Search

ProLong Antifade Mountants and Reagents |
Enhance the quality of your fluorescence microscopy imaging with our selection of ProLong antifade mountants and ProLong Live antifade reagents. Designed to suppress photobleaching and preserve fluorescent signal intensity, these products offer excellent protection for your samples. Our newest offering, ProLong RapidSet antifade mountants, provide a fast-curing antifade solution for researchers needing same-day imaging and high-throughput workflows.
ProLong antifade mountants for fixed cells offer reliable, long-term protection for fixed samples, helping ensure that the fluorescence signals of your labeled target molecules remain robust for extended storage and analysis. ProLong Live antifade reagents cater to live-cell imaging needs by maintaining cell viability and fluorescence signal over extended imaging sessions. Explore our complete lineup of antifade mountants—including fast-setting and long-term imaging options—for your fluorescent microscopy needs.
Product highlights
ProLong antifade mountants for fixed cells
- Inhibits photobleaching across the spectrum, even after prolonged storage
- Low background across the spectrum
- Hard setting mountants
- Available with or without DAPI
- Ready-to-use benchtop formulations
- Choice of refractive index based on your needs: 1.52 for ProLong Glass reagent, 1.49 for ProLong RapidSet reagent (after 1 hour), and 1.47 for ProLong Diamond and ProLong Gold reagents
- Need a faster workflow? Explore ProLong RapidSet Antifade Mountant—a quick-curing, high-performance mountant excellent for same-day imaging and time-sensitive studies.
ProLong antifade reagents for live cells
- Inhibits photobleaching in live cells
- Compatible with fluorescent dyes and proteins across the spectrum
- Extends imaging times in time-lapse experiments
- Minimal effects on viability and proliferation
- Ready-to-use 100X formulation
- Refractive index: 1.3
Select the optimal ProLong antifade for your imaging experiment
ProLong antifade mountants are ready-to-use, hard-setting reagents used for the long-term imaging of fixed animal cells and tissues.
| NEWProLong RapidSet antifade mountant | ProLong Glass | ProLong Diamond | ProLong Gold | ||||
| ProLong Glass antifade mountant | ProLong Glass antifade mountant with NucBlue stain | ProLong Diamond antifade mountant | ProLong Diamond antifade mountant with DAPI | ProLong Gold antifade mountant | ProLong Gold antifade mountant with DAPI | ||
| Curing time | 1 hour | 18–60 hours (depending on specimen thickness) | 24 hours | 24 hours | |||
|---|---|---|---|---|---|---|---|
| Sample thickness | Up to 80 µm sample thickness | Up to 150 µm sample thickness | Up to 10 µm sample thickness | Up to 10 µm sample thickness | |||
| Refractive index (after curing) | 1.49 (after 1 hr curing) RI 1.52 (after 24 hr curing) | 1.52 (after 24 hr curing) | 1.47 (after 24 hr curing) | 1.47 (after 24 hr curing) | |||
| Sample storage temperature | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | |||
| Optimal microscope objective* | Oil-immersion | Oil-immersion | Glycerol-corrected | Glycerol-corrected | |||
| Compatible fluorophores | Most dyes and fluorescent proteins | Most dyes and fluorescent proteins | Most dyes and fluorescent proteins | Alexa Fluor dyes | |||
| Reagent storage temperature | 2-8°C | 2-8°C | 2-8°C | Room temperature | |||
| Sizes | 50 slides 5 x 50 slides | 5 x 2 mL 2 mL 10 mL | 5 x 2 mL 2 mL 10 mL | 5 x 2 mL 2 mL 10 mL | |||
| User guide | User Guide: ProLong Glass Antifade Mountant | User Guide: ProLong Gold and Diamond Antifade Mountants | |||||
*All objective types are compatible with these reagents
Excellent image resolution in less time with ProLong RapidSet mountant
In microscopy, the refractive index (RI) of a material determines how much light is bent or refracted when it enters that material. Significant differences in RI between the specimen and the mounting medium can cause optical distortions. To minimize these distortions, it is crucial to reduce the RI differences between the specimen and the mounting medium.
ProLong RapidSet antifade is a new hard-setting mountant with the ability to be fully cured within 1 hour, while approaching the refractive index and archive-ability of ProLong Glass. Unlike other mountants that require several hours to days to harden, ProLong RapidSet mountant can achieve an RI of 1.49 within 1 hour (Figure 1, 2), allowing for same-day imaging without sacrificing image quality.
Figure 1. ProLong RapidSet mountant achieves an RI of 1.49 within 1 hour, faster than other mountants that harden over several days. Comparison of refractive index (RI) vs. time for ProLong RapidSet antifade mountant and other mounting media was conducted over 4 days using an Abbe Refractometer (589 nm) with a closed prism to simulate hardening after cover slipping. While most mounting media harden over several days via water evaporation, ProLong RapidSet mountant hardens in 1 hour with an initial RI near 1.49, higher than most other hardening mountants. If water evaporates from mounted samples, it achieves an RI of 1.52, excellent for high-resolution fluorescence imaging with crown glass coverslips.

Figure 2. Fast hardening with ProLong RapidSet antifade mountant for improved image resolution in a fraction of the time. Free-floating cryopreserved rat brain sections (100 µm thick) stained with NucBlue Fixed Cell ReadyProbes Reagent (DAPI) and mounted in A) ProLong Glass antifade mountant hardened overnight using protocol Option C, B) ProLong Glass antifade mountant imaged 3 hours after mounting, and C) ProLong RapidSet antifade mountant imaged 3 hours after mounting. Refractive Index (RI) values approximated from refractometry measurements and rounded to the nearest one hundredth. (A) ProLong Glass antifade mountant (RI = 1.52) enables high-resolution imaging in thick samples when fully hardened, which takes 16 to 72 hours via water evaporation. (B) Imaging a sample in ProLong Glass antifade mountant (initial RI = 1.38) after three hours of hardening shows decreased fluorescence beyond 30 microns due to RI mismatch. (C) ProLong RapidSet antifade mountant (RI ~1.49) hardens in 1 hour, offering better RI matching. A sample imaged after three hours in ProLong RapidSet mountant shows improved resolution beyond 30 microns compared to (B), though not as high as (A) due to slight RI mismatch (1.49 vs. 1.52). Samples imaged with a Zeiss™ LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss™ Zen software.
Performance of ProLong antifade mountants for fixed cells
Increase optical clarity for sharp, bright images
Matching the RI of the mountant to the imaging system is critical for achieving high-resolution imaging in microscopy. By aligning the refractive index of the mountant with that of the imaging system, light rays can pass from the sample through the mountant and into the imaging system without significant refraction or reflection. This alignment of refractive indices minimizes light distortion and preserves the integrity of the captured images, helping ensure that any mismatch in refractive indices does not compromise the final image quality. Effective matching of the RI with products like ProLong RapidSet or ProLong Glass reagents (which offer RI values of ~1.52) facilitates high-resolution imaging, enabling clear, detailed, and accurate visualization of structures within the sample (Figure 3).

Figure 3. Matching the refractive index (RI) of the mountant to the imaging system is crucial for achieving high-resolution imaging. Free-floating cryopreserved rat brain sections (100 µm thick) stained with NucBlue Fixed Cell ReadyProbes Reagent (DAPI) and mounted in (A)ProLong Glass antifade mountant hardened overnight using protocol Option C, (B)ProLong RapidSet antifade mountant imaged 3 hours after mounting and (C) VectaShield® Vibrance® Antifade Mounting Media hardened for four days. Refractive Index (RI) values approximated from refractometry measurements and rounded to the nearest one hundredth. (A) ProLong Glass antifade mountant (RI = 1.52) exhibits high-resolution imaging in thick samples when fully hardened, which takes 16 to 72 hours via water evaporation. (B) ProLong RapidSet mountant (RI ~1.49) hardens in 1 hour, offering better RI matching. A sample imaged after just three hours in RapidSet mountant shows improved resolution beyond 30 microns compared to (B), though not as high as (A) due to slight RI mismatch (1.49 vs. 1.52). (C) A sample mounted VectaShield® Vibrance® Antifade Mounting Media (RI = 1.46) after extended hardening shows decreased fluorescence beyond 30 microns due to RI mismatch. Samples imaged with a Zeiss™ LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss™ Zen software.
Minimize photobleaching, increase fluorescent intensity
In fluorescence microscopy imaging, photobleaching is a common occurrence where fluorescent molecules lose their fluorescence when exposed to light. This can lead to a reduction in signal intensity and can compromise the quality of the resulting images. Photobleaching can be minimized by using antifade mountants such as ProLong RapidSet, ProLong Glass and ProLong Diamond reagents. These antifade mountants stabilize the fluorophores and preserve signal strength over prolonged imaging sessions (Figure 4, 5). By reducing the effects of photobleaching, these mountants help ensure clearer and more reliable imaging results.
Figure 4. Antifade protection for improved photostability during fluorescence imaging. Cultured cells stained with Fluorescein (FITC) Phalloidin Labeling Probe and mounted in ProLong Glass, ProLong Diamond and ProLong Rapidset mountants were photobleached by constant illumination for one minute under exposure matched light settings using an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. Data is plotted as %Fluorescence retained (fluorescence at tn/fluorescence at t0).
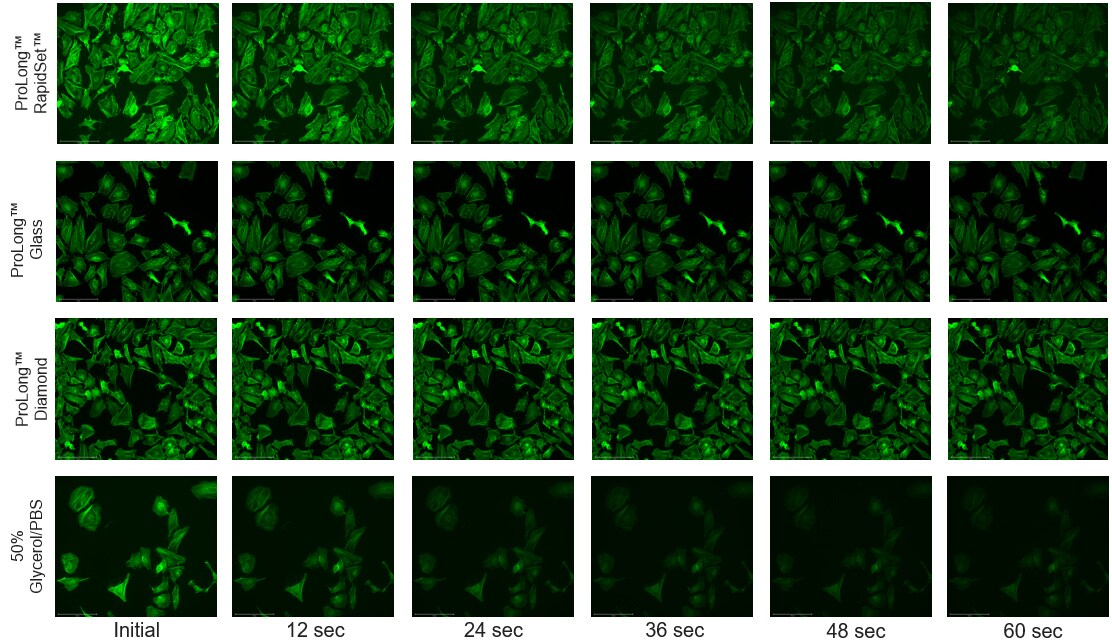
Figure 5. A 60-second time-lapse showing the enhanced resistance to photobleaching afforded by ProLong antifade mountants.Fluorescein (FITC) Phalloidin Labeling Probe stained cells mounted in ProLong Glass, ProLong Diamond, and ProLong RapidSet antifade mountants were imaged for 60 seconds with constant illumination using an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. Images taken at 12 second intervals are compared.
Signal preservation across the spectrum
ProLong antifade mountants offer low background and protection against photobleaching across the visible and near-infrared spectrum in addition to exhibiting less initial quenching of the fluorescent signal (Table 1, Figure 6). They also help provide extra protection for traditional dyes such as FITC and fluorescent proteins such as GFP.
Table 1. Photobleaching resistance across the spectrum.
| Fluorophore | Ex/Em (nm) | Fluorophore performance* | ||
|---|---|---|---|---|
| ProLong RapidSet | ProLong Glass | ProLong Diamond | ||
| Alexa Fluor 405 | 400/424 | ++ | + | +++ |
| Alexa Fluor Plus 405 | 405/450 | ++ | + | +++ |
| DAPI | 345/455 | +++ | +++ | +++ |
| Hoechst 33342 | 350/461 | ++ | +++ | +++ |
| Alexa Fluor 420 | 420/500 | ++ | ++ | ++ |
| emGFP | 488/510 | +++ | +++ | +++ |
| Fluorescein | 494/518 | +++ | +++ | +++ |
| Alexa Fluor 488 | 495/519 | +++ | +++ | +++ |
| Alexa Fluor Plus 488 | 495/519 | +++ | +++ | +++ |
| Alexa Fluor 514 | 518/543 | +++ | +++ | +++ |
| Alexa Fluor 555 | 555/565 | +++ | +++ | +++ |
| Alexa Fluor Plus 555 | 555/565 | +++ | +++ | +++ |
| Cy3 | 550/570 | +++ | ++ | ++ |
| Alexa Fluor 546 | 556/575 | +++ | ++ | ++ |
| Tetramethylrhodamine | 555/580 | +++ | ++ | +++ |
| TagRFP | 555/584 | +++ | +++ | ++ |
| Alexa Fluor 568 | 578/603 | +++ | +++ | +++ |
| mCherry | 575/610 | +++ | +++ | +++ |
| Texas Red | 595/615 | +++ | +++ | +++ |
| Alexa Fluor 594 | 590/617 | +++ | +++ | +++ |
| Alexa Fluor Plus 594 | 590/617 | +++ | ++ | +++ |
| TO-PRO-3 | 642/661 | ++ | +++ | +++ |
| Alexa Fluor 647 | 652/668 | ++ | ++ | +++ |
| Alexa Fluor Plus 647 | 652/668 | ++ | ++ | +++ |
| Cy5 | 650/670 | ++ | +++ | +++ |
| SYTOX Deep Red | 660/682 | ++ | +++ | ++ |
| Alexa Fluor 700 | 702/723 | +++ | +++ | +++ |
| Alexa Fluor Plus 750 | 760/788 | +++ | ++ | + |
| Key: +++ = Best performance, ++ = Better performance, + = Good performance.*Metrics including fluorescence retention, fluorescence signal-to-background, and labeling/sample appearance were used to rate fluorophore performance. | ||||

Figure 6. Formulated to protect fluorescent dyes and proteins across the spectrum. Cells stained with Alexa Fluor 488 Phalloidin Labeling Probe (Green), mouse anti-tublin primary antibody detected with Alexa Fluor 647 Goat anti-Mouse Secondary Antibody (Red), and NucBlue Fixed Cell ReadyProbes Reagent (Blue, DAPI), were mounted in ProLong Glass antifade mountant, ProLong Diamond antifade mountant and ProLong RapidSet antifade mountant, alongside VectaShield® Vibrance® and HardSet™ Antifade Mounting Media products and imaged using equivalent exposure settings on an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. ProLong antifade mountants are compatible with various fluorescent proteins and dye chemistries to reduce photobleaching and prevent fluorescence quenching before imaging. Note that Alexa Fluor dye 647 loses fluorescence in VectaShield® HardSet™ Antifade Mounting Media due to incompatibility.
Excellent axial resolution of ProLong Glass mountant
ProLong Glass antifade mountant features a RI of approximately 1.52, which is similar to that of cover glass, immersion oil, and oil objective optics. This exceptional optical clarity means ProLong Glass reagent delivers up to 75% improvement in axial resolution and 3–4 times more imageable focal depth compared to mountants with refractive index of 1.47 (Figure 7), producing sharp images for monolayers of cells, tissue slices, or 3D cell cultures up to 150 μm thickness/focal depth (Figure 8).
Figure 8. Axial and lateral confocal images of A549 spheroid mounted with ProLong Glass antifade mountant. A549 spheroid grown in Nunclon Sphera microplates at 100 cells/well were labeled with NucBlue Live ReadyProbes Reagent, MitoTracker Orange, and CellROX Deep Red Reagent for oxidative stress detection for 1 hour at 37oC. Cells were then fixed with 4% PFA, transferred to a #1.5 coverslip, and mounted in ProLong Glass. Sample was allowed to cure overnight exposed to air, and the coverslip was mounted to the slide using 100% glycerol. Image acquisition and analysis was performed using a Zeiss LSM710 Confocal running Zen software.
ProLong live antifade reagent comes in 100 x concentration liquid that can be used on live cells and tissues.
| ProLong Live antifade reagent | |
| Imaging type | Live |
| Optimal microscope objective* | Glycerol-corrected Water- or air-corrected |
| Refractive index | 1.3 |
| Incubation time | 15 min to 2 hours (2 hours recommended) |
| Compatible fluorophores | All dyes and fluorescent proteins |
| Reagent storage temperature | 2–8°C (up to 30 days) ≤–20°C (up to 6 months) |
| Sizes | 5 x 1 mL 1 mL |
| User guide | UserGuide: ProLong Live Antifade Reagent |
*All objective types are compatible with these reagents
Performance of ProLong antifade reagents for live cells
Minimize photobleaching, increase signal intensity
ProLong Live antifade reagent helps to prevent photobleaching by reducing the photochemical reactions that lead to loss of fluorescence during live cell imaging. ProLong Live reagent contains enzymes that metabolize free radical singlet oxygen that is involved in degrading dye molecules, resulting in photobleaching. By stabilizing the fluorescent dyes or proteins in live cells, ProLong Live antifade reagent helps to preserve the fluorescence signal over time (Figure 9). This enables researchers longer and clearer live cell imaging experiments with high-quality images.

Figure 9. Photobleach protection using ProLong Live antifade reagent. HeLa cells were transduced with CellLight Mitochondria-GFP (green) for 24 hours, then labeled with NucBlue Live ReadyProbes Reagent (Hoechst 33342 dye, blue) for 15 minutes. ProLong Live antifade reagent was added to one sample and incubated for 2 hours. Samples incubated with ProLong Live antifade reagent (top panel) retained more signal at all time points, compared to control samples in complete medium alone (bottom panel).
Preserve signal across the spectrum
ProLong Live antifade reagent is compatible with a wide range of fluorescence, making it versatile for various experimental setups and applications. Moreover, ProLong Live reagent can preserve the signals of fluorescent proteins, functional probes like MitoTracker dyes, and counterstains such as Hoechst 33342 for extended imaging protocols and more accurate measurements (shown above in Figure 9, Figure 10).
Figure 10. Effective photobleaching protection with multiple fluorophores. HeLa or U2OS cells expressing fluorescent proteins (CellLight MitoGFP and MitoRFP) or treated with fluorescent dyes (MitoTracker Green, Red, or Deep Red or Hoechst 33342 dye) were treated with ProLong Live reagent and scanned using a high-content analysis instrument in confocal mode. The degree of photobleaching protection is expressed as the fraction of initial fluorescence intensity remaining following repeat exposures. EC50 (half-maximal fluorescence signal) is indicated for each graph, and the overall signal protection offered by ProLong Live reagent (compared to untreated samples) is calculated based on the scan number where treated and untreated samples reach the EC50 value. The addition of ProLong Live reagent permitted (A) 50% more captures with CellLight MitoGFP (EmGFP); (B) 100% more captures with CellLight MitoRFP (TagRFP); (C) 120% more captures with MitoTracker Green; (D) 67% more captures with MitoTracker Red; (E) 85% more captures with MitoTracker Deep Red; (F) More captures (amount not calculated) with Hoechst 33342 dye.
Maintain cellular viability and proliferation
ProLong Live antifade reagent is formulated to be gentle on live cells, allowing for extended imaging without compromising cell viability or proliferation (Figure 11). This feature is crucial for long-term imaging experiments where cell health and functionality need to be maintained.
Figure 11. Cell viability and proliferation are not affected by ProLong Live reagent treatment. HeLa cells were plated at a concentration of 1,000 cells/well in a 96-well plate and treated with a working concentration of ProLong Live antifade reagent for the period indicated. Cell viability was detected using (A)LIVE/DEAD Red dye, (B)CyQUANT Direct reagent, and (C)PrestoBlue reagent. Cell proliferation was detected with (D)Click-iT Plus EdU reagent. Error bars = standard deviation.
ProLong antifade mountants are ready-to-use, hard-setting reagents used for the long-term imaging of fixed animal cells and tissues.
| NEWProLong RapidSet antifade mountant | ProLong Glass | ProLong Diamond | ProLong Gold | ||||
| ProLong Glass antifade mountant | ProLong Glass antifade mountant with NucBlue stain | ProLong Diamond antifade mountant | ProLong Diamond antifade mountant with DAPI | ProLong Gold antifade mountant | ProLong Gold antifade mountant with DAPI | ||
| Curing time | 1 hour | 18–60 hours (depending on specimen thickness) | 24 hours | 24 hours | |||
|---|---|---|---|---|---|---|---|
| Sample thickness | Up to 80 µm sample thickness | Up to 150 µm sample thickness | Up to 10 µm sample thickness | Up to 10 µm sample thickness | |||
| Refractive index (after curing) | 1.49 (after 1 hr curing) RI 1.52 (after 24 hr curing) | 1.52 (after 24 hr curing) | 1.47 (after 24 hr curing) | 1.47 (after 24 hr curing) | |||
| Sample storage temperature | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | Room temperature 4°C ≤-20°C (optimal) | |||
| Optimal microscope objective* | Oil-immersion | Oil-immersion | Glycerol-corrected | Glycerol-corrected | |||
| Compatible fluorophores | Most dyes and fluorescent proteins | Most dyes and fluorescent proteins | Most dyes and fluorescent proteins | Alexa Fluor dyes | |||
| Reagent storage temperature | 2-8°C | 2-8°C | 2-8°C | Room temperature | |||
| Sizes | 50 slides 5 x 50 slides | 5 x 2 mL 2 mL 10 mL | 5 x 2 mL 2 mL 10 mL | 5 x 2 mL 2 mL 10 mL | |||
| User guide | User Guide: ProLong Glass Antifade Mountant | User Guide: ProLong Gold and Diamond Antifade Mountants | |||||
*All objective types are compatible with these reagents
Excellent image resolution in less time with ProLong RapidSet mountant
In microscopy, the refractive index (RI) of a material determines how much light is bent or refracted when it enters that material. Significant differences in RI between the specimen and the mounting medium can cause optical distortions. To minimize these distortions, it is crucial to reduce the RI differences between the specimen and the mounting medium.
ProLong RapidSet antifade is a new hard-setting mountant with the ability to be fully cured within 1 hour, while approaching the refractive index and archive-ability of ProLong Glass. Unlike other mountants that require several hours to days to harden, ProLong RapidSet mountant can achieve an RI of 1.49 within 1 hour (Figure 1, 2), allowing for same-day imaging without sacrificing image quality.
Figure 1. ProLong RapidSet mountant achieves an RI of 1.49 within 1 hour, faster than other mountants that harden over several days. Comparison of refractive index (RI) vs. time for ProLong RapidSet antifade mountant and other mounting media was conducted over 4 days using an Abbe Refractometer (589 nm) with a closed prism to simulate hardening after cover slipping. While most mounting media harden over several days via water evaporation, ProLong RapidSet mountant hardens in 1 hour with an initial RI near 1.49, higher than most other hardening mountants. If water evaporates from mounted samples, it achieves an RI of 1.52, excellent for high-resolution fluorescence imaging with crown glass coverslips.

Figure 2. Fast hardening with ProLong RapidSet antifade mountant for improved image resolution in a fraction of the time. Free-floating cryopreserved rat brain sections (100 µm thick) stained with NucBlue Fixed Cell ReadyProbes Reagent (DAPI) and mounted in A) ProLong Glass antifade mountant hardened overnight using protocol Option C, B) ProLong Glass antifade mountant imaged 3 hours after mounting, and C) ProLong RapidSet antifade mountant imaged 3 hours after mounting. Refractive Index (RI) values approximated from refractometry measurements and rounded to the nearest one hundredth. (A) ProLong Glass antifade mountant (RI = 1.52) enables high-resolution imaging in thick samples when fully hardened, which takes 16 to 72 hours via water evaporation. (B) Imaging a sample in ProLong Glass antifade mountant (initial RI = 1.38) after three hours of hardening shows decreased fluorescence beyond 30 microns due to RI mismatch. (C) ProLong RapidSet antifade mountant (RI ~1.49) hardens in 1 hour, offering better RI matching. A sample imaged after three hours in ProLong RapidSet mountant shows improved resolution beyond 30 microns compared to (B), though not as high as (A) due to slight RI mismatch (1.49 vs. 1.52). Samples imaged with a Zeiss™ LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss™ Zen software.
Performance of ProLong antifade mountants for fixed cells
Increase optical clarity for sharp, bright images
Matching the RI of the mountant to the imaging system is critical for achieving high-resolution imaging in microscopy. By aligning the refractive index of the mountant with that of the imaging system, light rays can pass from the sample through the mountant and into the imaging system without significant refraction or reflection. This alignment of refractive indices minimizes light distortion and preserves the integrity of the captured images, helping ensure that any mismatch in refractive indices does not compromise the final image quality. Effective matching of the RI with products like ProLong RapidSet or ProLong Glass reagents (which offer RI values of ~1.52) facilitates high-resolution imaging, enabling clear, detailed, and accurate visualization of structures within the sample (Figure 3).

Figure 3. Matching the refractive index (RI) of the mountant to the imaging system is crucial for achieving high-resolution imaging. Free-floating cryopreserved rat brain sections (100 µm thick) stained with NucBlue Fixed Cell ReadyProbes Reagent (DAPI) and mounted in (A)ProLong Glass antifade mountant hardened overnight using protocol Option C, (B)ProLong RapidSet antifade mountant imaged 3 hours after mounting and (C) VectaShield® Vibrance® Antifade Mounting Media hardened for four days. Refractive Index (RI) values approximated from refractometry measurements and rounded to the nearest one hundredth. (A) ProLong Glass antifade mountant (RI = 1.52) exhibits high-resolution imaging in thick samples when fully hardened, which takes 16 to 72 hours via water evaporation. (B) ProLong RapidSet mountant (RI ~1.49) hardens in 1 hour, offering better RI matching. A sample imaged after just three hours in RapidSet mountant shows improved resolution beyond 30 microns compared to (B), though not as high as (A) due to slight RI mismatch (1.49 vs. 1.52). (C) A sample mounted VectaShield® Vibrance® Antifade Mounting Media (RI = 1.46) after extended hardening shows decreased fluorescence beyond 30 microns due to RI mismatch. Samples imaged with a Zeiss™ LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss™ Zen software.
Minimize photobleaching, increase fluorescent intensity
In fluorescence microscopy imaging, photobleaching is a common occurrence where fluorescent molecules lose their fluorescence when exposed to light. This can lead to a reduction in signal intensity and can compromise the quality of the resulting images. Photobleaching can be minimized by using antifade mountants such as ProLong RapidSet, ProLong Glass and ProLong Diamond reagents. These antifade mountants stabilize the fluorophores and preserve signal strength over prolonged imaging sessions (Figure 4, 5). By reducing the effects of photobleaching, these mountants help ensure clearer and more reliable imaging results.
Figure 4. Antifade protection for improved photostability during fluorescence imaging. Cultured cells stained with Fluorescein (FITC) Phalloidin Labeling Probe and mounted in ProLong Glass, ProLong Diamond and ProLong Rapidset mountants were photobleached by constant illumination for one minute under exposure matched light settings using an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. Data is plotted as %Fluorescence retained (fluorescence at tn/fluorescence at t0).

Figure 5. A 60-second time-lapse showing the enhanced resistance to photobleaching afforded by ProLong antifade mountants.Fluorescein (FITC) Phalloidin Labeling Probe stained cells mounted in ProLong Glass, ProLong Diamond, and ProLong RapidSet antifade mountants were imaged for 60 seconds with constant illumination using an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. Images taken at 12 second intervals are compared.
Signal preservation across the spectrum
ProLong antifade mountants offer low background and protection against photobleaching across the visible and near-infrared spectrum in addition to exhibiting less initial quenching of the fluorescent signal (Table 1, Figure 6). They also help provide extra protection for traditional dyes such as FITC and fluorescent proteins such as GFP.
Table 1. Photobleaching resistance across the spectrum.
| Fluorophore | Ex/Em (nm) | Fluorophore performance* | ||
|---|---|---|---|---|
| ProLong RapidSet | ProLong Glass | ProLong Diamond | ||
| Alexa Fluor 405 | 400/424 | ++ | + | +++ |
| Alexa Fluor Plus 405 | 405/450 | ++ | + | +++ |
| DAPI | 345/455 | +++ | +++ | +++ |
| Hoechst 33342 | 350/461 | ++ | +++ | +++ |
| Alexa Fluor 420 | 420/500 | ++ | ++ | ++ |
| emGFP | 488/510 | +++ | +++ | +++ |
| Fluorescein | 494/518 | +++ | +++ | +++ |
| Alexa Fluor 488 | 495/519 | +++ | +++ | +++ |
| Alexa Fluor Plus 488 | 495/519 | +++ | +++ | +++ |
| Alexa Fluor 514 | 518/543 | +++ | +++ | +++ |
| Alexa Fluor 555 | 555/565 | +++ | +++ | +++ |
| Alexa Fluor Plus 555 | 555/565 | +++ | +++ | +++ |
| Cy3 | 550/570 | +++ | ++ | ++ |
| Alexa Fluor 546 | 556/575 | +++ | ++ | ++ |
| Tetramethylrhodamine | 555/580 | +++ | ++ | +++ |
| TagRFP | 555/584 | +++ | +++ | ++ |
| Alexa Fluor 568 | 578/603 | +++ | +++ | +++ |
| mCherry | 575/610 | +++ | +++ | +++ |
| Texas Red | 595/615 | +++ | +++ | +++ |
| Alexa Fluor 594 | 590/617 | +++ | +++ | +++ |
| Alexa Fluor Plus 594 | 590/617 | +++ | ++ | +++ |
| TO-PRO-3 | 642/661 | ++ | +++ | +++ |
| Alexa Fluor 647 | 652/668 | ++ | ++ | +++ |
| Alexa Fluor Plus 647 | 652/668 | ++ | ++ | +++ |
| Cy5 | 650/670 | ++ | +++ | +++ |
| SYTOX Deep Red | 660/682 | ++ | +++ | ++ |
| Alexa Fluor 700 | 702/723 | +++ | +++ | +++ |
| Alexa Fluor Plus 750 | 760/788 | +++ | ++ | + |
| Key: +++ = Best performance, ++ = Better performance, + = Good performance.*Metrics including fluorescence retention, fluorescence signal-to-background, and labeling/sample appearance were used to rate fluorophore performance. | ||||

Figure 6. Formulated to protect fluorescent dyes and proteins across the spectrum. Cells stained with Alexa Fluor 488 Phalloidin Labeling Probe (Green), mouse anti-tublin primary antibody detected with Alexa Fluor 647 Goat anti-Mouse Secondary Antibody (Red), and NucBlue Fixed Cell ReadyProbes Reagent (Blue, DAPI), were mounted in ProLong Glass antifade mountant, ProLong Diamond antifade mountant and ProLong RapidSet antifade mountant, alongside VectaShield® Vibrance® and HardSet™ Antifade Mounting Media products and imaged using equivalent exposure settings on an EVOS M7000 Imaging System equipped with a 20x air coverslip corrected objective. ProLong antifade mountants are compatible with various fluorescent proteins and dye chemistries to reduce photobleaching and prevent fluorescence quenching before imaging. Note that Alexa Fluor dye 647 loses fluorescence in VectaShield® HardSet™ Antifade Mounting Media due to incompatibility.
Excellent axial resolution of ProLong Glass mountant
ProLong Glass antifade mountant features a RI of approximately 1.52, which is similar to that of cover glass, immersion oil, and oil objective optics. This exceptional optical clarity means ProLong Glass reagent delivers up to 75% improvement in axial resolution and 3–4 times more imageable focal depth compared to mountants with refractive index of 1.47 (Figure 7), producing sharp images for monolayers of cells, tissue slices, or 3D cell cultures up to 150 μm thickness/focal depth (Figure 8).
Figure 8. Axial and lateral confocal images of A549 spheroid mounted with ProLong Glass antifade mountant. A549 spheroid grown in Nunclon Sphera microplates at 100 cells/well were labeled with NucBlue Live ReadyProbes Reagent, MitoTracker Orange, and CellROX Deep Red Reagent for oxidative stress detection for 1 hour at 37oC. Cells were then fixed with 4% PFA, transferred to a #1.5 coverslip, and mounted in ProLong Glass. Sample was allowed to cure overnight exposed to air, and the coverslip was mounted to the slide using 100% glycerol. Image acquisition and analysis was performed using a Zeiss LSM710 Confocal running Zen software.
ProLong live antifade reagent comes in 100 x concentration liquid that can be used on live cells and tissues.
| ProLong Live antifade reagent | |
| Imaging type | Live |
| Optimal microscope objective* | Glycerol-corrected Water- or air-corrected |
| Refractive index | 1.3 |
| Incubation time | 15 min to 2 hours (2 hours recommended) |
| Compatible fluorophores | All dyes and fluorescent proteins |
| Reagent storage temperature | 2–8°C (up to 30 days) ≤–20°C (up to 6 months) |
| Sizes | 5 x 1 mL 1 mL |
| User guide | UserGuide: ProLong Live Antifade Reagent |
*All objective types are compatible with these reagents
Performance of ProLong antifade reagents for live cells
Minimize photobleaching, increase signal intensity
ProLong Live antifade reagent helps to prevent photobleaching by reducing the photochemical reactions that lead to loss of fluorescence during live cell imaging. ProLong Live reagent contains enzymes that metabolize free radical singlet oxygen that is involved in degrading dye molecules, resulting in photobleaching. By stabilizing the fluorescent dyes or proteins in live cells, ProLong Live antifade reagent helps to preserve the fluorescence signal over time (Figure 9). This enables researchers longer and clearer live cell imaging experiments with high-quality images.

Figure 9. Photobleach protection using ProLong Live antifade reagent. HeLa cells were transduced with CellLight Mitochondria-GFP (green) for 24 hours, then labeled with NucBlue Live ReadyProbes Reagent (Hoechst 33342 dye, blue) for 15 minutes. ProLong Live antifade reagent was added to one sample and incubated for 2 hours. Samples incubated with ProLong Live antifade reagent (top panel) retained more signal at all time points, compared to control samples in complete medium alone (bottom panel).
Preserve signal across the spectrum
ProLong Live antifade reagent is compatible with a wide range of fluorescence, making it versatile for various experimental setups and applications. Moreover, ProLong Live reagent can preserve the signals of fluorescent proteins, functional probes like MitoTracker dyes, and counterstains such as Hoechst 33342 for extended imaging protocols and more accurate measurements (shown above in Figure 9, Figure 10).
Figure 10. Effective photobleaching protection with multiple fluorophores. HeLa or U2OS cells expressing fluorescent proteins (CellLight MitoGFP and MitoRFP) or treated with fluorescent dyes (MitoTracker Green, Red, or Deep Red or Hoechst 33342 dye) were treated with ProLong Live reagent and scanned using a high-content analysis instrument in confocal mode. The degree of photobleaching protection is expressed as the fraction of initial fluorescence intensity remaining following repeat exposures. EC50 (half-maximal fluorescence signal) is indicated for each graph, and the overall signal protection offered by ProLong Live reagent (compared to untreated samples) is calculated based on the scan number where treated and untreated samples reach the EC50 value. The addition of ProLong Live reagent permitted (A) 50% more captures with CellLight MitoGFP (EmGFP); (B) 100% more captures with CellLight MitoRFP (TagRFP); (C) 120% more captures with MitoTracker Green; (D) 67% more captures with MitoTracker Red; (E) 85% more captures with MitoTracker Deep Red; (F) More captures (amount not calculated) with Hoechst 33342 dye.
Maintain cellular viability and proliferation
ProLong Live antifade reagent is formulated to be gentle on live cells, allowing for extended imaging without compromising cell viability or proliferation (Figure 11). This feature is crucial for long-term imaging experiments where cell health and functionality need to be maintained.
Figure 11. Cell viability and proliferation are not affected by ProLong Live reagent treatment. HeLa cells were plated at a concentration of 1,000 cells/well in a 96-well plate and treated with a working concentration of ProLong Live antifade reagent for the period indicated. Cell viability was detected using (A)LIVE/DEAD Red dye, (B)CyQUANT Direct reagent, and (C)PrestoBlue reagent. Cell proliferation was detected with (D)Click-iT Plus EdU reagent. Error bars = standard deviation.
Ordering information
Image gallery ProLong antifade mountants for fixed cells

U2-OS cells mounted with ProLong RapidSet antifade mountant. U-2 OS cells labeled with NucBlue Fixed Cell ReadyProbes Reagent (Blue, DAPI), Alexa Fluor 488 Phalloidin Labeling Probe (Green), CellLight Mitochondria-RFP, BacMam 2.0 (Yellow), and immunostained with beta-Tubulin Monoclonal Antibody (2 28 33) detected with Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 647 (Red) then mounted in ProLong RapidSet antifade mountant. Samples imaged with a Zeiss LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss Zen software.

U2-OS cells mounted with ProLong RapidSet antifade mountant. U-2 OS cells labeled with NucBlue Fixed Cell ReadyProbes Reagent (Blue, DAPI), Alexa Fluor 488 Phalloidin Labeling Probe (Green), CellLight Mitochondria-RFP, BacMam 2.0 (Yellow), and immunostained with beta-Tubulin Monoclonal Antibody (2 28 33) detected with Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 647 (Red) then mounted in ProLong RapidSet antifade mountant. Sample was imaged on an EVOS M7000 Imaging System equipped with an Olympus 20X Objective, semi-apo, 0.5NA/2.1WD20x air objective.

U2-OS cells mounted with ProLong RapidSet antifade mountant. U-2 OS cells labeled with NucBlue Fixed Cell ReadyProbes Reagent (Blue, DAPI), Alexa Fluor 488 Phalloidin Labeling Probe (Green), and CellLight Mitochondria-RFP, BacMam 2.0 (Red) then mounted in ProLong RapidSet antifade mountant. Sample was imaged on an EVOS M7000 Imaging System equipped with an Olympus 40X Objective, fluorite, 0.60NA/2.7–4.0WD, correction collar (0–2.0 mm).

Deep tissue imaging with ProLong RapidSet antifade mountant. Cryo-preserved rat brain sections (100 µm thick), stained for tubulin (red) with Mouse Anti-Beta3-Tubulin and GFAP (yellow) Rabbit Anti-GFAP. Targets were detected with Alexa Fluor Plus 594 Goat Anti-Mouse and Alexa Fluor Plus 647 Goat Anti-Rabbit dyes. Nuclei (cyan) were stained with NucBlue fixed cell ReadyProbes Reagent (DAPI). Slides were mounted with ProLong RapidSet antifade mountant and imaged 3 hours just later with a Zeiss LSM 980 confocal microscope equipped with a Plan-Apochromat 63×/1.4 NA Oil immersion objective. Z-projections were generated using Zeiss™ Zen software

FFPE human intestine imaging with ProLong RapidSet antifade mountant. FFPE human small intestine section labeled with DAPI (Blue), Vimentin Monoclonal Antibody (V9) detected with Alexa Fluor Plus 488 Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody (Green), Cytokeratin 15 Polyclonal Antibody detected with Alexa Fluor Plus 647 Goat anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody (Red), and Alexa Fluor Plus 750 Antibody Alpha-Smooth Muscle Actin Monoclonal Antibody (1A4) (White) then mounted in ProLong RapidSet antifade mountant. The tissue section was scanned on the EVOS S1000 Spatial Imaging System at 20x magnification.

FFPE mouse brain imaging with ProLong RapidSet antifade mountant. FFPE mouse brain sagittal slice stained with DAPI (Blue), rabbit anti-GFAP primary antibody detected with Alexa Fluor Plus 488 Goat anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody (Green), MAP2 Monoclonal Antibody (AP18) detected with Alexa Fluor Plus 647 Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody (Red), and Alexa Fluor Plus 750 Antibody Alpha-Smooth Muscle Actin Monoclonal Antibody (1A4) (White) then mounted in ProLong RapidSet antifade mountant. The tissue section was scanned on the EVOS S1000 Spatial Imaging System at 20x magnification.

HeLa cells mounted with Prolong Glass antifade mountant. HeLa cells expressing CellLight Mitochondria-GFP were fixed and permeabilized. Tubulin was probed with Mouse Anti-beta-3 Tubulin Antibody and Alexa Fluor 647 Goat Anti-Mouse Secondary Antibody. Actin was stained with Alexa Fluor 594 Phalloidin and nuclei labeled with NucBlue Fixed Cell ReadyProbes Reagent. Cells were mounted with ProLong Glass antifade mountant and imaged on a Zeiss LSM 710 confocal microscope using a plan- apochromat 63X/1.4 NA oil immersion objective.

Tissue section mounted with ProLong Glass antifade mountant. Rat duodenal cryo section was stained with Histone H3 primary antibody, followed by goat anti-mouse Alexa Fluor 647 secondary antibody following standard IHC protocol. The tissue sections were then stained with CellMask Orange Actin Tracking Stain at 1X concentration and Hoechst 34580 for 1 hr. The tissue section was then mounted with ProLong Glass antifade mountant. Images were taken using an EVOS M7000 imaging system.

Cryo-preserved rat tissue section mounted with ProLong Glass antifade mountant. Cryo-preserved rat brain sections (100-µm thick) were stained for tubulin (red) with Mouse Anti-Beta3-Tubulin and GFAP (yellow) Rabbit Anti-GFAP. Targets were detected with Alexa Fluor Plus 594 Goat Anti-Mouse and Alexa Fluor Plus 647 Goat Anti-Rabbit. Nuclei (cyan) were stained with DAPI. Slides were mounted with ProLong Glass antifade mountant and imaged with a Zeiss LSM 710 confocal microscope using a plan- apochromat 63X/1.4 NA oil immersion objective.

Induced pluripotent stem cells (iPSCs) mounted with ProLong Diamond antifade mountant. iPSCs were cultured on a mouse embryonic feeder layer stained using Alexa Fluor dye-conjugated, live-cell qualified (sterile-filtered, bioburden tested, endotoxin-free) antibodies for fibroblast marker CD44 (green) and PSC marker TRA-1-60. Imaging was performed after fixing the cells and applying ProLong Diamond antifade mountant. Images were acquired on an EVOS FL Imaging System.

Neuronal stem cell (NSCs) mounted with ProLong Diamond antifade mountant. NSCs were generated from an iPSC line using Gibco PSC Neural Induction Medium. Cells were cultured on a Geltrex coated plate stained for the following NSC markers using the Human Neural Stem Cell Immunocytochemistry Kit: Nestin (green) and SOX2 (red) with DAPI (blue) nuclear DNA counterstaining. Images were acquired on an EVOS FL Imaging System.

HeLa cells mounted with ProLong Diamond antifade mountant. HeLa cells were fixed, permeabilized and blocked with BlockAid, stained with ActinGreen 488 Ready Probes Reagent, NucBlue Live ReadyProbes Reagent, and AlexaFluor 594 Goat Anti-Mouse IgG (H+L) Antibody against ATP Synthase, and mounted using ProLong Diamond mountant.

Neonatal human dermal fibroblasts (HDFn) cells mounted using ProLong Gold antifade mountant. HDFn cells were grown on coverslips, fixed with 4% formaldehyde, and permeabilized with 0.2% Triton X-100. Tubulin was detected with anti-tubulin-a (bovine) antibody (mouse IgG1, monoclonal 236-10501) and visualized using Alexa Fluor 594 goat anti-mouse IgG. Actin filaments were labeled with Alexa Fluor 488 phalloidin, and nuclei were stained with Hoechst 33342. Coverslips were mounted on slides using ProLong Gold antifade reagent. The multiple-exposure image was acquired on a Zeiss LSM 710 confocal microscope at 63x and 16-slice Z-stack maximum intensity projection.

Neonatal human dermal fibroblasts (HDFn) cells mounted with ProLong Gold antifade mountant. HDFn cells were fixed, permeabilized, and blocked using the Image-iTFixation/Permeabilization Kit. Golgi were labeled with an anti- golgin 97 antibody followed by detection with an Alexa Fluor 488 secondary. Cells were also stained with Alexa Fluor 594 phalloidin to label actin and NucBlue Fixed to label nuclei. Finally, cells were mounted in ProLong Gold antifade reagent.

BPAE cells mounted using ProLong Gold antifade mountant. BPAE cells were fixed, permeabilized, and blocked using the Image-iT Fixation/Permeabilization Kit. Mitochondria were labeled with an anti-CVIP antibody followed by detection with an Alexa Fluor 488 secondary. Cells were also stained with Alexa Fluor 594 phalloidin to label actin and NucBlue Fixed to label nuclei. Finally, cells were mounted in ProLong Gold antifade reagent.
Image gallery of ProLong antifade reagents for live cells

U2OS cells mounted with ProLong Live antifade reagent. U20S cells were transduced with CellLight Talin-GFP (green) and CellLight Golgi-RFP (orange) for 24 hours then labeled with MitoTracker Deep Red (purple) and NucBlue Live ReadyProbes Reagent (Hoechst 33342) (blue) for 15 minutes. For photobleach protection, the cells were incubated with ProLong Live antifade reagent for 90 minutes before imaging.

HeLa Cells mounted with ProLong Live antifade reagent. HeLa cells were transduced with CellLight Mitochondria-RFP (red) and CellLight Talin-GFP (green) for 24 hours then labeled with NucBlue Live ReadyProbes Reagent (Hoechst 33342) (blue) for 15 minutes. To protect cells from photobleaching, cells were incubated with ProLong Live antifade reagent for 90 minutes before imaging on EVOS Cell Imaging System.

Imaging protocols
Protocols that fit your needs in imaging ranging from sample and assay preparation to staining, labeling, and data analysis strategies.
Additionally, search through our library of online microscopy protocols.
Imaging tools
Find helpful tools below to help you plan your imaging experiments.
SpectraViewer Supports all levels of experimental complexity, use the tool to compare excitation and emission spectra of fluorophores and reagents.
Stain-iT Cell Staining Simulator Visualize staining your cell without wasting your reagents, antibodies, or time.
Cell Imaging Resource Center
Access articles, webinars and videos, experimental design, and more for imaging microscopy and high-content analysis
Fixed Cell Imaging
Utilize established tools and protocols to capture and analyze high-resolution images, facilitating the study of cellular structures and architectures
Live Cell Imaging
Examine cells in their natural state to maintain cellular functions allowing for the study of various cellular processes
Publications
BioProbes 73 article: ProLong Live Antifade Reagent: Protection from photobleaching for live-cell imaging
BioProbes 72 article: ProLong Diamond and SlowFade Diamond Antifade Mountants for fixed cell imaging
Posters
Antifade mounting media to help improve image quality in 3D biological samples
Support
Cell Imaging Support Center
Find technical information, tips and tricks, and troubleshooting help for your experimental problems
Cy™ is a trademark or registered trademark of GE Healthcare.
For Research Use Only. Not for use in diagnostic procedures.








