Search
TO-PRO-3 Stain Protocol

Nuclear stain for fixed cells
TO-PRO-3 is a cell-impermeant dye that gives strong and selective staining of the nucleus in cultured cells and paraffin sections. TO-PRO-3 and other carbocyanine monomers have very strong binding affinity for dsDNA, with dissociation constants in the micromolar range. The long-wavelength fluorescence of TO-PRO-3 is outside the spectral range of tissue autofluorescence as well as commonly used fluorophores in the DAPI and FITC channels.
This protocol can be used for:
- Nucleic acid (nuclear) staining in fluorescence microscopy
This protocol should not be used for:
- Flow cytometry
You will need the following for this protocol:
- Cells growing in culture
- TO-PRO-3 stain (Cat. No. T3605)
- Fluorescence microscope
Protocol
Labeling fixed cells
First, fix and permeabilize cultured cells with a protocol appropriate for your sample.
|
|
| TO-PRO-3 | |
|---|---|
| Excitation/Emission (nm) | 642/661 |
| Standard filter set | Far Red |
| EVOS Light Cube | Cy®5 |
| Storage conditions | ≤–20°C |
Protocol tips
- Warm to room temperature and briefly centrifuge the DMSO solution to the bottom of the vial each time before use.
- Try multiple dye concentrations in the range from 100 nM to 5 µM to determine the optimal concentration.
- RNase treatment is not necessary but could improve nuclear signals over cytoplasmic RNA background.
- Treat all nucleic acid binding dyes as potential mutagens and handle with care.
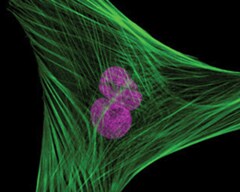
Muntjac fibroblast labeled with probes for actin and the nucleus. The actin cytoskeleton was visualized using the green-fluorescent Alexa Fluor 488 phalloidin. The nucleus was stained with TO-PRO-3 reagent.