Search
DAPI Protocol for Fluorescence Imaging

Nuclear counterstain for fluorescence microscopy
A popular nuclear and chromosome counterstain, Thermo Scientific™ DAPI (4′,6-diamidino-2-phenylindole) emits blue fluorescence upon binding to AT regions of DNA.
This protocol can be used for:
- Nucleic acid (nuclear) staining in fluorescence microscopy
This protocol should not be used for:
- Flow cytometry
You will need the following for this protocol:
- Cells growing in culture
- DAPI, hydrochloride (Cat. No. D1306)
- Phosphate-buffered saline (PBS) or other suitable buffer
- Fluorescence microscope
Simulate your staining experiment before you run it
The Stain-iT simulator can help reduce trial and error and wasted reagents.
Protocol
Preparing solutions
Labeling fixed cells
First, fix and permeabilize cultured cells with a protocol appropriate for your sample.
Spectral information and storage
| DAPI | |
|---|---|
| Excitation/Emission (nm) | 358/461 |
| Standard filter set | DAPI |
| Invitrogen™ EVOS™ Light Cube | DAPI |
| Storage conditions | ≤–20°C |
Protocol tips
- DAPI is a known mutagen and should be handled with care.
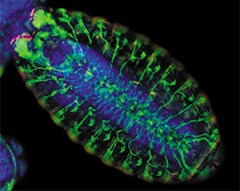
Drosophila melanogaster embryo staining. Wild type (Canton-S) Drosophila melanogaster embryo exhibiting microtubule staining (green fluorescence), denticle band staining (red fluorescence), and nuclear staining (blue-fluorescent DAPI).