis detection reagents (see above). HCS LipidTOX neutral lipid stains can also be used to monitor the formation and differentiation of adipocytes, a process called adipogenesis. Adipogenesis is of acute interest to the biomedical and drug discovery community as it plays an important role in diseases such as obesity, diabetes and atherosclerosis.
Described more thoroughly in Other Nonpolar and Amphiphilic Probes—Section 13.5, HCS LipidTOX neutral lipid stains are available with green, red and deep red fluorescence emission:
- HCS LipidTOX Green neutral lipid stain (H34475), with excitation/emission maxima ~495/505 nm (Figure 13.2.11)
- HCS LipidTOX Red neutral lipid stain (H34476), with excitation/emission maxima ~577/609 nm
- HCS LipidTOX Deep Red neutral lipid stain (H34477), with excitation/emission maxima ~637/655 nm
HCS LipidTOX neutral lipid stains are designed for fixed–end point workflows in which formaldehyde-fixed cells in microplates are processed, imaged and analyzed. These stains can easily be detected with fluorescence microscopes or HCS readers equipped with standard filter sets.
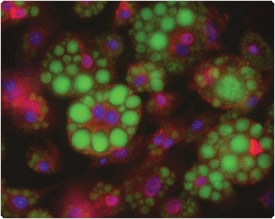
Figure 13.2.11 LipidTOX Green neutral lipid stain and fatty acid–binding protein (FABP4) antibody labeling in adipocytes. Adipocytes differentiated from 3T3-L1 mouse fibroblasts were fixed with formaldehyde and permeabilized with saponin before labeling with rabbit anti–fatty acid binding protein (FABP4) IgG (red). These cells were then stained with LipidTOX Green neutral lipid stain (H34475, green), counterstained with DAPI (D1306, D21490; blue) and mounted in ProLong Gold antifade reagent (P36930).
HCS LipidTOX Phospholipidosis and Steatosis Detection Kit
The detection and analysis of pre-lethal mechanisms in toxicological profiling and compound screening are extremely important components of the drug discovery process. The cationic amphiphilic drugs are among the most prominent examples of compounds that impact lipid metabolism of cells. These drugs tend to become enriched in lysosomes to high concentrations and inhibit the normal metabolism of phospholipids, which in turn causes the intracellular accumulation of phospholipids and the formation of lamellar bodies. Other drug classes more adversely affect various aspects of fatty acid or neutral lipid metabolism, leading to the cytoplasmic accumulation of neutral lipid as lipid droplets or globules.
The HCS LipidTOX Phospholipidosis and Steatosis Detection Kit (H34158) provides a complete set of reagents for performing validated HCS assays to detect and distinguish these two facets of cytotoxicity—the intracellular accumulation of phospholipids (phospholipidosis) and of neutral lipids (steatosis)—in mammalian cell lines after exposure to test compounds.![]() This kit includes an aqueous, red-fluorescent formulation of labeled phospholipids (LipidTOX Red phospholipid stain, excitation/emission ~595/615 nm) and a ready-to-use, highly selective green-fluorescent stain for neutral lipids (LipidTOX Green neutral lipid stain, excitation/emission ~495/505 nm), which can be used sequentially for the analysis of phospholipidosis and steatosis, respectively, or can be used separately for single-parameter analysis.
This kit includes an aqueous, red-fluorescent formulation of labeled phospholipids (LipidTOX Red phospholipid stain, excitation/emission ~595/615 nm) and a ready-to-use, highly selective green-fluorescent stain for neutral lipids (LipidTOX Green neutral lipid stain, excitation/emission ~495/505 nm), which can be used sequentially for the analysis of phospholipidosis and steatosis, respectively, or can be used separately for single-parameter analysis.
After incubation with LipidTOX Red phospholipid stain and a test compound, the cells are fixed with formaldehyde and labeled with LipidTOX Green neutral lipid stain (Figure 13.2.12). Neither LipidTOX Red phospholipid stain, nor LipidTOX Green phospholipid stain described above, requires sonication or organic solvents. Furthermore, LipidTOX Green neutral lipid stain (as well as the other LipidTOX neutral lipid stains described above) is more selective than nile red, allowing you to easily distinguish neutral lipids (such as those in adipocytes and cells undergoing steatosis) from other types of lipids.
Each HCS LipidTOX Phospholipidosis and Steatosis Detection Kit provides:
- LipidTOX Red phospholipid stain
- LipidTOX Green neutral lipid stain
- Hoechst 33342 for nuclear labeling
- Propranolol, a positive-control compound for inducing phospholipidosis
- Cyclosporin A, a positive-control compound for inducing steatosis
- Dimethylsulfoxide (DMSO)
- Detailed protocols (HCS LipidTOX Phospholipidosis and Steatosis Detection Kit)
Sufficient reagents are provided for 1200 assays (H34158, 10-plate size), based on assay volumes of 100 µL per well. This kit is designed for fixed–end point workflows in which formaldehyde-fixed cells in microplates are processed, imaged and analyzed. The fluorescent stains used for the analysis of phospholipidosis and steatosis can easily be detected with fluorescence microscopes or HCS readers equipped with standard filter sets.
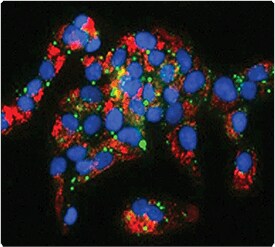
Figure 13.2.12 Multiplex detection of phospholipidosis and steatosis in HepG2 cells using the HCS LipidTOX Phospholipidosis and Steatosis Detection Kit (H34158). HepG2 cells were co-incubated with tamoxifen and LipidTOX Red phospholipid stain, followed by fixation with formaldehyde and labeling with HCS LipidTOX Green neutral lipid stain and Hoechst 33342 (H1399, H3570, H21492).
For a detailed explanation of column headings, see Definitions of Data Table Contents
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Notes |
|---|---|---|---|---|---|---|---|---|
| ADIFAB fatty acid indicator | ~15,350 | FF,L,AA | H2O | 365 | 10,500 | 432 | H2O | 1 |
| A10070 PED-A1 | 880.68 | FF,D,L | DMSO | 505 | 92,000 | 512 | MeOH | 16 |
| A10072 Red/Green BODIPY PC-A2 (ratiometric phospholipase A2 substrate) | 986.67 | FF,D,L | DMSO | 505 | 85,000 | 567 | MeOH | 17, 18 |
| biotin DHPE | 1019.45 | FF,D | see Notes | <300 | none | 2 | ||
| biotin-X DHPE | 1132.61 | FF,D | see Notes | <300 | none | 2 | ||
| 1,2-bis-(1-pyrenebutanoyl)-sn-glycero-3-phosphocholine | 797.88 | FF,D,L | see Notes | 342 | 75,000 | 471 | EtOH | 3 |
| 1,2-bis-(1-pyrenedecanoyl)-sn-glycero-3-phosphocholine | 966.20 | FF,D,L | see Notes | 340 | 62,000 | 473 | EtOH | 4 |
| BODIPY 500/510 C4, C9 | 404.31 | F,L | DMSO | 509 | 101,000 | 515 | MeOH | 5 |
| B7701 bis-BODIPY FL C11-PC | 1029.80 | FF,D,L | see Notes | 505 | 123,000 | 512 | MeOH | 2, 6 |
| dansyl DHPE | 1026.44 | FF,D,L | see Notes | 336 | 4500 | 517 | MeOH | 2 |
| DAUDA | 434.59 | F,L | DMSO, EtOH | 335 | 4800 | 519 | MeOH | |
| β-DPH HPC | 782.01 | FF,D,L | see Notes | 354 | 81,000 | 428 | MeOH | 2, 7 |
| 2-decanoyl-1-(O-(11-(4,4-difluoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene-3-propionyl)amino) undecyl)-sn-glycero-3-phosphocholine | 854.86 | FF,D,L | see Notes | 506 | 71,000 | 512 | EtOH | 2 |
| D3792 β-BODIPY FL C12-HPC | 895.95 | FF,D,L | see Notes | 506 | 86,000 | 513 | EtOH | 2, 5 |
| β-BODIPY 500/510 C12-HPC | 881.93 | FF,D,L | see Notes | 509 | 86,000 | 513 | EtOH | 2, 5 |
| D3800 BODIPY FL DHPE | 1067.23 | FF,D,L | see Notes | 505 | 87,000 | 511 | MeOH | 2, 5 |
| β-BODIPY FL C5-HPC | 797.77 | FF,D,L | see Notes | 503 | 80,000 | 512 | MeOH | 2, 5 |
| β-BODIPY FL C5-HPA | 746.68 | FF,D,L | see Notes | 504 | 79,000 | 511 | MeOH | 2, 5 |
| β-BODIPY 530/550 C5-HPC | 921.91 | FF,D,L | see Notes | 534 | 64,000 | 552 | MeOH | 2, 5 |
| D3821 BODIPY FL C16 | 474.44 | F,L | DMSO | 505 | 90,000 | 512 | MeOH | 5 |
| D3822 BODIPY FL C12 | 418.33 | F,L | DMSO | 505 | 87,000 | 511 | MeOH | 5 |
| D3823 BODIPY 500/510 C1, C12 | 404.31 | F,L | DMSO | 508 | 97,000 | 514 | MeOH | 5 |
| BODIPY 500/510 C8, C5 | 404.31 | F,L | DMSO | 509 | 100,000 | 515 | MeOH | 5 |
| BODIPY 530/550 C12 | 542.47 | F,L | DMSO | 534 | 76,000 | 552 | MeOH | 5 |
| D3834 BODIPY FL C5 | 320.15 | F,L | DMSO, MeCN | 505 | 96,000 | 511 | MeOH | 5 |
| D3835 BODIPY 558/568 C12 | 472.40 | F,L | DMSO | 559 | 91,000 | 568 | MeOH | 5 |
| D3861 BODIPY 581/591 C11 (lipid peroxidation sensor) | 504.43 | F,L | DMSO | 582 | 140,000 | 591 | MeOH | 8 |
| BODIPY FL C11 | 404.31 | F,L | DMSO | 505 | 92,000 | 510 | MeOH | 5 |
| D23739 PED6 | 1136.13 | FF,D,L | DMSO | 505 | 92,000 | 511 | MeOH | 2, 9 |
| F362 fluorescein DHPE | 1182.54 | FF,D,L | see Notes | 496 | 88,000 | 519 | MeOH | 2, 10 |
| β-py-c10-HPC | 850.13 | FF,D,L | see Notes | 342 | 37,000 | 376 | MeOH | 2, 11, 12 |
| β-py-C10-PG | 856.09 | FF,D,L | see Notes | 341 | 38,000 | 376 | MeOH | 2, 11, 12 |
| H34350 HCS LipidTOX Green Phospholipidosis Detection Reagent | ~1100 | F,L | H2O | 495 | 84,000 | 525 | MeOH | 15 |
| H34351 HCS LipidTOX Red Phospholipidosis Detection Reagent | ~1400 | F,L | H2O | 595 | 112,000 | 615 | MeOH | 15 |
| L1392 rhodamine DHPE | 1333.81 | FF,D,L | see Notes | 560 | 75,000 | 581 | MeOH | 2 |
| M12652 Marina Blue DHPE | 944.14 | FF,D,L | see Notes | 365 | 18,000 | 460 | MeOH | 2, 10 |
| NBD-X | 294.27 | L | DMSO | 467 | 23,000 | 539 | MeOH | 13 |
| N360 NBD-PE | 956.25 | FF,D,L | see Notes | 463 | 21,000 | 536 | MeOH | 2, 13 |
| 12-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)dodecanoic acid | 378.43 | L | DMSO | 467 | 24,000 | 536 | MeOH | 13 |
| NBD C6-HPC | 771.89 | FF,D,L | see Notes | 465 | 21,000 | 533 | EtOH | 2, 13 |
| NBD C12-HPC | 856.05 | FF,D,L | see Notes | 465 | 22,000 | 534 | EtOH | 2, 13 |
| O12650 Oregon Green 488 DHPE | 1086.25 | FF,D,L | see Notes | 501 | 85,000 | 526 | MeOH | 2, 10 |
| 1-pyrenedecanoic acid | 372.51 | L | DMF, DMSO | 341 | 43,000 | 377 | MeOH | 11, 12 |
| 1-pyrenedodecanoic acid | 400.56 | L | DMF, DMSO | 341 | 44,000 | 377 | MeOH | 11, 12 |
| 1-pyrenehexadecanoic acid | 456.67 | L | DMF, DMSO | 341 | 43,000 | 377 | MeOH | 11, 12 |
| 1-pyrenebutanoic acid | 288.35 | L | DMF, DMSO | 341 | 43,000 | 376 | MeOH | 11, 12 |
| Pacific Blue DMPE | 961.17 | FF,D,L | see Notes | 411 | 40,000 | 454 | MeOH | 2 |
| cis-parinaric acid | 276.42 | FF,LL,AA | EtOH | 304 | 77,000 | 416 | MeOH | 14, 15 |
| TRITC DHPE | 1236.68 | FF,D,L | see Notes | 540 | 93,000 | 566 | MeOH | 2 |
| T1395MP Texas Red DHPE | 1381.85 | FF,D,L | see Notes | 583 | 115,000 | 601 | MeOH | 2 |
| ||||||||
For Research Use Only. Not for use in diagnostic procedures.

