Search
Loading and Calibration of Intracellular Ion Indicators—Note 19.1
There are two major prerequisites for measuring intracellular ion concentrations using fluorescent indicators:
- Loading: The indicator must be localized in the region (most commonly the cytosol but sometimes the mitochondria) where the ion concentration is to be measured.
- Calibration: The fluorescence of the indicator must be quantitatively related to the concentration of the free ion.
This technical note focuses on these prerequisites. Further information on the practical aspects of ion measurements using fluorescent indicators can be found in several reviews.![]()
Cell loading methods can be divided into two groups. Bulk loading procedures are applicable to large populations of cells and include:
- Acetoxymethyl (AM) ester loading

- ATP-induced permeabilization

- Electroporation

- Hypoosmotic shock

- Influx pinocytic cell-loading reagent
 (I14402, Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8)
(I14402, Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8) - Coupling to cell-penetrating peptides (CPP)

- Ballistic microprojectile delivery

Procedures such as microinjection ![]() and infusion from whole-cell patch pipettes
and infusion from whole-cell patch pipettes ![]() must be carried out one cell at a time. Reviews of some of these techniques have been published;
must be carried out one cell at a time. Reviews of some of these techniques have been published;![]() see also Techniques for loading molecules into the cytoplasm—Table 14.1 (Choosing a Tracer—Section 14.1).
see also Techniques for loading molecules into the cytoplasm—Table 14.1 (Choosing a Tracer—Section 14.1).
The AM Ester Loading Technique
The noninvasive and technically straightforward AM ester technique is by far the most popular method for loading fluorescent ion indicators (Figure 1). The carboxylate groups of indicators for Ca2+ and other cations and the phenolic hydroxyl groups of pH indicators are derivatized as acetoxymethyl or acetate esters, respectively, rendering the indicator permeant to membranes and insensitive to ions. Once inside the cell, these derivatized indicators are hydrolyzed by ubiquitous intracellular esterases, releasing the ion-sensitive polyanionic indicator.
In practice, a 1–10 mM stock solution of the ester probe in anhydrous dimethylsulfoxide (DMSO) is prepared and divided into appropriately sized aliquots that can be stored desiccated at –20°C. This procedure will curtail the spontaneous ester hydrolysis that can occur in moist environments. Before loading, the DMSO stock solution should be diluted at least 1:200 in serum-free culture medium to a final concentration of about 1–10 µM. The nonionic and nondenaturing detergent Pluronic F-127 or the related PowerLoad reagent (P3000MP, P6866, P6867, P10020; Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8) are frequently added to help disperse the indicator in the loading medium.![]() After incubation at 20–37°C for 15–60 minutes, the cells should be washed two to three times with fresh serum-free culture medium (serum may contain esterase activity). The loading medium should also be free of amino acids or buffers containing primary or secondary amines because aliphatic amines may cleave the AM esters and prevent loading. The overall loading efficiency is typically 10–40%, depending on the molecular structure of the indicator, the type of cells and the incubation conditions.
After incubation at 20–37°C for 15–60 minutes, the cells should be washed two to three times with fresh serum-free culture medium (serum may contain esterase activity). The loading medium should also be free of amino acids or buffers containing primary or secondary amines because aliphatic amines may cleave the AM esters and prevent loading. The overall loading efficiency is typically 10–40%, depending on the molecular structure of the indicator, the type of cells and the incubation conditions.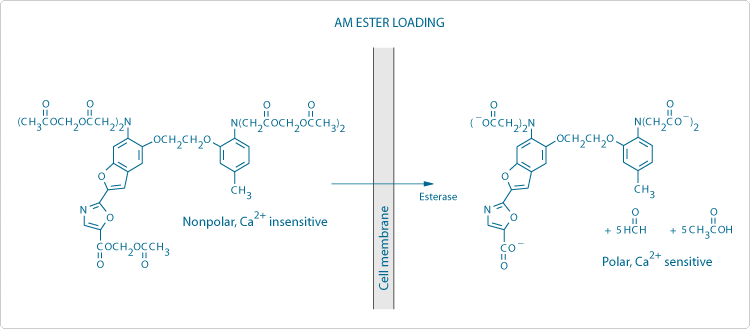
Figure 1. Schematic diagram of the processes involved in loading cells using membrane-permeant acetoxymethyl (AM) ester derivatives of fluorescent indicators, in this case fura-2. Note the generation of potentially toxic by-products (formaldehyde and acetic acid).
Problems with AM Ester Loading
- Compartmentalization: For calibration purposes (see below), it is usually assumed that fluorescent indicators are homogeneously distributed in the cytosol and equally responsive to variations of intracellular ion concentration. However, AM esters and their hydrolysis products are capable of accumulating in any membrane-enclosed structure within the cell. In addition, indicators in polyanionic form may be sequestered within organelles via active transport processes.
 Compartmentalization is usually more pronounced at higher loading temperatures and is particularly acute in plant and fungal cells.
Compartmentalization is usually more pronounced at higher loading temperatures and is particularly acute in plant and fungal cells. The extent of compartmentalization can be assessed by image analysis, as well as fluorometrically using membrane permeabilization reagents, such as Triton X-100.
The extent of compartmentalization can be assessed by image analysis, as well as fluorometrically using membrane permeabilization reagents, such as Triton X-100.
- Incomplete AM ester hydrolysis: Residual unhydrolyzed AM esters may be present extracellularly due to incomplete removal by washing. Inside the cell, low levels of intracellular esterase activity, which can vary considerably from one cell type to another, may produce only partial AM ester hydrolysis.
 Because even partially hydrolyzed AM esters are Ca2+-insensitive, detection of their fluorescence as part of the total signal leads to an underestimation of the Ca2+ concentration.
Because even partially hydrolyzed AM esters are Ca2+-insensitive, detection of their fluorescence as part of the total signal leads to an underestimation of the Ca2+ concentration. Fluorescence quenching by Mn2+, which only binds with high affinity to completely de-esterified indicators, can be used to quantitate these effects. Note that although some indicators are fluorescent in the AM ester form, others are not (Summary of Molecular Probes fluorescent Ca2+ indicators—Table 19.1).
Fluorescence quenching by Mn2+, which only binds with high affinity to completely de-esterified indicators, can be used to quantitate these effects. Note that although some indicators are fluorescent in the AM ester form, others are not (Summary of Molecular Probes fluorescent Ca2+ indicators—Table 19.1). - Extracellular AM ester hydrolysis: High levels of extracellular esterase activity can make AM ester loading ineffective, particularly for in vivo applications.

- Leakage: Extrusion of anionic indicators from cells by organic ion transporters can be reduced by cooling the sample or by applying inhibitors such as probenecid (P36400, Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8), sulfinpyrazone and MK571.
 AM esters have been shown to be extruded by the P-glycoprotein multidrug transporter
AM esters have been shown to be extruded by the P-glycoprotein multidrug transporter  (Probes for Cell Adhesion, Chemotaxis, Multidrug Resistance and Glutathione—Section 15.6). Ratiometric measurements (see below) help to minimize the impact of indicator leakage on experimental data.
(Probes for Cell Adhesion, Chemotaxis, Multidrug Resistance and Glutathione—Section 15.6). Ratiometric measurements (see below) help to minimize the impact of indicator leakage on experimental data.
Ion Dissociation Constants
The dissociation constant (Kd) is the key conversion parameter linking fluorescence signals to ion concentrations, assuming that the indicator is operating as an equilibrium sensor. This conventional assumption requires that the concentration of the indicator is close to the Kd value. Because intracellular indicator concentrations can easily reach 10–100 µM, even if the externally applied concentration is only 1–10 µM, this assumption is not always valid.![]() For pH indicators, Kd is conventionally expressed as its negative log (pKa). The concentration range over which an indicator produces an observable response is approximately 0.1 × Kd to 10 × Kd. For ratiometric measurements, the response range also depends on wavelength-dependent parameters.
For pH indicators, Kd is conventionally expressed as its negative log (pKa). The concentration range over which an indicator produces an observable response is approximately 0.1 × Kd to 10 × Kd. For ratiometric measurements, the response range also depends on wavelength-dependent parameters.![]() For BAPTA-based Ca2+ indicators in particular, the Kd is very sensitive to a number of environmental factors, including temperature, pH, ionic strength and interactions of the indicator with proteins.
For BAPTA-based Ca2+ indicators in particular, the Kd is very sensitive to a number of environmental factors, including temperature, pH, ionic strength and interactions of the indicator with proteins.![]() Examination of published data shows that values of Kd determined in situ within cells can be up to fivefold higher than values determined in vitro
Examination of published data shows that values of Kd determined in situ within cells can be up to fivefold higher than values determined in vitro![]() (Comparison of in vitro and in situ Kd values for various Ca2+ indicators—Table 19.2), underscoring the importance of performing calibrations to determine the Kd directly in the system under study.
(Comparison of in vitro and in situ Kd values for various Ca2+ indicators—Table 19.2), underscoring the importance of performing calibrations to determine the Kd directly in the system under study.
Calibration Methodology
Calibration procedures basically consist of recording fluorescence signals corresponding to a series of precisely manipulated ion concentrations. The resulting sigmoidal titration curve is either linearized by means of a Hill plot or analyzed directly by nonlinear regression to yield Kd. For in vitro calibrations of Ca2+ indicators, EGTA buffering is widely used to produce defined Ca2+ concentrations that can be calculated from the Kd of the Ca2+-EGTA complex.![]() This technique is used in the Calcium Calibration Buffer Kits (Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8). In situ calibrations of intracellular indicators generally utilize an ionophore to equilibrate the controlled external ion concentration with the ion concentration within the cell.
This technique is used in the Calcium Calibration Buffer Kits (Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8). In situ calibrations of intracellular indicators generally utilize an ionophore to equilibrate the controlled external ion concentration with the ion concentration within the cell.![]() Commonly used ionophores include:
Commonly used ionophores include:
- A-23187 (A1493), 4-bromo A-23187 (B1494) or ionomycin (I24222) for Ca2+ and Mg2+ (Chelators, Calibration Buffers, Ionophores and Cell-Loading Reagents—Section 19.8)
- Nigericin (N1495; Probes Useful at Near-Neutral pH—Section 20.2, Detecting Chloride, Phosphate, Nitrite and Other Anions—Section 21.2) for H+ and Cl–
- Gramicidin (G6888, Fluorescent Na+ and K+ Indicators—Section 21.1) for Na+
- Valinomycin (V1644, Fluorescent Na+ and K+ Indicators—Section 21.1) for K+
Ratiometric Calibration
Indicators that show an excitation or emission spectral shift upon ion binding can be calibrated using a ratio of the fluorescence intensities measured at two different wavelengths, resulting in the cancellation of artifactual variations in the fluorescence signal that might otherwise be misinterpreted as changes in ion concentration (Figure 2). Note that background levels must be subtracted from the component fluorescence intensities before calculation of the ratio. Examples of indicators exhibiting ion-dependent spectral shifts include the Ca2+ indicators fura-2 (Figure 3) and indo-1 (Fluorescent Ca2+ Indicators Excited with UV Light—Section 19.2), and the pH indicators BCECF and SNARF-1 (Probes Useful at Near-Neutral pH—Section 20.2). The ratio of two intensities with opposite ion-sensitive responses (for example, 340 nm/380 nm in Figure 3) gives the largest possible dynamic range of ratio signals for a particular indicator. Alternatively, the ratio of an ion-sensitive intensity to an ion-insensitive intensity (measured at a spectral isosbestic point, e.g., 360 nm in Figure 3) can be used (Figure 2). Ratiometric measurements reduce or eliminate variations of several determining factors in the measured fluorescence intensity, including indicator concentration, excitation path length, excitation intensity and detection efficiency.![]() Artifacts that are eliminated include photobleaching and leakage of the indicator, variable cell thickness, and nonuniform indicator distribution within cells (due to compartmentalization) or among populations of cells (due to loading efficacy variations).
Artifacts that are eliminated include photobleaching and leakage of the indicator, variable cell thickness, and nonuniform indicator distribution within cells (due to compartmentalization) or among populations of cells (due to loading efficacy variations).
 | Figure 2. Simulated data demonstrating the practical importance of ratiometric fluorescence techniques. This figure represents an ion indicator that exhibits a fluorescence intensity increase in response to ion binding at wavelength λ1 and a corresponding decrease at λ3. Fluorescence measured at an isosbestic point (λ2) is independent of ion concentration. The intracellular indicator concentration diminishes rapidly due to photobleaching, leakage (assuming the extracellular indicator is not detectable) or some other process. The change of intracellular ion concentration due to a stimulus applied at the time indicated by the arrow is unambiguously identified by recording the fluorescence intensity ratios λ1/λ3 or λ1/λ2. |
For Research Use Only. Not for use in diagnostic procedures.
